Cleaner Chemistry, Better Peptides: The Rise of N-to-C Synthesis
Abstract
As the demand for peptide-based therapeutics grows, so too does the need for sustainable manufacturing methods. Traditional solid-phase peptide synthesis (SPPS), typically performed in the C-to-N direction, has long dominated the field—but it comes at a high environmental cost, consuming large amounts of reagents, solvents, and energy. In contrast, N-to-C peptide synthesis, a method that mimics the natural direction of peptide biosynthesis, is gaining renewed attention for its green chemistry potential. Once sidelined due to concerns about epimerization and scalability, recent advances in coupling strategies—such as thioacids, vinyl esters, and transamidation—have made N-to-C synthesis both feasible and efficient. This blog explores the scientific breakthroughs enabling this shift, compares the environmental benefits of the two approaches, and discusses the challenges that remain. As regulatory and sustainability pressures intensify, N-to-C synthesis may represent the future of cleaner, smarter peptide production.
Introduction: The Peptide Revolution Needs a Sustainability Overhaul
Peptides are revolutionizing modern medicine. As therapeutics, they offer precision, potency, and low toxicity—making them ideal candidates for treating cancer, metabolic disorders, infections, and more. But while demand for peptides continues to surge, the methods used to synthesize them are under increasing environmental scrutiny.
The industry standard, solid-phase peptide synthesis (SPPS), typically assembles peptides from the C-terminal to the N-terminal. Since its debut in the 1960s, C-to-N SPPS has enabled automation and high-throughput production. However, it comes at a steep environmental cost. The process relies on atom-inefficient protecting groups like Fmoc, excess reagents, and large volumes of toxic solvents.
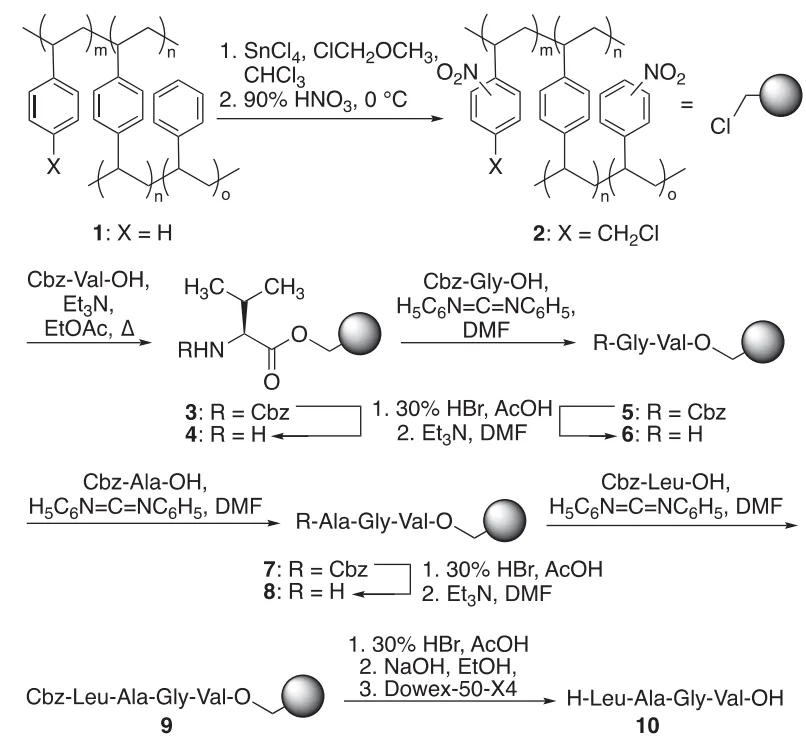
Merrifield C- to N-direction solid-phase peptide synthesis
As a result, peptide production has an exceptionally high process mass intensity (PMI)—up to 80 times greater than that of small-molecule drugs and nearly double that of biologics. This inefficiency raises serious concerns as pharmaceutical manufacturers move toward greener, more sustainable practices.
Now, scientists are revisiting an alternative: N-to-C peptide synthesis. This approach, which mimics nature’s ribosomal peptide assembly, was historically sidelined due to fears of epimerization. But with recent advances in coupling chemistry and process design, those challenges are being overcome.
This blog series explores how N-to-C synthesis—once seen as impractical—may offer a cleaner, more sustainable path forward in peptide manufacturing. From improved atom economy to reduced waste, the case for reversing direction has never been stronger.
Understanding the Chemistry: What Is N-to-C Peptide Synthesis?
To understand why N-to-C peptide synthesis is gaining traction, it helps to first look at how peptides are conventionally built. In standard C-to-N solid-phase peptide synthesis (C-N SPPS), peptide chains are assembled starting from the carboxyl (C-) terminal, attaching each new amino acid via its free amine group. This directionality has been favored for decades due to its compatibility with protecting group strategies that limit racemization and simplify purification.
However, nature takes a different approach.
Inside cells, peptides and proteins are synthesized in the N-to-C direction, where amino acids are joined starting from the amine (N-) terminal. The ribosome, nature’s protein factory, orchestrates this process with remarkable efficiency and fidelity. Mimicking this natural assembly has long been a goal in synthetic chemistry—but early attempts at N-to-C synthesis faced technical challenges. Chief among them was epimerization, a loss of stereochemistry that can compromise peptide activity and purity.
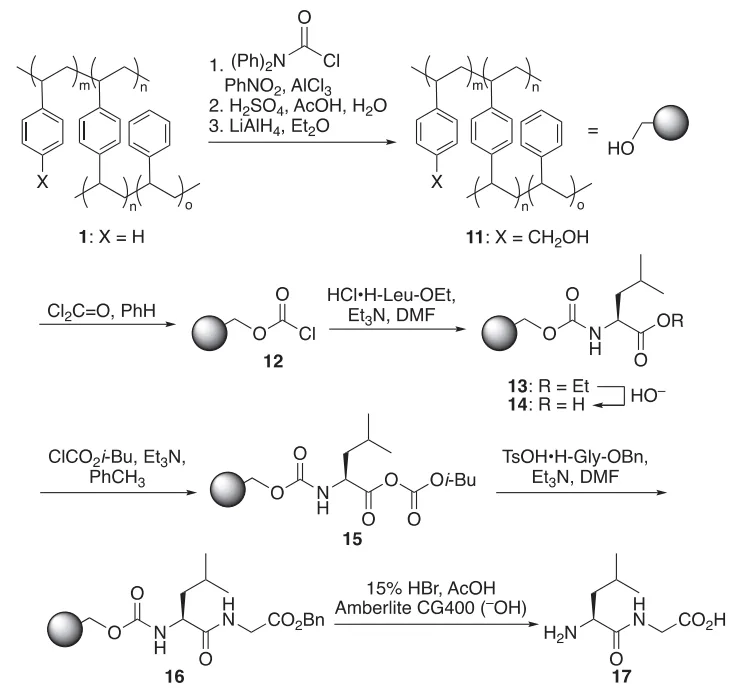
Letsinger N- to C-direction solid-phase peptide synthesis
Despite these concerns, the concept of N-to-C synthesis never fully disappeared. Researchers continued to explore new methods, from acyl azide chemistry to thiocarboxylic acid couplings, in search of ways to make the approach viable. Over the past two decades, these efforts have yielded promising results—strategies that minimize side reactions while improving atom economy and sustainability.
Today, N-to-C synthesis isn’t just feasible—it’s increasingly practical, especially for producing peptides with modified C-termini or in greener, less wasteful ways.
Breaking the Racemization Barrier: 21st Century Innovations
For decades, the primary barrier to adopting N-to-C peptide synthesis was the fear of epimerization—the unintended inversion of stereochemistry during coupling steps. Unlike traditional methods that use protected amino acids to guard against this risk, early N-to-C approaches lacked the same level of control. As a result, they were largely dismissed as impractical for high-fidelity synthesis.
But that’s changing.
Thanks to innovative chemistry, modern N-to-C synthesis can now proceed with minimal or no epimerization—even without the heavy reliance on protective groups. Several techniques have been instrumental in this breakthrough:
- Acyl azide chemistry, first explored in the 1970s and now refined, allows for mild and clean activation of amino acids from the carboxyl end.
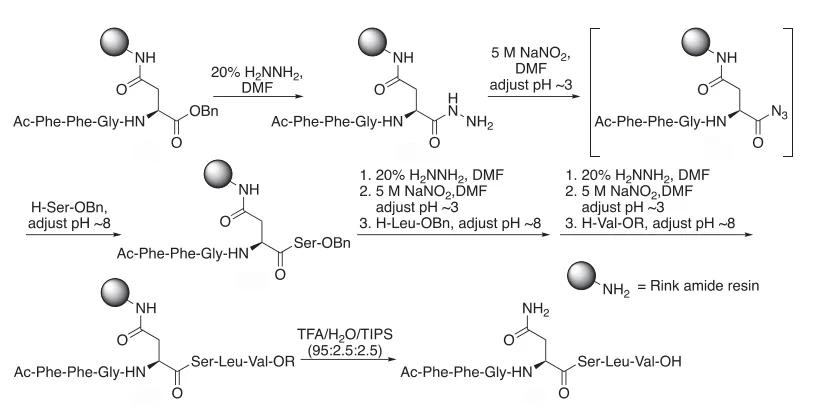
- Thioacids, inspired by natural biosynthesis, enable peptide bond formation under gentle conditions, reducing the risk of side reactions.
- Vinyl esters, activated via allenones or ynamides, have proven effective in building complex peptides like carfilzomib—a cancer drug—without racemization and with 68% overall yield.
- Transamidation, using twisted amide or metal-catalyzed systems, enables precise amide bond formation in the N-to-C direction with strong stereochemical control.
These strategies have expanded the toolkit for peptide chemists, making N-to-C synthesis not only viable but in some cases superior—particularly when cleaner reactions and greener solvents are a priority.
What was once a theoretical curiosity is now a proven, scalable approach. As these innovations mature, they’re setting the stage for a more sustainable era in peptide manufacturing.
Why N-to-C Matters for Green Chemistry
Beyond technical feasibility, what truly sets N-to-C peptide synthesis apart is its potential to support green chemistry principles—a growing priority for pharmaceutical and biotech industries striving to reduce their environmental footprint.
One of the core advantages of N-to-C synthesis lies in its superior atom economy. Unlike conventional C-to-N methods that depend heavily on bulky protecting groups like Fmoc and excess coupling agents, N-to-C strategies can often proceed with minimal protection, especially on the carboxyl side. Fewer protecting groups mean fewer reagents, less waste, and simplified purification workflows.
Additionally, many modern N-to-C methods use milder, more environmentally friendly solvents. For example, newer coupling strategies avoid problematic solvents like DMF, which is now subject to regulatory restrictions in parts of Europe. Methods based on thioacids, vinyl esters, or transamidation often function well in solvents like DMSO, THF, or even water-rich systems, aligning better with sustainability goals.
There’s also a practical edge: N-to-C synthesis can streamline the production of C-terminally modified peptides, a growing class of bioactive compounds used in drug discovery. These modifications are more easily introduced when the C-terminus is built last, rather than being anchored to a resin.
In short, N-to-C synthesis opens a path toward leaner, cleaner peptide production—reducing chemical input, energy consumption, and hazardous waste. As industries face increasing pressure to quantify and cut their PMI (process mass intensity), this approach offers a compelling route to greener, more responsible manufacturing.
Challenges and the Road Ahead
While N-to-C peptide synthesis offers clear sustainability advantages, several challenges still limit its widespread adoption—especially in large-scale or automated production.
One key limitation is the lack of robust anchoring strategies for the N-terminal end. Unlike C-to-N SPPS, which benefits from decades of resin development and optimized workflows, N-to-C methods often require custom linkers or resins that aren’t yet standardized. This makes automation and high-throughput synthesis—especially for combinatorial libraries—more difficult to implement at scale.
There’s also a learning curve. Many peptide chemists are deeply familiar with traditional SPPS protocols and may hesitate to shift to a less established, albeit promising, workflow. Further investment in training, process validation, and equipment compatibility will be needed to fully integrate N-to-C synthesis into industrial pipelines.
That said, momentum is building. The successful N-to-C synthesis of complex peptides like carfilzomib, with superior yields and cleaner profiles, signals that this approach is not only viable but in some cases preferable. Combined with advances in green reagents, solvent-free technologies like mechanochemistry, and novel purification supports, the foundation is being laid for a new generation of peptide manufacturing.
As the pressure to reduce chemical waste and meet environmental standards intensifies, N-to-C peptide synthesis is poised to shift from niche curiosity to mainstream solution. With continued innovation and collaborative investment, reversing the direction of peptide assembly might just move the entire field forward.
 LinkPeptide
LinkPeptide