Designing Peptide Receptor Agonists and Antagonists: Strategies Driving Modern Drug Discovery
Introduction: Why Peptide–Receptor Interactions Matter
Peptide–protein interactions are among the most fundamental mechanisms controlling biological systems. From hormone signaling to immune responses, these interactions regulate essential physiological processes such as metabolism, pain perception, cardiovascular function, and neural activity.
In fact, peptide ligands—including hormones, neurotransmitters, and cytokines—represent a significant portion of current therapeutic targets and drug candidates. As a result, designing peptide receptor agonists and antagonists has become a central focus in modern drug discovery.
This article explores the core design strategies, structure–activity relationships (SAR), and emerging approaches that enable the development of highly selective and biologically active peptide ligands.
What Are Peptide Receptor Agonists and Antagonists?
Peptide ligands interact with receptors—most commonly G protein-coupled receptors (GPCRs)—to regulate cellular responses.
- Agonists activate receptors, triggering a biological response
- Antagonists bind receptors but block activation, preventing signaling
- Inverse agonists reduce baseline receptor activity
These ligands are critical for treating diseases such as:
- Diabetes (e.g., insulin analogues)
- Pain (opioid peptides)
- Cardiovascular disorders
- Neurological and endocrine diseases
However, natural peptides are rarely ideal drugs due to:
- Rapid enzymatic degradation
- Short half-life
- Poor membrane permeability
This makes rational peptide design essential.
Core Strategy: From Natural Peptides to Optimized Ligands
The design of peptide therapeutics typically begins with a natural ligand and evolves through systematic modification.
The workflow diagram outlines a widely used strategy:
- Identify biologically active peptides
- Study structure–activity relationships (SAR)
- Define pharmacophore elements
- Introduce conformational constraints
- Develop optimized peptides or peptidomimetics
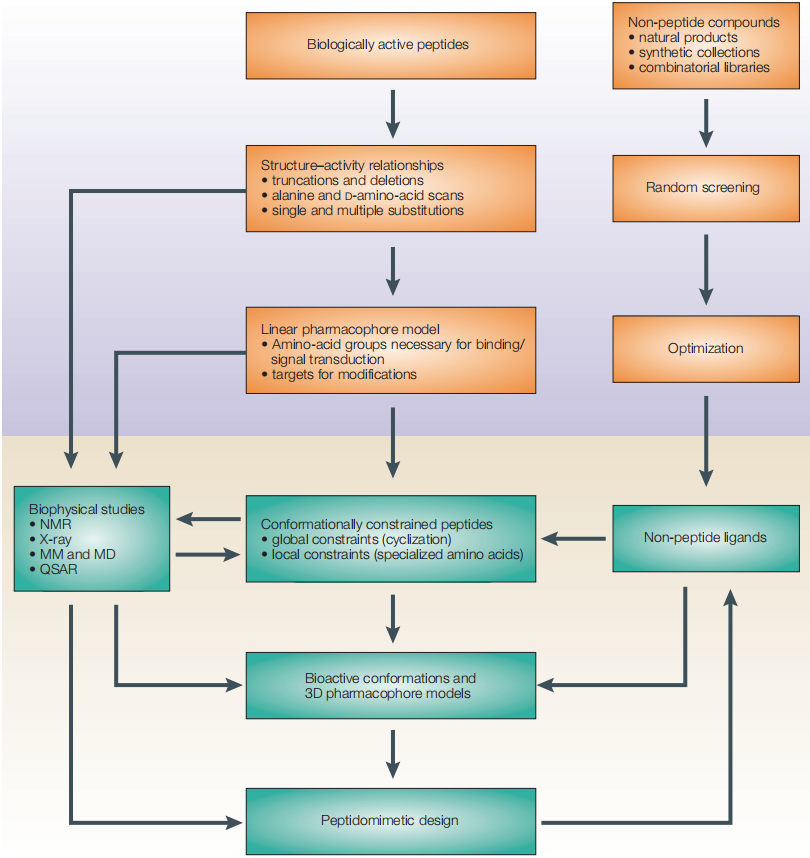
This stepwise approach allows researchers to transform unstable natural peptides into potent, selective, and drug-like molecules.
Step 1: Identifying the Pharmacophore
A pharmacophore represents the minimal structural features required for biological activity.
Key methods:
1. Truncation Studies
Peptides are systematically shortened from both ends to determine:
- Minimum active sequence
- Essential residues for activity
This reduces molecular complexity and guides further optimization.
2. Amino Acid Scanning
Techniques such as:
- Alanine scanning
- Glycine scanning
- D-amino acid substitution
help determine the role of individual residues in:
- Binding affinity
- Signal transduction
For example, replacing residues one-by-one reveals which side chains are critical for receptor interaction.
Step 2: Understanding Structure–Activity Relationships (SAR)
Once key residues are identified, researchers explore how structural changes affect function.
SAR analysis examines:
- Side-chain properties (hydrophobicity, charge, size)
- Chirality and stereochemistry
- Backbone flexibility
These insights guide the design of peptides with:
- Improved potency
- Enhanced selectivity
- Reduced off-target effects
Step 3: Conformational Control—A Critical Design Principle
Peptides are inherently flexible, but biological activity depends on specific 3D conformations.
Local Conformational Constraints
Modifications such as:
- D-amino acid substitution
- N-methylation
- α-substituted amino acids
can stabilize structures like:
- α-helices
- β-sheets
- β-turns
These constraints help identify the bioactive conformation required for receptor binding.
Global Conformational Constraints (Cyclization)
Cyclization is one of the most powerful strategies in peptide design.
The illustrations on page 5 show different cyclization approaches, including:
- Head-to-tail cyclization
- Disulfide bridges
- Side chain-to-side chain linkages
Benefits include:
- Increased structural stability
- Resistance to enzymatic degradation
- Enhanced receptor selectivity
- Improved bioavailability
Case Study: Enkephalin Optimization
A classic example is the modification of enkephalin, a natural opioid peptide.
By introducing:
- Disulfide bridges
- Conformational constraints
researchers developed DPDPE, a cyclic peptide with:
- High receptor selectivity
- Strong biological activity
- Improved stability and brain penetration
This demonstrates how structural engineering transforms natural peptides into viable drug candidates.
Step 4: Side-Chain Engineering and Topography
Beyond backbone structure, side-chain orientation plays a crucial role in receptor recognition.
Peptides contain flexible side chains defined by torsional angles (χ angles). These determine:
- Binding interactions
- Signal activation
By introducing conformationally constrained amino acids, researchers can:
- Lock side chains into specific orientations
- Enhance receptor selectivity
- Fine-tune biological activity
Designing Antagonists: A Different Challenge
Unlike agonists, antagonists require a different design strategy.
Key differences:
- Bind receptor without activation
- Often have distinct conformations
- Require different pharmacophore features
Research shows that:
- Agonists and antagonists may bind the same receptor
- But stabilize different receptor conformations
Strategies for Antagonist Design
- Sequence Truncation
- Removes activation domains
- Retains binding ability
- Peptide Fragmentation
- Separates “message” (activation) and “address” (binding) regions
- Combinatorial Libraries
- Screen large peptide sets for antagonist activity
- Conformational Restriction
- Locks peptide into inactive-binding conformations
Case Study: Glucagon Antagonists
Glucagon regulates glucose metabolism, making it a key diabetes target.
Studies showed:

- Removing specific residues converts agonists into antagonists
- Multiple modifications were required to eliminate residual activity
This highlights the complexity of antagonist design, especially in larger peptides.
Agonist vs Antagonist: Structural Differences
A critical insight from modern research:
Agonists and antagonists do not just differ in function—they differ in structure, conformation, and receptor interaction dynamics.
Even subtle changes in:
- Side-chain orientation
- Backbone conformation
- Peptide flexibility
can switch activity from agonist to antagonist.
Toward Peptidomimetics and Non-Peptide Drugs
Once the active peptide structure is understood, researchers can design:
- Peptidomimetics (peptide-like molecules)
- Non-peptide ligands
These offer advantages such as:
- Oral bioavailability
- Increased stability
- Lower production cost
This transition represents a key step in translating peptide biology into therapeutics.
Systems Biology: The Future of Peptide Drug Design
Traditional drug design focused on single targets, but this approach is increasingly insufficient.
Modern insights show:
- Diseases are multifactorial
- Multiple pathways interact
- Receptor systems adapt dynamically
Future strategies must consider:
- Network-level interactions
- Multi-target modulation
- System-wide effects
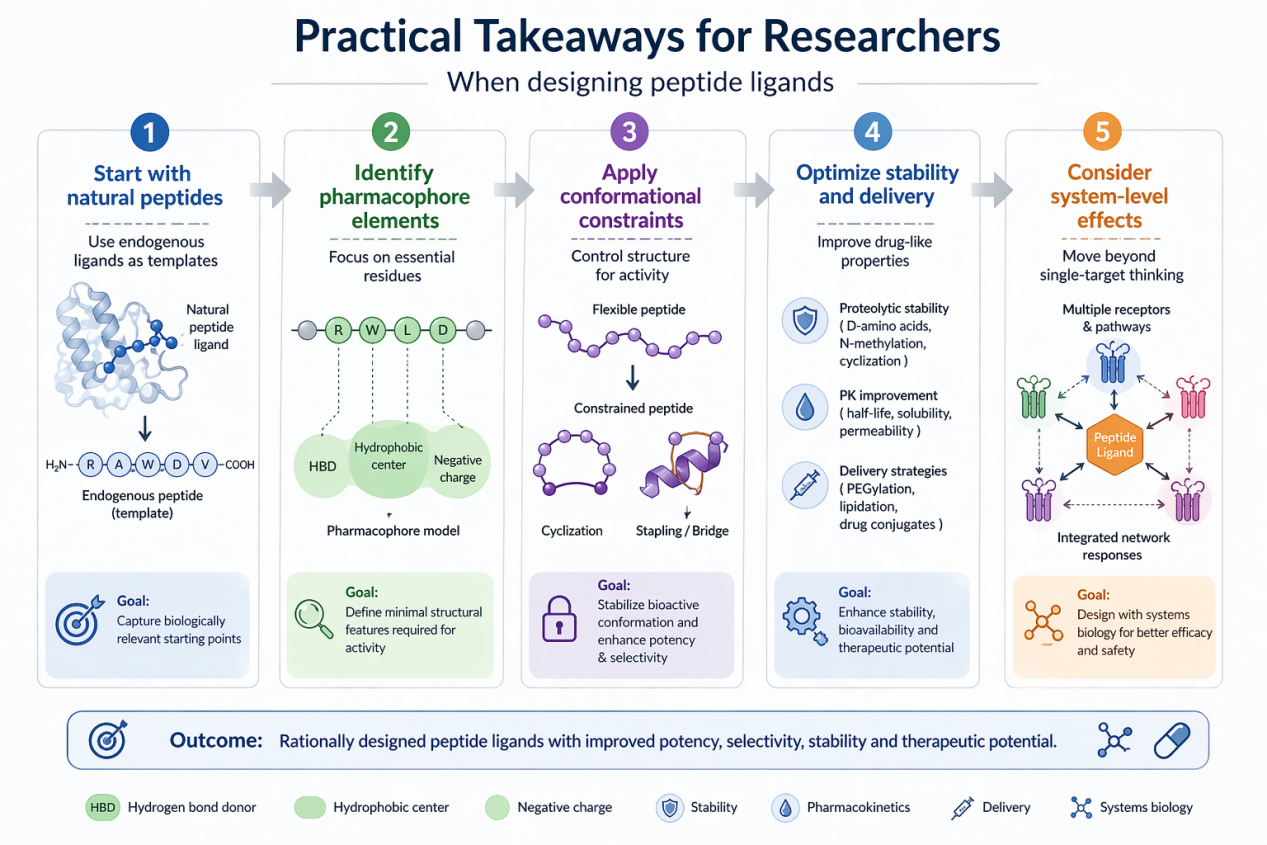
Practical Takeaways for Researchers
When designing peptide ligands:
1. Start with natural peptides
Use endogenous ligands as templates
2. Identify pharmacophore elements
Focus on essential residues
3. Apply conformational constraints
Control structure for activity
4. Optimize stability and delivery
Improve drug-like properties
5. Consider system-level effects
Move beyond single-target thinking
How LinkPeptide Supports Peptide Drug Design
At LinkPeptide, we support advanced peptide research and drug development with:
- Custom peptide synthesis
- Structure optimization support
- High-resolution analytical validation (HPLC, MS)
- Design consultation for functional peptides
From early-stage discovery to advanced optimization, we help researchers translate peptide concepts into reliable experimental tools and therapeutic candidates.
Conclusion
Designing peptide receptor agonists and antagonists is a sophisticated process that integrates:
- Structural biology
- Chemistry
- Pharmacology
- Systems biology
By combining structure–activity insights, conformational control, and modern analytical techniques, researchers can develop highly selective and effective peptide-based therapeutics.
As drug discovery evolves, peptides and their mimetics will continue to play a central role in targeting complex biological systems.
Reference
Hruby, V. J. (2002). Designing peptide receptor agonists and antagonists. Nature reviews Drug discovery, 1(11), 847-858. https://doi.org/10.1038/nrd939
Kessler, H. (1982). Conformation and biological activity of cyclic peptides. Angewandte Chemie International Edition in English, 21(7), 512-523. https://doi.org/10.1002/anie.198205121
Shenderovich, M. D., Liao, S., Qian, X., & Hruby, V. J. (2000). A three‐dimensional model of the δ‐opioid pharmacophore: Comparative molecular modeling of peptide and nonpeptide ligands. Biopolymers: Original Research on Biomolecules, 53(7), 565-580. https://doi.org/10.1002/(SICI)1097-0282(200006)53:7%3C565::AID-BIP4%3E3.0.CO;2-5
Hruby, V. J. (2001). Design in Topographical Space of Peptide and Peptidomimetic Ligands That Affect Behavior. A Chemist’s Glimpse at the Mind− Body Problem. Accounts of Chemical Research, 34(5), 389-397. https://doi.org/10.1021/ar990063q
 LinkPeptide
LinkPeptide