Terlipressin: A Game-Changer in Hepatorenal Syndrome Treatment and the Road Ahead
Abstract
Terlipressin has emerged as a pivotal treatment in managing Hepatorenal Syndrome (HRS), a severe complication of cirrhosis characterized by acute kidney injury (AKI). Recently approved by the FDA, terlipressin offers new hope for improving renal function and survival rates in HRS patients. By stimulating vasopressin receptors, it effectively increases renal blood flow and reduces portal pressure, making it the most effective vasoconstrictor available for HRS treatment. However, its use presents several challenges, including systemic inflammation, adverse ischemic events, and its impact on liver transplant candidacy via changes to the MELD score. Despite these obstacles, terlipressin remains a valuable tool in the therapeutic landscape of HRS, with ongoing research needed to refine patient selection, dosage regimens, and long-term outcomes. As clinicians and researchers continue to gather real-world data, terlipressin may redefine the management of this life-threatening condition.
Unraveling Hepatorenal Syndrome: A Complex Clinical Challenge
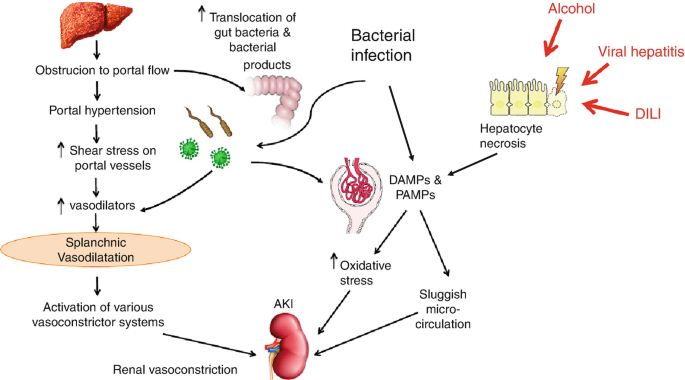
Hepatorenal Syndrome (HRS) is a life-threatening condition associated with advanced cirrhosis, where renal function deteriorates without apparent structural damage to the kidneys. It is classified as a form of acute kidney injury (AKI), occurring in 15-43% of hospitalized patients with cirrhosis and AKI. HRS arises from a significant reduction in renal blood flow due to splanchnic vasodilation and a decrease in effective arterial volume. The condition poses a serious challenge, with a reported in-hospital mortality rate of approximately 26%, and significant economic burdens associated with its management.
Until recently, the management of HRS relied on the use of vasoconstrictors, such as noradrenaline, octreotide, and midodrine, combined with intravenous albumin. These treatments aim to increase renal perfusion by constricting the splanchnic vasculature, yet response rates vary, and patient outcomes remain suboptimal. In this context, the FDA’s approval of terlipressin marks a significant milestone in the treatment of HRS. As a synthetic vasopressin analogue, terlipressin acts on V1 receptors in vascular smooth muscle to reverse HRS-AKI and reduce mortality rates. Its effectiveness, along with its widespread use in Europe and Asia, highlights its potential to transform HRS management in the U.S.
Terlipressin: Unlocking New Pathways in HRS Treatment
Terlipressin, a synthetic vasopressin analogue, has become the most effective vasoconstrictor for treating Hepatorenal Syndrome (HRS). Its primary mechanism of action involves stimulating V1 receptors located on vascular smooth muscle cells. This stimulation leads to vasoconstriction, particularly in the splanchnic vasculature, increasing renal blood flow and thereby reversing the effects of HRS. Terlipressin also reduces portal pressure, a crucial benefit for patients with cirrhosis. Unlike other vasoconstrictors, such as noradrenaline and midodrine, terlipressin has demonstrated superior effectiveness in clinical settings due to its ability to reduce serum creatinine levels and reverse acute kidney injury (AKI).
Terlipressin’s efficacy has been confirmed in several clinical trials, showing complete response rates of 33-81% among HRS patients. This complete response is characterized by a reduction in serum creatinine to less than 1.5 mg/dL on two consecutive measurements. The drug’s effectiveness is even more pronounced when treatment is initiated early, with patients showing significantly improved outcomes when baseline serum creatinine levels are relatively low. Additionally, terlipressin has been associated with improved transplant-free survival rates, offering patients a viable option while awaiting liver transplantation.
Navigating Therapeutic Obstacles: Challenges in Terlipressin Use
Despite terlipressin’s effectiveness in treating Hepatorenal Syndrome (HRS), its use presents several clinical challenges. One key challenge is late initiation of treatment. The efficacy of terlipressin diminishes significantly when treatment is delayed, particularly in patients with elevated baseline serum creatinine levels. For instance, the CONFIRM trial demonstrated a much lower response rate in patients whose mean serum creatinine was above 3.5 mg/dL. Early intervention is crucial for maximizing treatment benefits and reversing acute kidney injury (AKI).
Another challenge is the impact of systemic inflammation on terlipressin’s efficacy. In cases where HRS-AKI is accompanied by acute-on-chronic liver failure (ACLF), terlipressin’s ability to improve renal function is often diminished. ACLF involves multiple organ failures, and mortality rates can range from 20% to 80% depending on the severity of the condition. The systemic inflammation seen in ACLF may hinder the expected outcomes of terlipressin, especially in more advanced cases.
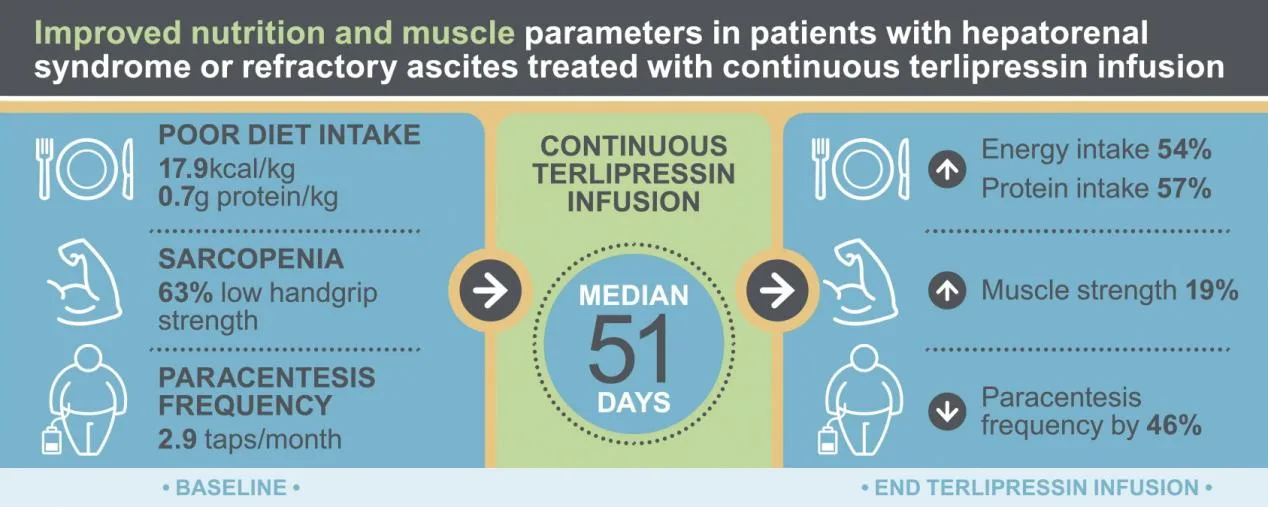
Furthermore, terlipressin’s influence on serum creatinine levels affects liver transplantation eligibility. As serum creatinine is a component of the Model for End-Stage Liver Disease (MELD) score, reducing creatinine levels can deprioritize patients on transplant lists, complicating their management.
Balancing Efficacy and Risk: Pitfalls of Terlipressin Therapy
While terlipressin offers substantial clinical benefits for Hepatorenal Syndrome (HRS), its use is associated with several risks and potential pitfalls. The most concerning adverse effects of terlipressin are related to ischemia, which can result in severe complications such as digital infarction, gut ischemia, cerebral ischemia, and even cardiac hypoperfusion leading to pulmonary edema. These risks are particularly pronounced in elderly patients and those with a history of peripheral, cardiac, or cerebrovascular disease.
Terlipressin’s vasoconstrictive mechanism can exacerbate underlying cardiovascular conditions. As a result, patients require thorough cardiovascular assessments, including electrocardiograms (ECGs) and echocardiography, before initiating treatment. Another significant risk is hyponatremia, which necessitates close monitoring of sodium levels during therapy. To mitigate these complications, continuous infusion of terlipressin, rather than bolus administration, has been suggested as a safer alternative.
Additionally, terlipressin is contraindicated in patients with grade 3 acute-on-chronic liver failure (ACLF), as these individuals are at high risk for developing fatal ischemic or respiratory complications. For these patients, the risks of terlipressin may outweigh its potential benefits, especially for those awaiting liver transplantation.
Looking Beyond: Future Prospects in HRS Management with Terlipressin
The recent FDA approval of terlipressin has transformed the management of Hepatorenal Syndrome (HRS), offering a more effective treatment option than previously available vasoconstrictors. While terlipressin has shown promising results in reversing acute kidney injury (AKI) and improving transplant-free survival, its use must be carefully considered in light of potential complications, including ischemic events and respiratory failure in patients with advanced liver failure. Early intervention and appropriate patient selection are critical to maximizing the drug’s benefits.
Looking ahead, further research is needed to refine treatment strategies for HRS patients, particularly those with acute-on-chronic liver failure (ACLF) and high MELD scores. Investigating the long-term effects of outpatient terlipressin infusions, as well as its role in bridging patients to liver transplantation, could significantly improve outcomes. Additionally, the medical community must address the impact of terlipressin on liver transplantation candidacy, especially with its influence on MELD scores.
Terlipressin represents a critical step forward in HRS treatment, but optimizing its use will require continued real-world data collection and strategic clinical approaches to mitigate risks while enhancing patient outcomes.
 LinkPeptide
LinkPeptide