Voclosporin: A Breakthrough Calcineurin Inhibitor in the Treatment of Lupus Nephritis
Abstract
Voclosporin, a next-generation calcineurin inhibitor, marks a significant breakthrough in the treatment of lupus nephritis (LN), a severe complication of systemic lupus erythematosus (SLE). Approved by the FDA in 2021, voclosporin works through dual mechanisms: modulating the immune response by inhibiting T-cell activation and protecting kidney function by stabilizing podocytes. Clinical trials have demonstrated its ability to achieve faster reductions in proteinuria and improve Complete Renal Response (CRR) rates when combined with standard therapies like mycophenolate mofetil and corticosteroids. Voclosporin’s favorable pharmacokinetics, including the absence of a need for therapeutic drug monitoring, further enhance its clinical appeal. With a manageable safety profile, voclosporin is poised to redefine the management of lupus nephritis and may also have potential in other immune-related conditions under investigation.
The Clinical Challenge of Lupus Nephritis: Scope and Impact
Lupus nephritis (LN) is a severe manifestation of systemic lupus erythematosus (SLE), a chronic autoimmune disease characterized by widespread inflammation. Approximately 40% of patients with SLE develop lupus nephritis, with a higher incidence in specific ethnic groups such as Asians, Blacks, and Hispanics. This complication significantly impacts patient outcomes, as LN leads to progressive kidney damage and is a major cause of morbidity and mortality in SLE patients. Studies indicate that up to 30% of LN patients progress to end-stage kidney disease (ESKD) within 15 years of diagnosis.
The clinical presentation of LN varies, ranging from asymptomatic hematuria or proteinuria to more severe forms such as nephrotic syndrome or acute kidney injury. Diagnosis typically requires renal biopsy, which allows classification of the disease into six histological classes. Treatment of lupus nephritis aims to reduce proteinuria and preserve kidney function, often involving immunosuppressive therapies such as mycophenolate mofetil (MMF), cyclophosphamide, and corticosteroids. Despite these treatments, response rates remain suboptimal, and many patients face challenges with relapses and drug toxicity.
Given these difficulties, there has been a significant need for novel therapies that offer better efficacy and safety profiles. Voclosporin, a recently approved calcineurin inhibitor, is one such therapy that promises improved outcomes for lupus nephritis patients.
Voclosporin’s Innovation: Mechanism and Development Pathway
Voclosporin is a novel calcineurin inhibitor (CNI) developed to address the limitations of older CNIs like cyclosporine and tacrolimus. It works by inhibiting calcineurin, a calcium-dependent phosphatase crucial for T-cell activation. This inhibition reduces cytokine production and suppresses immune responses, making voclosporin highly effective in treating lupus nephritis (LN), where immune system dysfunction leads to kidney damage. In addition to its immunosuppressive effects, voclosporin stabilizes kidney podocytes, preserving the integrity of the glomerular filtration barrier—a key factor in reducing proteinuria.
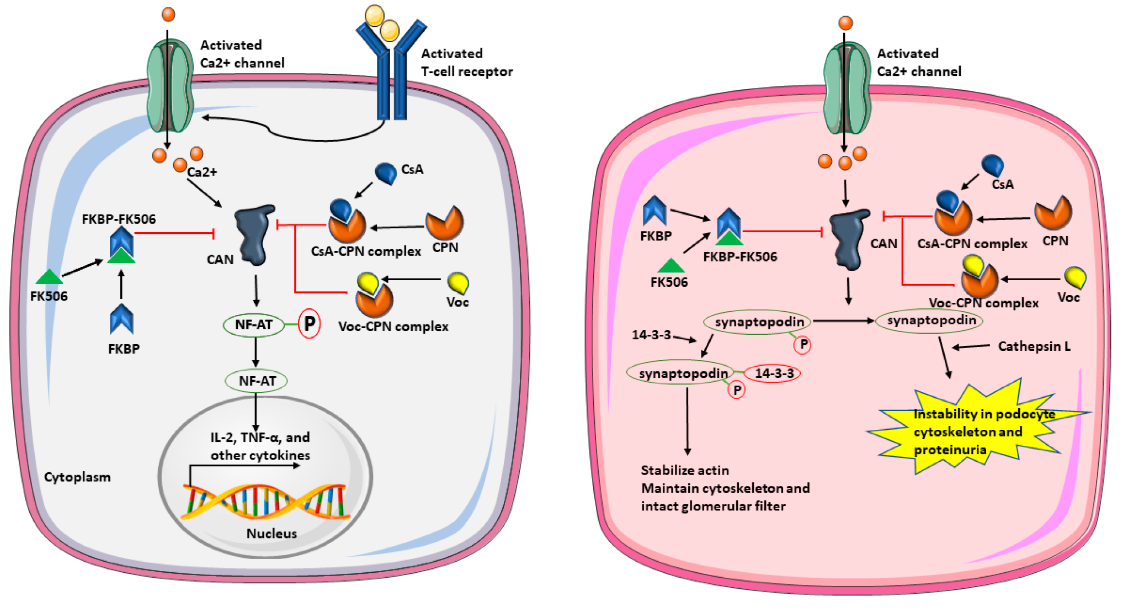
Voclosporin was specifically designed to improve both the efficacy and safety of traditional CNIs. While chemically similar to cyclosporine, it incorporates structural modifications that enhance potency and improve metabolic stability. A key modification at the amino acid-1 position increases pharmacokinetic predictability and eliminates the need for therapeutic drug monitoring. Furthermore, voclosporin demonstrates a more favorable lipid, glucose, and metabolic profile, helping to avoid complications such as nephrotoxicity, hypertension, and hyperlipidemia, which are commonly associated with older CNIs.
In 2021, voclosporin was approved by the FDA as an adjunct therapy for lupus nephritis. Its dual action—modulating immune responses and reducing proteinuria—marks a significant advancement over conventional treatments for LN.
Unveiling the Pharmacological Dynamics of Voclosporin
Voclosporin possesses distinct pharmacokinetic and pharmacodynamic characteristics that set it apart from traditional calcineurin inhibitors (CNIs). After oral administration, the drug is rapidly absorbed, with peak plasma levels reached within 1 to 4 hours. It follows a linear, dose-proportional pharmacokinetic profile, meaning that systemic exposure increases proportionally with the dose. Unlike older CNIs such as cyclosporine and tacrolimus, voclosporin does not require weight-based dosing, making its clinical use more straightforward.
Upon absorption, voclosporin binds extensively to plasma proteins (around 97%) and undergoes metabolism primarily via the liver enzyme CYP3A4. While the drug is metabolized into several compounds, the parent molecule remains the most pharmacologically active. Excretion is mainly fecal, with minimal urinary clearance, which reduces the renal burden—a notable advantage for patients with kidney involvement.
Pharmacodynamically, voclosporin inhibits calcineurin in a dose-dependent manner, preventing the activation of nuclear factor of activated T-cells (NFAT). This blockade suppresses the transcription of pro-inflammatory cytokines, curbing T-cell proliferation. In addition to its immunosuppressive effects, voclosporin stabilizes kidney podocytes, preventing protein leakage and thereby reducing proteinuria in lupus nephritis patients. These dual effects—immune regulation and kidney protection—position voclosporin as a highly effective treatment for lupus nephritis.
Overall, its favorable pharmacokinetics and reliable calcineurin inhibition have been crucial to voclosporin’s success in clinical practice.
Voclosporin’s Safety Landscape and Emerging Therapeutic Horizons
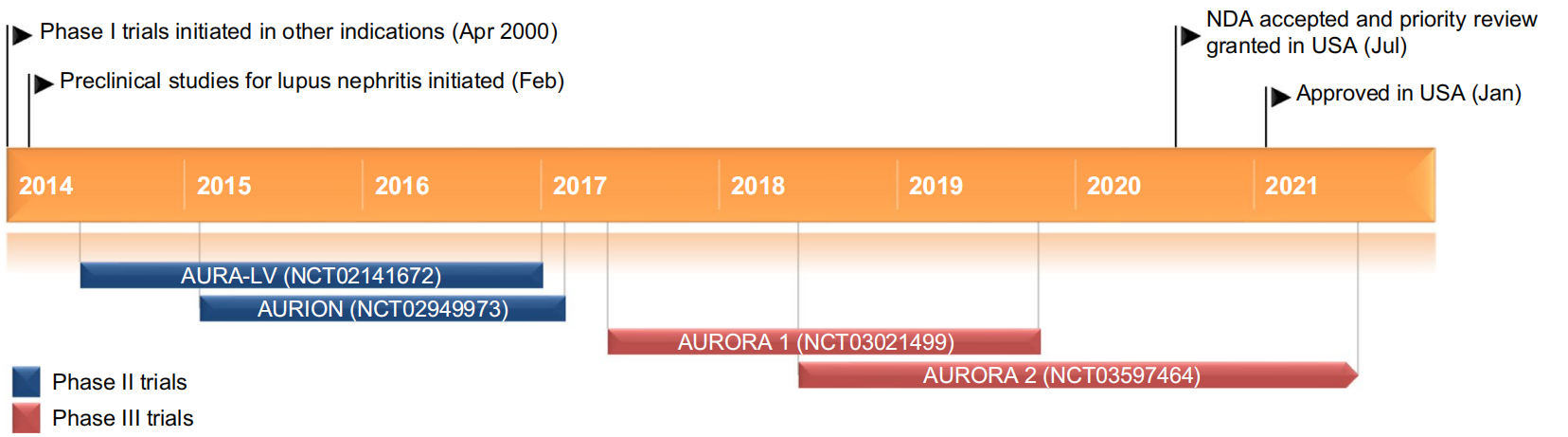
The clinical efficacy of voclosporin in lupus nephritis (LN) has been thoroughly validated through a series of key trials. These studies primarily assessed the drug’s ability to enhance renal outcomes when used in conjunction with standard therapies like mycophenolate mofetil (MMF) and corticosteroids. The most notable data emerged from the Phase II AURA-LV and Phase III AURORA 1 trials, both of which underscored voclosporin’s capacity to achieve Complete Renal Response (CRR) and significantly reduce proteinuria in patients with active LN.
In the AURA-LV trial, voclosporin-treated patients exhibited a markedly higher CRR rate at 24 weeks compared to the placebo group. The AURORA 1 trial confirmed these results, showing sustained CRR improvement at 52 weeks, with 40.8% of voclosporin patients achieving CRR versus 22.5% of those on placebo. Furthermore, voclosporin reduced proteinuria, a critical marker of kidney function, more rapidly than placebo.
Voclosporin’s efficacy was consistent across diverse patient demographics, including age, race, and prior immunosuppressant use. The drug maintained a favorable safety profile throughout the trials, with manageable side effects. These outcomes firmly position voclosporin as a critical therapeutic advancement in lupus nephritis, offering enhanced renal protection and improved long-term prognosis.
Safety Profile and Future Applications of Voclosporin
Voclosporin has demonstrated a generally favorable safety profile in clinical trials, especially compared to traditional calcineurin inhibitors such as cyclosporine and tacrolimus. The most common adverse effects associated with voclosporin include decreases in estimated glomerular filtration rate (eGFR), hypertension, diarrhea, and headaches. These side effects are expected with calcineurin inhibitors but were generally manageable with dose adjustments. One of the major safety concerns—nephrotoxicity—was observed in some patients, but it was mitigated through monitoring and dosage modifications.
Notably, voclosporin does not require therapeutic drug monitoring, which is a significant advantage over older CNIs. This simplifies its clinical use, reducing the burden on both patients and healthcare providers. Furthermore, voclosporin’s metabolic profile is more favorable, exhibiting fewer side effects related to lipid and glucose metabolism compared to cyclosporine and tacrolimus.
Beyond lupus nephritis, voclosporin is being explored for potential applications in other conditions requiring immunosuppressive therapy. Research is ongoing to evaluate its use in kidney transplantation and COVID-19-related kidney complications. These investigations suggest voclosporin could become a broader tool in managing immune-mediated diseases, given its effective immune suppression and renal protection mechanisms.
As clinical experience with voclosporin grows, it is likely that its role in managing various autoimmune and renal conditions will expand, providing new avenues for therapy with a safer profile compared to older treatments.
 LinkPeptide
LinkPeptide