Beyond Single Signals: How Bioactive Peptides Shape Cellular Communication Networks
Introduction: Why Bioactive Peptides Matter More Than Ever
For decades, bioactive peptides have been studied as key signaling molecules in biology. From insulin regulation to neuropeptide signaling, they play essential roles in maintaining physiological balance. But modern research is revealing a deeper reality: cells don’t respond to just one peptide at a time—they respond to a complex network of peptide signals.
This shift in understanding is redefining how researchers approach peptide biology, drug discovery, and systems-level physiology. Instead of viewing peptides as isolated messengers, scientists now recognize them as part of an integrated signaling network that fine-tunes cellular responses.
What Are Bioactive Peptides?
Bioactive peptides are short chains of amino acids derived from larger precursor proteins. These precursors—called pre-propeptides—are processed through enzymatic cleavage and post-translational modifications to produce active signaling molecules.

Key characteristics:
- Generated through enzymatic cleavage (e.g., prohormone convertases)
- Often modified (amidation, acetylation, etc.) for activity
- Bind primarily to G protein-coupled receptors (GPCRs)
- Regulate processes such as metabolism, hormone release, and neural signaling
Unlike small signaling molecules like steroids or biogenic amines, peptides offer:
- Higher specificity
- Greater adaptability through evolution
- Ability to encode complex biological information
From “One Signal, One Effect” to Complex Networks
Traditional endocrinology followed a simple model:
One peptide → One receptor → One biological effect
However, this model no longer holds.
Modern research shows:
- Cells produce multiple peptides simultaneously
- Cells express many different peptide receptors
- One peptide can trigger multiple signaling pathways
For example, pancreatic islets contain not just insulin, glucagon, and somatostatin—but dozens of additional bioactive peptides, each influencing secretion and metabolism.
This means:
Cells are not responding to a single signal—but to an entire “peptidome”, the full set of peptides present at a given time.
The Peptidome: A New Layer of Biological Complexity
Advances in peptidomics (mass spectrometry-based peptide profiling) have revealed that biological systems contain far more peptides than previously expected .
These peptides:
- Exist in multiple processed forms
- Have varying receptor affinities
- Often act locally (paracrine/autocrine signaling)
In tissues like:
- Pancreas
- Hypothalamus–pituitary axis
- Brain
…the diversity of peptides creates a highly dynamic signaling environment.
Instead of asking:
“What does this peptide do?”
Researchers now ask:
“How does this peptide interact within a network of signals?”
Bioactive Peptides as Signaling Networks
The key insight from the paper is this:
Bioactive peptides function as part of a network-based signaling system, not isolated pathways.
This network includes:
- Peptides
- Receptors
- Enzymes (that activate or degrade peptides)
- Intracellular signaling cascades
How the Network Works
The signaling process can be simplified as:
Multiple peptide inputs → Integrated signaling network → Coordinated cellular response
Inside the cell:
- Pathways interact through shared signaling nodes
- Signals are processed using network motifs (basic regulatory patterns)
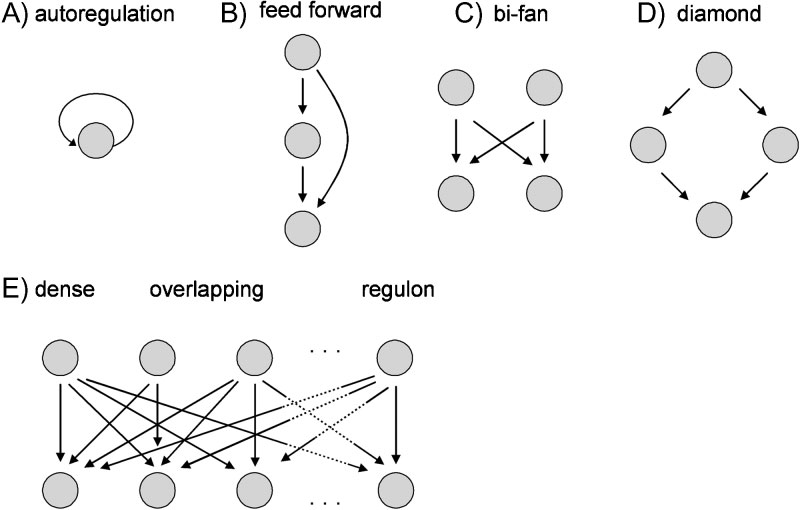
The picture illustrates these motifs—such as feed-forward loops and bi-fan structures—which are common in biological signaling systems and allow complex information processing.
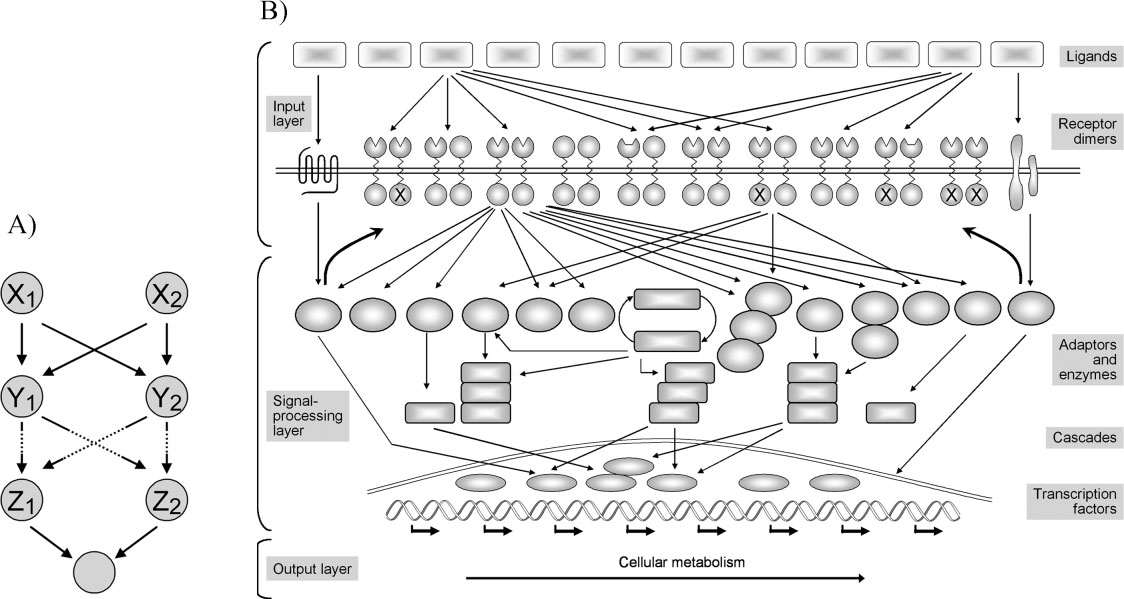
This architecture is similar to:
- Neural networks
- Artificial intelligence systems
Why Cells Need Network-Based Signaling
Biological systems must respond to:
- Changing environments
- Multiple simultaneous stimuli
- Complex physiological demands
A single-signal system cannot handle this complexity.
Instead, network-based signaling enables:
- Signal integration (combining multiple inputs)
- Specificity (different outputs from similar signals)
- Robustness (resistance to system failures)
- Adaptability (dynamic response to change)
For example:
A pancreatic beta cell integrates signals from glucose, hormones, and dozens of peptides before deciding how much insulin to release.
The Role of Peptide Processing in Signal Diversity
An often-overlooked factor is peptide processing.
After secretion, peptides can be:
- Cleaved into shorter fragments
- Modified by extracellular enzymes
- Converted into forms with different activities
These processes create:
- New signaling variants
- Different receptor interactions
- Fine-tuned biological responses
This means:
Even a single peptide precursor can generate multiple functional signals, greatly expanding signaling complexity.
Systems Biology: A New Framework for Peptide Research
To understand this complexity, researchers are turning to systems biology.
This approach:
- Models biological systems as networks
- Uses computational tools to predict behavior
- Integrates data from genomics, proteomics, and peptidomics
According to the paper, future progress depends on answering key questions:
- Which receptors does a cell express?
- Which pathways are activated?
- How do signals interact dynamically?
Implications for Drug Discovery and Research
This shift from linear to network thinking has major implications.
1. Drug Targeting Becomes More Complex
Targeting a single receptor may not be sufficient:
- Multiple pathways may compensate
- Off-target effects may arise from network interactions
2. Peptide Therapeutics Gain New Value
Peptides are uniquely suited for network modulation because:
- They are highly specific
- They can be engineered
- They mimic natural signaling systems
3. Precision Medicine Becomes More Achievable
Understanding peptide networks enables:
- Better biomarker discovery
- More accurate disease modeling
- Personalized therapeutic strategies
Evolution: Why Peptide Networks Exist
From an evolutionary perspective:
- Protein signaling came first
- Gene duplication increased signaling complexity
- Shorter peptides evolved as efficient signaling molecules
As networks expanded:
- More interactions emerged
- Signaling became more sophisticated
The diagram on page 10 shows how gene duplication increases network complexity, leading to more interconnected signaling systems.
Practical Takeaways for Researchers
If you work with peptides, here are key insights:
1. Don’t study peptides in isolation
Always consider:
- Co-expressed peptides
- Receptor diversity
- Environmental context
2. Use multiple analytical tools
Combine:
- HPLC
- LC-MS
- Peptidomics
3. Expect variability
Peptide behavior depends on:
- Processing
- Concentration
- Cellular context
4. Think in systems, not pathways
Shift from:
“What does this peptide do?”
To:
“How does this peptide interact within a network?”
How LinkPeptide Supports Advanced Peptide Research
At LinkPeptide, we recognize that peptide quality and characterization are critical for studying complex biological systems.
We support researchers with:
- High-quality custom peptide synthesis
- Analytical validation (HPLC, MS)
- Peptide design for functional studies
Because in modern research, peptide purity is only the starting point—understanding peptide interactions is the real challenge.
Conclusion: A New Era of Peptide Biology
Bioactive peptides are no longer just individual messengers—they are part of a highly interconnected signaling network that governs cellular behavior.
By integrating:
- Peptidomics
- Systems biology
- Advanced analytical tools
Researchers can move beyond simplified models and uncover the true complexity of biological regulation.
This network perspective is not just a theoretical shift—it’s a practical framework that will drive the next generation of discoveries in:
- Drug development
- Metabolic research
- Neuroscience
- Precision medicine
Reference
Boonen, K., Creemers, J. W., & Schoofs, L. (2009). Bioactive peptides, networks and systems biology. Bioessays, 31(3), 300-314. https://doi.org/10.1002/bies.200800055
Fuxe, K., Li, X. M., Tanganelli, S., Hedlund, P., O’Connor, W. T., Ferraro, L., … & Agnati, L. F. (1995). Receptor-receptor interactions and their relevance for receptor diversity. Focus on neuropeptide/dopamine interactions. Annals of the New York Academy of Sciences, 757, 365-376. https://doi.org/10.1111/j.1749-6632.1995.tb17495.x
B und Halbach, O. V. (2005). The renin-angiotensin system in the mammalian central nervous system. Current Protein and Peptide Science, 6(4), 355-371. https://doi.org/10.2174/1389203054546361
Barabasi, A. L., & Oltvai, Z. N. (2004). Network biology: understanding the cell’s functional organization. Nature reviews genetics, 5(2), 101-113. https://doi.org/10.1038/nrg1272
 LinkPeptide
LinkPeptide