Plecanatide: A Breakthrough in Treating Chronic Idiopathic Constipation – Mechanism, Efficacy, and Future Directions
Abstract
Plecanatide, a guanylate cyclase-C (GC-C) agonist, has gained attention as an effective treatment for chronic idiopathic constipation (CIC), a condition that can significantly diminish patients’ quality of life. By mimicking uroguanylin, a natural ligand, Plecanatide activates GC-C receptors in a pH-dependent way, encouraging bowel fluid secretion while avoiding systemic absorption. Clinical trials have shown it to be effective in enhancing bowel movements, improving stool consistency, and alleviating abdominal symptoms, with mild diarrhea being the most common side effect. Compared to traditional laxatives and other newer treatments, Plecanatide offers a more focused approach with fewer adverse effects. Although its cost may pose a challenge for widespread adoption, its demonstrated efficacy and potential use in treating irritable bowel syndrome with constipation (IBS-C) and other gastrointestinal conditions make it a valuable option for clinicians. Ongoing research may further unlock its broader therapeutic applications.
A New Era in Chronic Idiopathic Constipation Treatment
Chronic idiopathic constipation (CIC) is a prevalent gastrointestinal disorder that presents with infrequent, difficult, or incomplete bowel movements, often accompanied by abdominal discomfort and bloating. Unlike constipation associated with underlying organic diseases, CIC lacks an identifiable physiological cause, making its management particularly challenging. Constipation is a major health problem both domestically and globally. The average US and global prevalence is 14%–15%. Two and a half million outpatient doctor visits are attributed to constipation each year.In US alone, constipation results in a significant economic burden with an estimated ≥821 million spent annually on laxatives.Constipation severely impairs quality of life and causes significant psychological distress. Constipation affects females more than males, older individuals, and more people of lower socioeconomic status. Traditional management strategies for CIC often involve lifestyle modifications, such as increased fiber intake and fluid consumption, alongside the use of over-the-counter laxatives. However, these interventions are often insufficient for long-term relief, with many patients experiencing suboptimal results or significant side effects, such as bloating and cramping.
Plecanatide, a novel therapeutic agent, has gained attention for its potential to address these unmet needs. As a guanylate cyclase-C (GC-C) agonist, Plecanatide mimics the action of uroguanylin, a naturally occurring peptide involved in regulating intestinal fluid secretion. By stimulating the GC-C receptor in the gastrointestinal tract, Plecanatide enhances fluid secretion into the intestinal lumen, facilitating easier passage of stools. This mechanism is unique compared to traditional laxatives, as it promotes a more natural physiological response to constipation without significant systemic absorption or adverse effects. Plecanatide thus represents a significant advancement in the treatment of CIC, offering an effective and well-tolerated option for patients who have not responded to conventional therapies.
Innovative Mechanism of Action: How Plecanatide Works
Plecanatide exerts its therapeutic effect by mimicking uroguanylin, a peptide hormone essential for regulating fluid balance in the gastrointestinal tract. Acting as a selective agonist of guanylate cyclase-C (GC-C) receptors, Plecanatide binds to these receptors on the luminal surface of intestinal enterocytes. This binding triggers the conversion of guanosine triphosphate (GTP) into cyclic guanosine monophosphate (cGMP), which promotes the secretion of chloride and bicarbonate into the intestinal lumen. As a result, water is drawn into the bowel, softening stools and improving bowel motility, making it easier for patients to achieve regular bowel movements.
Plecanatide’s distinguishing feature is its pH-dependent binding, designed to mimic uroguanylin’s natural action in the slightly acidic environment of the upper small intestine. This pH-sensitive mechanism ensures its activity is confined to the gastrointestinal tract, minimizing systemic absorption and reducing the risk of systemic side effects, making it a safer option compared to other constipation therapies.
Pharmacokinetically, Plecanatide remains confined to the gastrointestinal system, where it is metabolized into smaller peptides and amino acids. Its lack of systemic distribution further enhances its safety profile, making it an ideal candidate for long-term use in patients with chronic idiopathic constipation.
Efficacy and Safety: Clinical Validation of Plecanatide
Plecanatide has undergone extensive clinical trials to assess its efficacy and safety in treating chronic idiopathic constipation (CIC). Results from Phase I, II, and III studies consistently highlight its therapeutic value, showing significant improvements in bowel function and patient-reported quality of life. Patients treated with Plecanatide experienced increased frequency of complete spontaneous bowel movements (CSBMs), improved stool consistency, and reduced straining. These outcomes were markedly superior to placebo, establishing Plecanatide as a reliable option for patients unresponsive to conventional therapies.
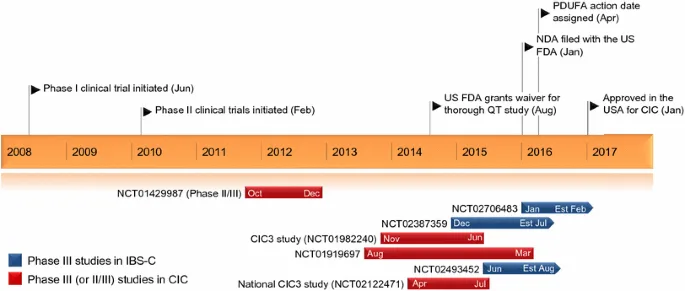
In terms of safety, Plecanatide exhibits a favorable profile. The most commonly reported adverse event across trials has been mild to moderate diarrhea, occurring in a minority of patients. Importantly, the incidence of diarrhea is dose-dependent and typically resolves without the need for medical intervention or discontinuation of treatment. Other side effects, such as nausea and abdominal pain, have been reported infrequently and are generally mild. Moreover, Plecanatide’s lack of systemic absorption significantly reduces the risk of severe adverse events or drug interactions, a concern with other pharmacological agents used in CIC management.
Long-term safety data from extended open-label trials further support Plecanatide’s tolerability, with few patients discontinuing treatment due to adverse effects. This safety, combined with its clinical efficacy, positions Plecanatide as a valuable treatment option for patients suffering from CIC, particularly those seeking alternatives to conventional laxatives or other pro-motility agents.
How Plecanatide Compares to Other Treatment Options
Plecanatide presents a notable advantage over both traditional and newer treatments for chronic idiopathic constipation (CIC) due to its targeted mechanism of action and favorable safety profile. Conventional therapies, such as osmotic and stimulant laxatives, primarily function by drawing water into the bowel or stimulating peristalsis. While commonly used, these treatments often yield inconsistent efficacy and are associated with significant side effects, including bloating, cramping, and electrolyte imbalances. Additionally, many patients achieve only partial symptom relief, underscoring the need for alternative therapeutic options.
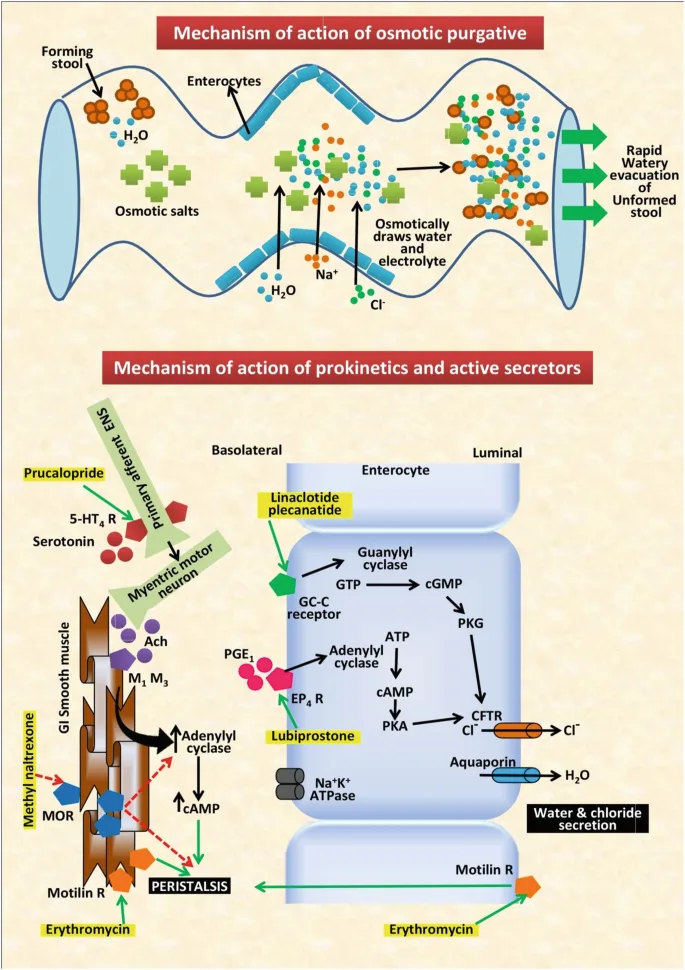
Compared to traditional laxatives, Plecanatide provides a more natural and physiological approach to managing CIC. As a guanylate cyclase-C (GC-C) agonist, it specifically targets the underlying pathophysiology of fluid secretion in the intestines, promoting bowel movements without disrupting electrolyte balance. This targeted action, combined with its pH-dependent receptor activation, results in fewer adverse effects and more consistent therapeutic outcomes.
Plecanatide also distinguishes itself from other newer agents, such as Linaclotide, another GC-C agonist. While both drugs share similar mechanisms, Plecanatide’s pH sensitivity more closely mimics the natural function of uroguanylin, offering a potentially better side-effect profile with reduced incidence of diarrhea. In addition, Plecanatide’s minimal systemic absorption provides an added safety benefit, particularly for patients with concerns about drug interactions or long-term systemic exposure.
In terms of cost-effectiveness, Plecanatide is priced comparably to other newer agents like Linaclotide and Lubiprostone, though its enhanced tolerability and patient satisfaction may justify its use as a preferred second-line treatment when traditional laxatives fail.
Looking Ahead: The Future of Plecanatide in CIC Therapy
Plecanatide marks a meaningful advancement in the therapeutic landscape for chronic idiopathic constipation (CIC), providing a well-tolerated and effective option for patients who have found conventional treatments inadequate. Its targeted mechanism of action, which mirrors the natural function of uroguanylin in activating guanylate cyclase-C (GC-C) receptors, enables precise regulation of intestinal fluid secretion. This action facilitates smoother and more consistent bowel movements, improving patient outcomes. Moreover, Plecanatide’s specificity in the gastrointestinal tract significantly reduces the risk of systemic side effects that are often associated with other treatments, positioning it as an optimal choice for long-term management of CIC.
Looking ahead, Plecanatide’s potential extends beyond CIC. Its role in treating irritable bowel syndrome with constipation (IBS-C) has already been established, and ongoing research into its effects on broader gastrointestinal disorders, such as inflammatory bowel disease (IBD) and colorectal cancer, suggests promising new applications. These developments highlight the growing importance of GC-C agonists in gastrointestinal health.
However, challenges remain, particularly regarding the drug’s affordability and long-term safety profile. While Plecanatide’s efficacy and tolerability have been demonstrated, further studies will be crucial to solidifying its position in both CIC and related conditions. As research continues, Plecanatide is poised to become an indispensable tool in the management of constipation-related disorders, providing clinicians and patients with a robust, reliable, and innovative therapeutic option.
 LinkPeptide
LinkPeptide