Daridorexant: A Breakthrough Dual Orexin Receptor Antagonist for Insomnia Treatment and Beyond
Abstract
Daridorexant, a novel dual orexin receptor antagonist (DORA), has emerged as a promising treatment for insomnia, offering improvements in both sleep onset and maintenance without the residual daytime effects commonly associated with traditional sedative-hypnotic agents. By selectively targeting orexin receptors OX1 and OX2, Daridorexant promotes sleep by inhibiting the brain’s wakefulness-promoting pathways. Clinical trials have demonstrated its efficacy in reducing wake time after sleep onset (WASO) and improving total sleep time (sTST), with a favorable safety profile, especially in elderly patients. The drug’s pharmacokinetics, characterized by a half-life of approximately 8 hours, support its once-daily dosing regimen, ensuring minimal impact on next-day functioning. Ongoing studies aim to explore its potential in additional therapeutic areas. This article delves into the pharmacodynamics, clinical trial outcomes, safety considerations, and future prospects of Daridorexant as a game-changing insomnia therapy.
Daridorexant: A New Approach to Insomnia Treatment
Insomnia disorder is a common sleep disorder associated with several comorbidities and major economic and societal impacts. Current guidelines recommend psychological and pharmacological therapies to reduce the time to fall asleep and the time spent awake after sleep onset, with subsequent improvements in daytime functioning. Among pharmacological approaches, benzodiazepines (BZD), ‘Z’-drugs, melatonin receptor agonists, and sedating antidepressants have been frequently used options for insomnia, but typically are not effective enough. Furthermore, currently available sleep medications that adequately address problems with sleep maintenance may be associated with common side effects of somnolence and drowsiness. Daridorexant represents a new class of insomnia medications known as dual orexin receptor antagonists (DORA), which offer a more targeted approach to managing sleep disorders.

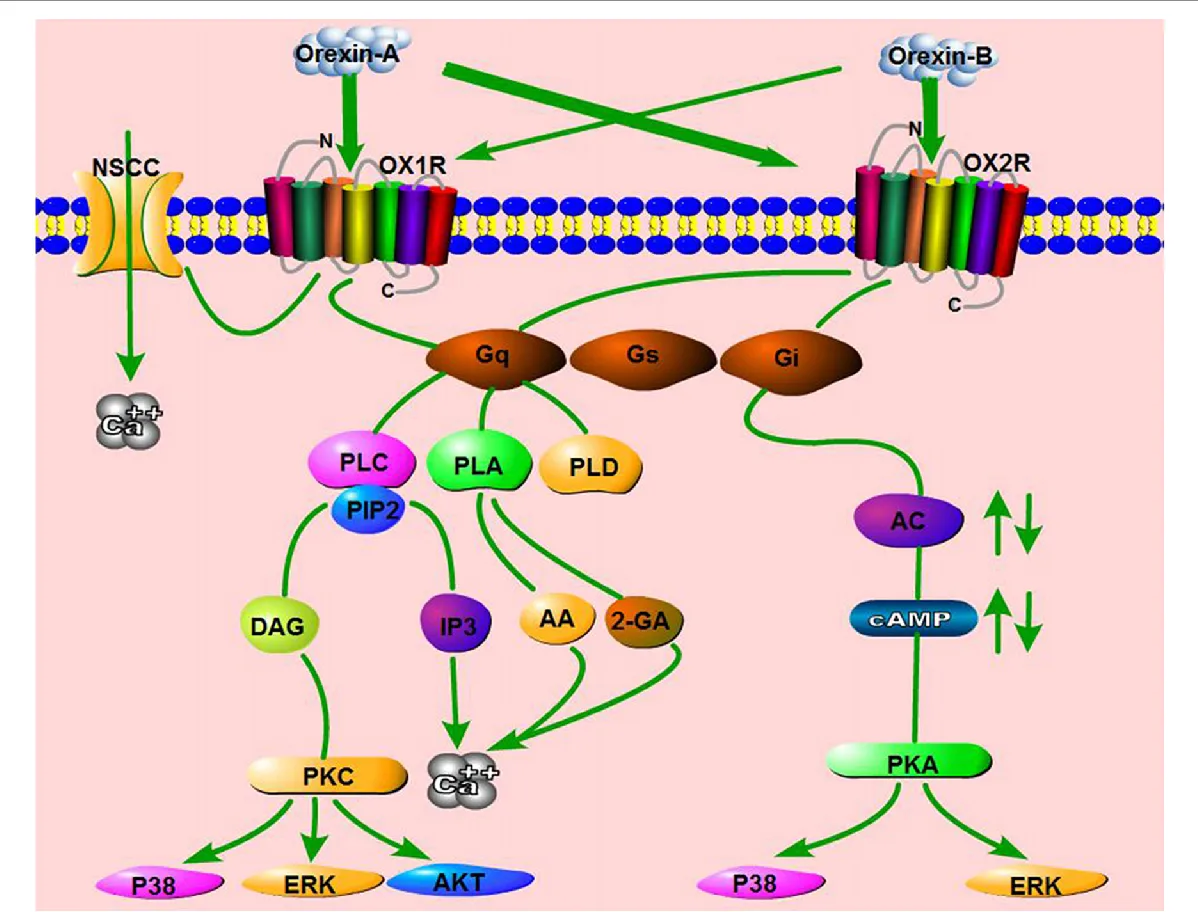
Daridorexant acts by inhibiting the activity of orexin A and orexin B, neuropeptides responsible for promoting wakefulness through their interaction with the orexin type 1 (OX1) and type 2 (OX2) receptors. By blocking these receptors, Daridorexant reduces the stimulation of wakefulness-promoting pathways, allowing for a more natural transition to sleep. This unique mechanism distinguishes Daridorexant from traditional sedative-hypnotic agents, as it specifically targets the brain’s wakefulness system without broadly depressing the central nervous system.
One of the key advantages of Daridorexant is its tailored pharmacological profile. It was selected for its optimal duration of action, with an expected effect of approximately 8 hours, ensuring that it helps with both sleep onset and maintenance. Furthermore, its pharmacokinetics were designed to minimize residual next-day effects, offering patients a solution that improves nighttime sleep without impairing daytime functioning. This makes Daridorexant a promising treatment option for patients struggling with insomnia.
Unveiling the Pharmacodynamics and Pharmacokinetics of Daridorexant
Daridorexant’s efficacy as an insomnia treatment is rooted in its precise pharmacodynamic and pharmacokinetic properties. As a dual orexin receptor antagonist (DORA), Daridorexant blocks the action of orexin A and orexin B on their respective receptors, OX1 and OX2, which are crucial in promoting wakefulness. The drug’s inhibition constants (Kb) for human OX1 and OX2 receptors are 0.52 nM and 0.78 nM, respectively, demonstrating its potent and selective action. Preclinical studies in animals showed that Daridorexant significantly increases time spent in both non-REM and REM sleep, while also reducing the latency to persistent sleep.
Daridorexant is designed to strike an ideal balance between effectiveness and safety. After taking it orally, the drug reaches peak levels in the bloodstream within 1 to 2 hours, making it an ideal choice for nightly insomnia treatment. With a half-life of around 8 hours, Daridorexant works throughout the night without causing excessive drowsiness the next day. It’s primarily broken down by the enzyme CYP3A4 and is mostly excreted as metabolites through feces and urine. Importantly, Daridorexant’s pharmacokinetics remain consistent across different patient groups, including those with mild-to-moderate kidney issues and elderly individuals, ensuring reliable performance across a range of users.
Additionally, Daridorexant’s pharmacokinetics are not significantly affected by food intake, although a high-fat meal may delay its absorption slightly. This makes it a convenient option for patients, who can take it regardless of meal timing.
Clinical Trials Highlight Daridorexant’s Efficacy in Sleep Improvement
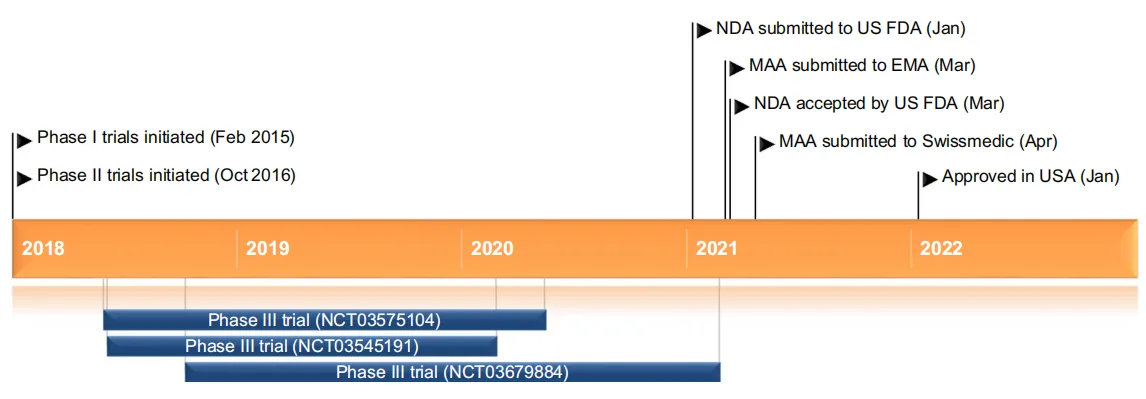
Daridorexant’s effectiveness has been thoroughly tested in several phase III trials, showing it can significantly improve sleep for people with insomnia. Two major studies (NCT03545191 and NCT03575104) focused on key sleep measures like wake time after sleep onset (WASO), the time it takes to fall into continuous sleep (LPS), and total sleep time (TST). In both studies, Daridorexant at 25 mg and 50 mg doses led to a notable reduction in WASO. These improvements were seen as early as one month into treatment and continued over the course of three months.
At the 50 mg dose, Daridorexant not only improved sleep but also boosted daytime functioning, as measured by the Insomnia Daytime Symptoms and Impacts Questionnaire (IDSIQ), which evaluates how insomnia affects next-day alertness and performance. This is a big advantage over many traditional sleep aids, which may help with sleep but often leave users feeling groggy and impaired the next day. By improving both sleep quality and daytime functioning, Daridorexant offers a more well-rounded solution for insomnia.
The trials also looked at a subgroup of elderly patients, aged 65 and older, who are often more susceptible to the side effects of traditional sedatives. The findings showed that Daridorexant was just as effective and safe in this group, making it a strong option for older adults, where insomnia is especially common. With consistent positive results across the board, Daridorexant shows great promise as a breakthrough treatment for insomnia, addressing both sleep issues and next-day alertness.
Evaluating the Safety and Side Effects of Daridorexant
Daridorexant has been shown to have a favorable safety profile, with most adverse effects being mild to moderate in severity. Across the phase III trials, the most commonly reported side effects were nasopharyngitis, headache, dizziness, and fatigue, with incidence rates similar to those observed in placebo groups. The drug’s overall tolerability is one of its key advantages, particularly compared to older insomnia treatments that are associated with a higher risk of dependency, next-day sedation, and cognitive impairment.
In rare cases, more serious side effects such as sleep paralysis, hypnagogic and hypnopompic hallucinations, and complex sleep behaviors (e.g., sleepwalking and sleep-driving) were reported. These events, while uncommon, are consistent with those observed in other medications targeting the central nervous system, and patients experiencing these should discontinue use immediately. Moreover, Daridorexant is not recommended for patients with severe respiratory conditions, such as obstructive sleep apnea (OSA) and chronic obstructive pulmonary disease (COPD), due to its potential to exacerbate respiratory depression.
Daridorexant’s risk of daytime impairment, a common concern with insomnia medications, is minimized by its short half-life of approximately 8 hours, which allows the drug to clear from the system in time for the next day’s activities. Nonetheless, caution is advised for patients when driving or operating machinery, particularly during the initial days of treatment. Importantly, Daridorexant does not show clinically relevant prolongation of the QT interval, making it safer from a cardiac standpoint compared to other sedative-hypnotic agents. Overall, its safety profile is highly favorable, particularly for long-term use in treating insomnia.
The Future of Daridorexant: Current Status and Expanding Horizons
Daridorexant received its first regulatory approval in the United States in January 2022 for the treatment of insomnia characterized by difficulties with sleep onset and/or sleep maintenance. This approval was based on the robust results from phase III clinical trials, which demonstrated significant improvements in both objective and subjective sleep measures. In addition to its U.S. approval, Daridorexant has also received a positive opinion from the Committee for Medicinal Products for Human Use (CHMP) in the European Union, with regulatory reviews ongoing in other countries such as Switzerland and Japan.
Currently, additional clinical trials are underway to assess Daridorexant’s safety and efficacy in broader patient populations, including potential long-term use. Ongoing research in Japan focuses on its effectiveness in Asian populations, while extended safety trials are evaluating its suitability for prolonged treatment. Beyond insomnia, Daridorexant’s dual orexin receptor antagonism is being explored for other sleep disorders and possibly psychiatric conditions, reflecting its versatile therapeutic potential.
As orexin-based therapies gain traction, Daridorexant is emerging as a key player in this evolving field. Its targeted mechanism, favorable safety profile, and proven ability to improve both sleep quality and daytime functioning position it as a breakthrough treatment. Future research is likely to expand its applications, offering hope to a wider range of patients.
 LinkPeptide
LinkPeptide