Angiotensinogen Unbound: Rethinking Its Role in Obesity-Related Hypertension
Abstract
Recent evidence challenges the conventional renin-angiotensin system (RAS) paradigm by highlighting angiotensinogen as more than a passive precursor to angiotensin II. A 2025 study in Peptides investigated 103 obese, treatment-naïve men and revealed that plasma angiotensinogen levels were significantly associated with 24-hour ambulatory systolic and diastolic blood pressure—independently of angiotensin II. These associations persisted across daytime and nighttime measurements, suggesting a stable, pressure-modulating role for angiotensinogen. Contrary to prior assumptions, angiotensinogen was not correlated with adiposity measures, reinforcing the liver, rather than adipose tissue, as the primary source. These findings support a paradigm shift in understanding RAS physiology and open the door to novel therapeutic strategies, particularly RNA interference therapies targeting hepatic angiotensinogen synthesis. This evolving perspective has important implications for the management of obesity-related hypertension and underscores the need for further longitudinal and mechanistic studies.
From Substrate to Signal: Revisiting Angiotensinogen in Blood Pressure Regulation
Hypertension remains a global health priority, with obesity as a major driver of its increasing prevalence. Central to blood pressure regulation is the renin-angiotensin system (RAS), where angiotensin II—a potent vasoconstrictor—has been the primary therapeutic focus. Pharmacological agents like angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs) have long targeted this peptide with proven clinical efficacy.
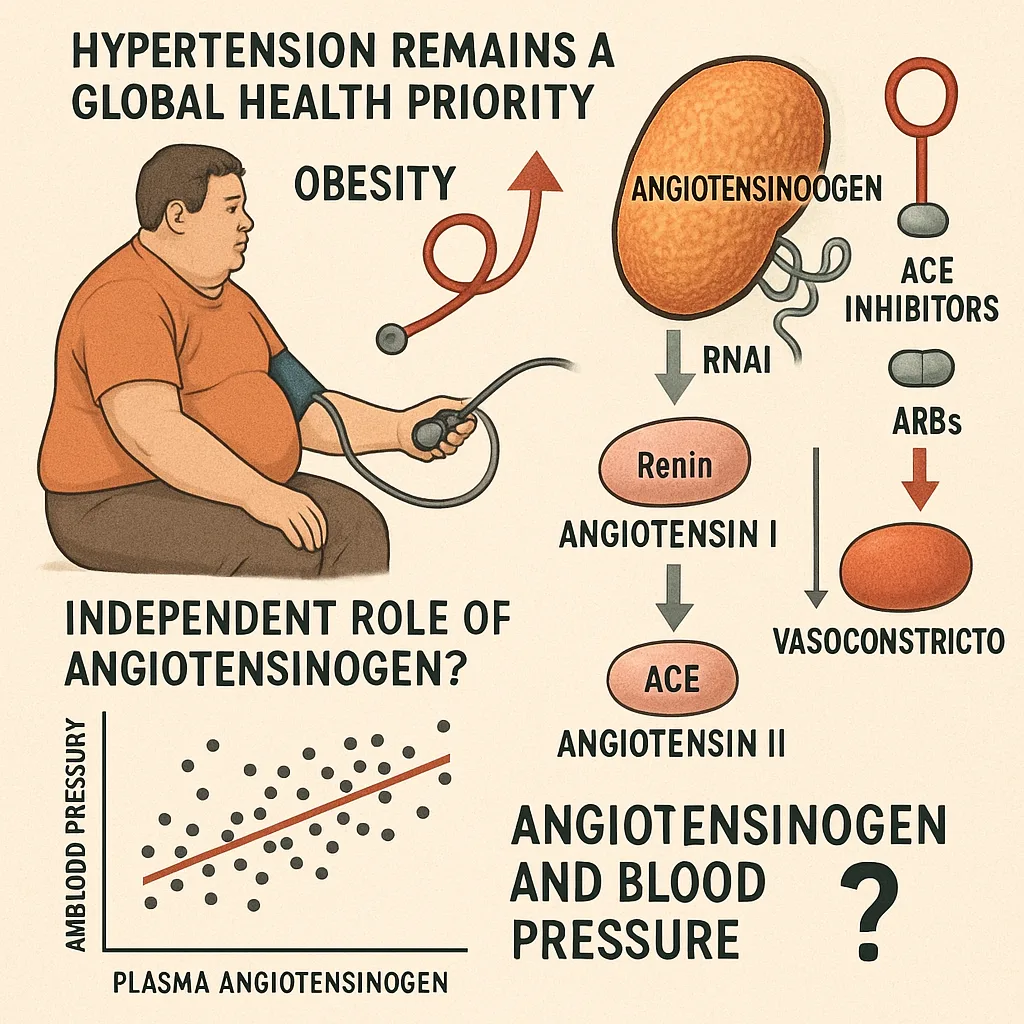
However, a growing body of evidence suggests that our angiotensin II–centered understanding of the RAS may be overly narrow.
At the top of the cascade sits angiotensinogen, a liver-derived glycoprotein historically considered a passive substrate for renin. Until recently, its role was largely dismissed as mechanistically inert beyond its enzymatic cleavage. But with the emergence of RNA interference (RNAi) therapies—notably those designed to inhibit hepatic angiotensinogen synthesis—scientific interest has shifted. This renewed focus raises a critical question: Can angiotensinogen itself modulate blood pressure independently of angiotensin II?
A recent study published in Peptides examines this question in depth. By analyzing 24-hour ambulatory blood pressure (ABP) in obese, treatment-naïve men, the researchers reveal that plasma angiotensinogen levels correlate strongly with ABP—even after adjusting for angiotensin II. These findings challenge conventional assumptions and suggest that angiotensinogen may play an active, previously underappreciated role in the pathophysiology of obesity-related hypertension.
Angiotensinogen and Ambulatory Blood Pressure: Evidence from Obese, Treatment-Naïve Men
To probe the potential role of angiotensinogen in blood pressure regulation beyond angiotensin II, the study recruited a cohort of 103 obese men (BMI ≥ 30 kg/m²), subdivided into hypertensive (n = 63) and normotensive (n = 40) groups. For comparison, a control group of 27 normal-weight, normotensive men (BMI 20.0–24.9 kg/m²) was also included. Importantly, all participants were treatment-naïve, free from diabetes, cardiovascular disease, or chronic illness, ensuring a metabolically clean sample.
Blood pressure was assessed using 24-hour ambulatory monitoring (ABP)—a method that offers superior prognostic value compared to office measurements. Biochemical profiling included fasting plasma concentrations of angiotensinogen, renin, angiotensin I, angiotensin II, and aldosterone, drawn after 60 minutes of supine rest to minimize acute RAS fluctuations.
The primary finding was both unexpected and compelling: Plasma angiotensinogen levels were significantly higher in hypertensive obese men compared to both normotensive obese and normal-weight individuals. Moreover, angiotensinogen levels showed a robust, independent association with 24-hour systolic and diastolic blood pressure, even after adjusting for age and plasma angiotensin II. Specifically, each 100 nmol/L increase in angiotensinogen was linked to a 1.8 mmHg rise in systolic BP and a 0.8 mmHg rise in diastolic BP.
These associations held true not only for overall 24-hour readings but also for daytime and nighttime measurements—underscoring the consistency and strength of angiotensinogen’s relationship with blood pressure across circadian variations.
Independent and Influential: Angiotensinogen Beyond the Angiotensin II Pathway
The renin-angiotensin system has long been conceptualized as a cascade in which angiotensinogen serves merely as raw material—converted to angiotensin I by renin, and then to angiotensin II, the active effector. Yet the current study disrupts this view, providing compelling evidence that angiotensinogen may influence blood pressure independently of angiotensin II.
What makes this finding so striking is the biological logic behind it. Unlike angiotensin II, which circulates in picomolar concentrations and fluctuates with acute stimuli, angiotensinogen exists in nanomolar quantities and remains relatively stable throughout the day. This stability suggests that it may serve as a chronic marker of RAS activity, analogous to how HbA1c reflects long-term glycemic control more reliably than a single glucose measurement.
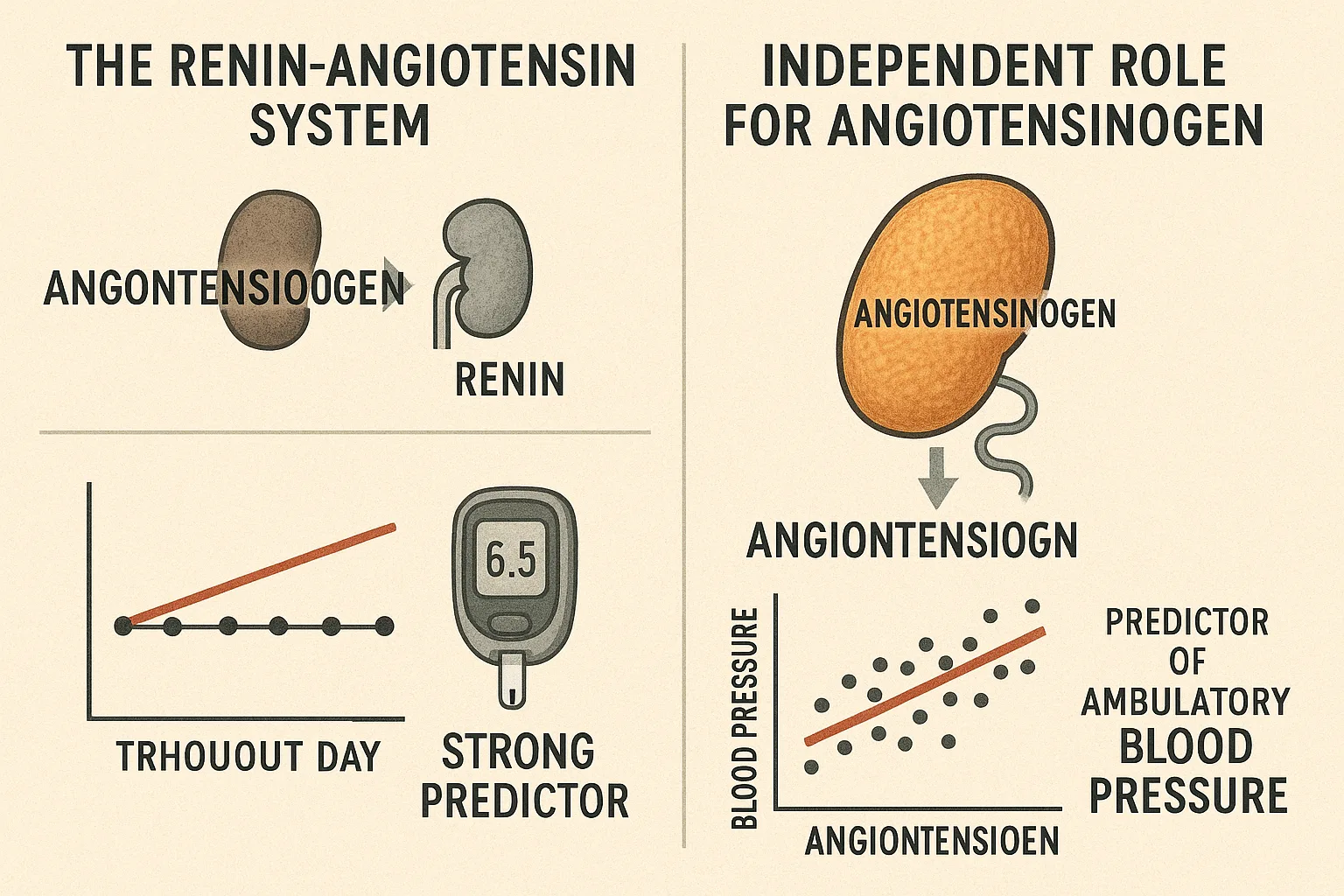
In this study, even after adjusting for plasma angiotensin II, angiotensinogen remained a strong predictor of 24-hour systolic and diastolic ABP. By contrast, angiotensin II showed inconsistent associations—linked only to diastolic BP and pulse, and not to systolic pressure or pulse pressure. These divergent patterns suggest that angiotensinogen may exert physiological effects via mechanisms that are not entirely dependent on angiotensin II generation.
One intriguing hypothesis is that angiotensinogen—or its metabolites—might affect vascular stiffness, thus influencing pulse pressure and systolic load. While this mechanism remains speculative, it introduces a novel dimension to the understanding of RAS physiology, particularly in the context of obesity, where vascular compliance is often compromised.
These findings justify deeper investigation into angiotensinogen as more than a passive precursor—and potentially as an active contributor to hypertensive pathology.
Liver Over Fat: Disentangling Adiposity from Circulating Angiotensinogen
Given the established links between obesity and elevated RAS activity, a reasonable assumption would be that adipose tissue contributes meaningfully to circulating angiotensinogen. Indeed, prior studies have reported local angiotensinogen expression in adipocytes, raising the possibility that increasing fat mass may drive systemic levels.
However, the findings from this study challenge that assumption.
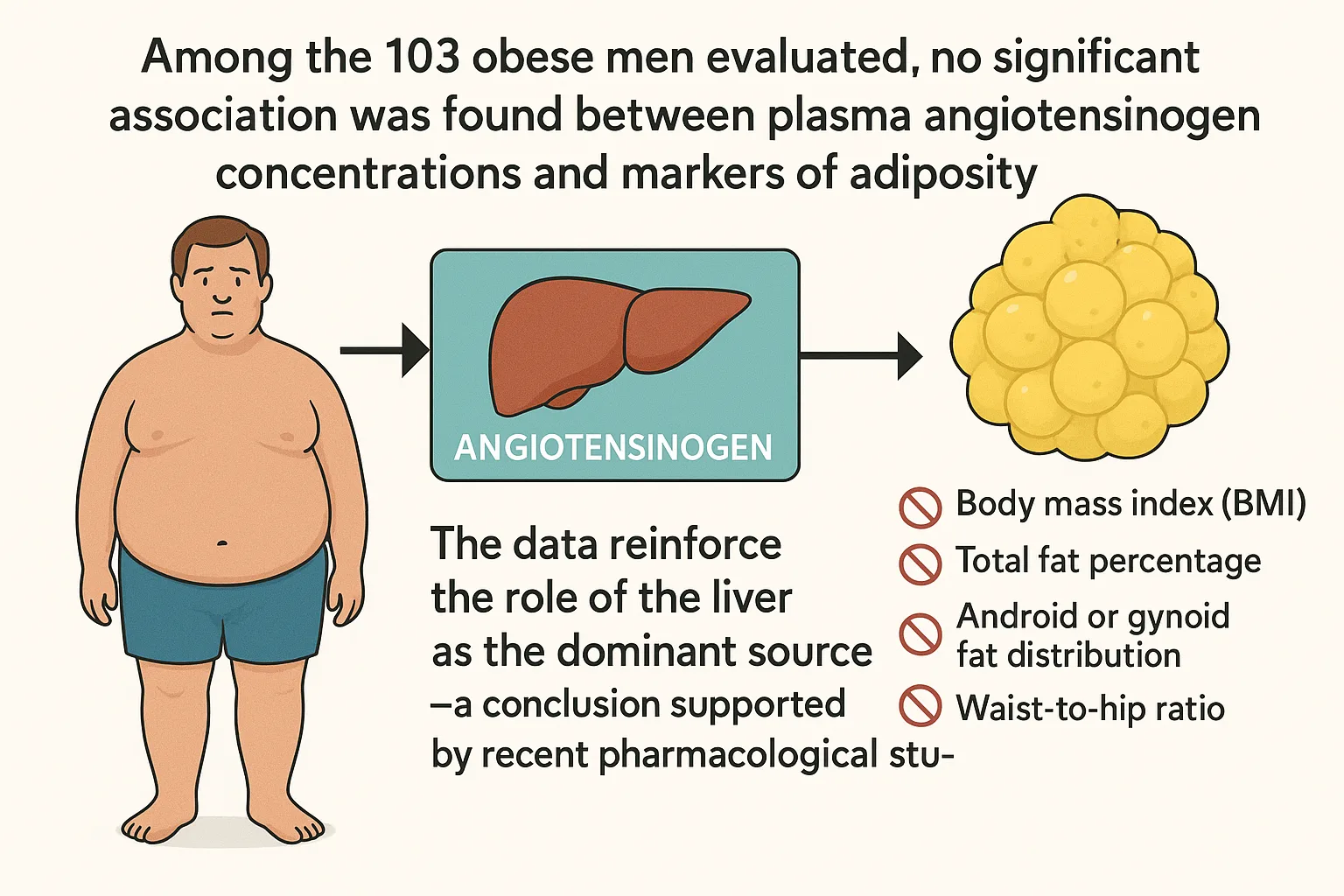
Among the 103 obese men evaluated, the researchers observed no significant association between plasma angiotensinogen concentrations and markers of adiposity—including body mass index (BMI), total fat percentage, android or gynoid fat distribution, or waist-to-hip ratio. This remained true even after adjusting for age and evaluating a wide spectrum of body composition values derived from dual-energy X-ray absorptiometry (DEXA) scans.
These null associations suggest that, despite the presence of angiotensinogen mRNA in adipose depots, adipose tissue is unlikely to be a major contributor to the circulating angiotensinogen pool. Instead, the data reinforce the role of the liver as the dominant source—a conclusion supported by recent pharmacological studies. For example, RNAi-based therapeutics that selectively silence hepatic angiotensinogen synthesis have been shown to reduce plasma concentrations by nearly 100%, without targeting adipose expression.
This clarification is more than academic. If angiotensinogen is indeed primarily liver-derived, then therapeutic strategies targeting hepatic synthesis—such as Zilebesiran and related agents—may be especially well-suited to mitigating systemic RAS overactivity in obesity-driven hypertension.
Targeting the Source: Therapeutic Implications for Hypertension in Obesity
The recognition that angiotensinogen may influence blood pressure independently of angiotensin II carries significant therapeutic implications—particularly in the management of obesity-related hypertension, where conventional strategies often face limitations.
The study’s findings align with a broader movement in cardiovascular pharmacology toward targeting upstream components of the renin-angiotensin system. Notably, the development of RNA interference (RNAi) therapeutics—such as Zilebesiran, which selectively inhibits hepatic angiotensinogen synthesis—reflects this shift. Clinical trials have already demonstrated that such agents can sustainably suppress plasma angiotensinogen levels, offering the promise of longer-lasting blood pressure control with potentially fewer dosing requirements.
Importantly, if angiotensinogen proves to be more than a passive precursor—functioning instead as an active contributor to vascular tone or arterial stiffness—then it represents a viable and perhaps underutilized therapeutic target. This could be especially relevant in populations with metabolic dysregulation, where traditional RAS blockade may not fully address the pathophysiological complexity.
However, caution is warranted. The present study is cross-sectional, includes only male participants, and assesses RAS activity at a single time point. Longitudinal studies with more diverse cohorts are needed to clarify causality and to understand whether angiotensinogen is simply a biomarker of RAS activation or a mechanistic driver of hypertension.
Nonetheless, this research marks a turning point. It challenges conventional hierarchies within the RAS and opens the door to novel therapeutic pathways that could improve outcomes in a growing population of patients for whom existing treatments remain insufficient.
 LinkPeptide
LinkPeptide