Ecnoglutide: A Breakthrough cAMP-Biased GLP-1 Analog Transforming Type 2 Diabetes and Obesity Management
Abstract
Ecnoglutide is a novel long-acting, cAMP-biased glucagon-like peptide-1 (GLP-1) analog designed to improve the management of type 2 diabetes and obesity. This GLP-1 analog selectively enhances cAMP production while minimizing receptor internalization, leading to potent glucose control and reduced body weight. Ecnoglutide’s engineered modifications, including the Ala8Val substitution and C18 diacid acylation, confer stability and prolonged half-life, allowing for once-weekly dosing. Preclinical studies demonstrate superior efficacy over semaglutide in reducing blood glucose and promoting weight loss in rodent models. Phase 1 clinical trials in healthy volunteers confirmed the safety, tolerability, and pharmacokinetic properties of ecnoglutide, with only mild adverse effects such as nausea and reduced appetite. With its promising profile, ecnoglutide represents a significant advancement in GLP-1 therapy for metabolic diseases, offering the potential for improved patient outcomes.
The Role of GLP-1 in Metabolic Regulation and the Need for Improved Therapies
Glucagon-like peptide-1 (GLP-1) is a key incretin hormone responsible for regulating glucose metabolism by stimulating insulin secretion in response to food intake[1]. It plays a crucial role in promoting blood sugar control, enhancing satiety, and reducing glucagon release, making it an important target in the management of metabolic disorders such as type 2 diabetes mellitus (T2DM) and obesity. GLP-1 receptor agonists have emerged as a therapeutic approach to enhance these metabolic benefits and improve patient outcomes[2].
Despite the efficacy of GLP-1 receptor agonists, current treatments face several limitations. First-generation GLP-1 analogs, such as exenatide and liraglutide, have short half-lives, necessitating frequent dosing and leading to poor patient compliance. Additionally, gastrointestinal side effects such as nausea and vomiting are common, reducing tolerability[3]. There is a growing need for novel therapies that offer extended half-lives, improved efficacy, and better tolerability.
Ecnoglutide, a newly developed GLP-1 analog, addresses these challenges by incorporating specific modifications that enhance its pharmacokinetic profile. These include the substitution of alanine with valine at position 8 (Ala8Val) and the conjugation of a C18 diacid fatty acid via acylation. These changes extend the half-life and promote receptor bias toward cAMP signaling, which is associated with enhanced metabolic effects and reduced receptor internalization. This design offers a promising solution for addressing the limitations of existing GLP-1 therapies and represents a significant step forward in the treatment of T2DM and obesity.
Ecnoglutide’s Structural Innovations and Biased GLP-1 Receptor Activation
Ecnoglutide was developed with structural modifications designed to overcome the limitations of existing GLP-1 receptor agonists, such as short half-life and frequent dosing requirements. The engineering of ecnoglutide involves a substitution of alanine with valine at position 8 (Ala8Val) and acylation with a C18 diacid fatty acid at lysine residues. This acylation process allows for increased binding to albumin, significantly extending the drug’s half-life and enabling once-weekly administration[4].
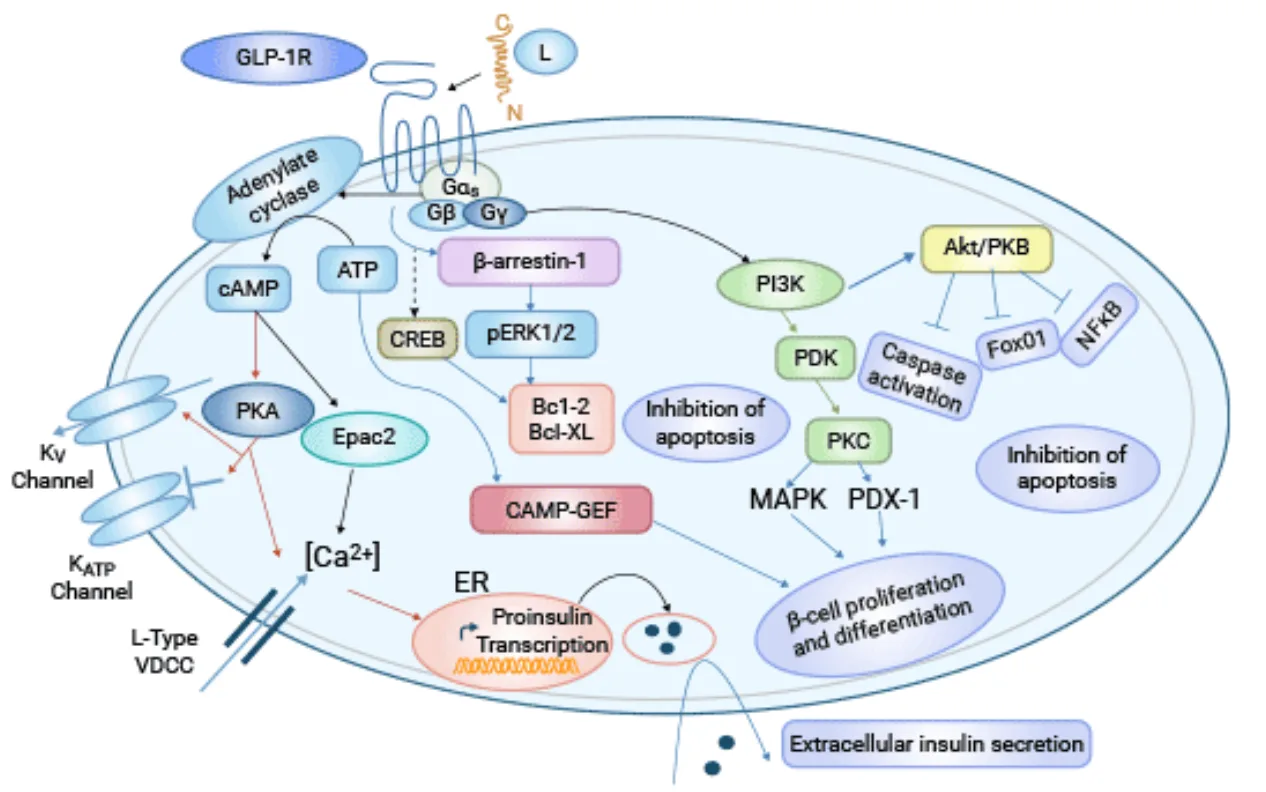
The Ala8Val substitution in ecnoglutide contributes to a cAMP-biased signaling pathway. Upon binding to the GLP-1 receptor, ecnoglutide selectively activates cAMP production, which is critical for stimulating insulin secretion and controlling blood glucose levels. Importantly, this biased signaling minimizes the recruitment of β-arrestin, which is responsible for receptor internalization and potential side effects like gastrointestinal intolerance.
This receptor signaling bias is thought to contribute to ecnoglutide’s enhanced metabolic benefits, including improved glucose control, increased insulin sensitivity, and reduced body weight. In preclinical studies, ecnoglutide demonstrated potent GLP-1 receptor binding, similar to semaglutide, while showing superior efficacy in activating the cAMP pathway.
By optimizing these structural and mechanistic features, ecnoglutide presents a promising therapeutic candidate that addresses key challenges in the management of type 2 diabetes and obesity, offering improved tolerability and longer-lasting effects compared to its predecessors.
Preclinical Success of Ecnoglutide: Superior Glucose Control and Extended Action
Ecnoglutide’s efficacy and pharmacokinetics have been extensively studied in preclinical models, demonstrating its superiority over other GLP-1 analogs like semaglutide. In rodent models of diabetes and obesity, ecnoglutide exhibited significant improvements in both glucose control and weight reduction. In db/db mice, a model of type 2 diabetes, ecnoglutide was shown to significantly reduce fasting blood glucose levels for up to 147 hours after a single dose, outperforming semaglutide in both glucose control and duration of effect[5].
In addition to glucose control, ecnoglutide demonstrated impressive weight loss benefits. In diet-induced obese (DIO) rats, ecnoglutide led to a more pronounced and sustained reduction in body weight compared to semaglutide, even after switching treatment groups. This prolonged weight loss effect is crucial in the management of both diabetes and obesity, as weight reduction improves insulin sensitivity and cardiovascular outcomes.
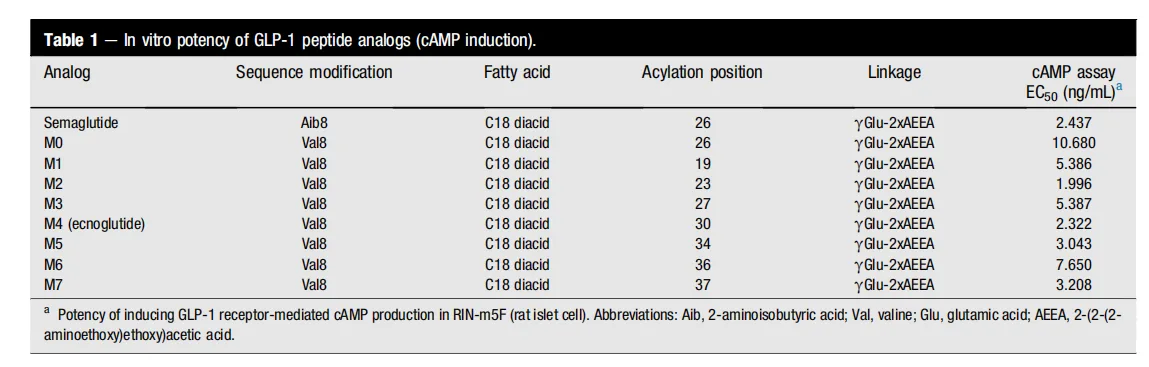
Pharmacokinetically, ecnoglutide benefits from an extended half-life, allowing for once-weekly administration. Its structural modifications, particularly the C18 diacid fatty acid acylation, enhance albumin binding, prolonging its residence time in the body. Preclinical pharmacokinetic studies in rats showed that ecnoglutide and semaglutide have similar Tmax and Cmax values, but ecnoglutide exhibited a longer duration of action, making it an ideal candidate for weekly administration in clinical settings.
These preclinical results underscore ecnoglutide’s potential as a next-generation GLP-1 analog, with superior efficacy and an improved pharmacokinetic profile that may translate into better patient outcomes.
Clinical Validation of Ecnoglutide: Safety, Tolerability, and Pharmacokinetic Insights
Ecnoglutide has shown promising results in early clinical trials, particularly in terms of safety, tolerability, and pharmacokinetics. A Phase 1 clinical study evaluated the safety and pharmacokinetics of ecnoglutide in healthy volunteers through both single ascending dose (SAD) and multiple ascending dose (MAD) cohorts. The study confirmed that ecnoglutide was well tolerated, with mild to moderate adverse events, including nausea, decreased appetite, and headaches. These adverse events are consistent with those seen in other GLP-1 receptor agonists, though no severe or life-threatening events were reported.
The pharmacokinetics of ecnoglutide were highly favorable, supporting its potential for once-weekly administration. The study revealed that ecnoglutide has a long half-life of approximately 124-138 hours, allowing sustained glucose control with weekly dosing[6]. Additionally, the pharmacokinetic profile demonstrated dose-proportional increases in exposure, ensuring predictable drug behavior across different dosing regimens.
Importantly, the clinical study demonstrated no significant accumulation of the drug in the body, even after multiple doses. This is crucial for ensuring long-term safety and minimizing the risk of drug-related adverse effects. The slow systemic absorption, with a median Tmax ranging from 12 to 72 hours, supports its extended action and reduced dosing frequency, which may improve patient compliance.
Overall, ecnoglutide’s clinical safety profile, combined with its favorable pharmacokinetics, positions it as a promising candidate for the treatment of type 2 diabetes and obesity, potentially offering enhanced patient convenience and reduced side effects compared to other GLP-1 receptor agonists.
Ecnoglutide’s Therapeutic Promise: Future Clinical Applications and Broader Impact
Ecnoglutide represents a significant advancement in the development of GLP-1 receptor agonists for the treatment of type 2 diabetes and obesity. Building upon preclinical and Phase 1 clinical studies, further clinical trials are needed to confirm its efficacy in broader patient populations. Ongoing Phase 2 and 3 studies aim to assess its long-term effects on glycemic control, weight reduction, and safety in patients with type 2 diabetes and obesity. These trials will also explore the potential for ecnoglutide to improve cardiovascular outcomes, an important aspect of diabetes management.
One exciting area of future research is the potential for ecnoglutide to be developed as an oral formulation. Preclinical data suggest that ecnoglutide’s gastrointestinal stability is superior to semaglutide, making it a promising candidate for oral delivery. This could further enhance patient compliance by eliminating the need for injections, a common barrier in long-term diabetes management.
In conclusion, ecnoglutide’s novel structure, cAMP-biased signaling, and favorable pharmacokinetic profile offer a promising therapeutic option for improving glucose control and weight management in patients with type 2 diabetes and obesity. As clinical trials progress, ecnoglutide may emerge as a first-line treatment with superior efficacy, tolerability, and convenience, addressing many of the limitations associated with current GLP-1 therapies.
 LinkPeptide
LinkPeptide