Fluorescent Cyclic Peptides: Advancing Precision in Cell Imaging and Molecular Diagnostics
Introduction: Why Cell Imaging Needs Better Molecular Probes
Modern biological research increasingly relies on high-resolution cell imaging to understand complex cellular processes, disease progression, and therapeutic responses. However, conventional imaging probes often lack the specificity, stability, or sensitivity required for precise molecular visualization.
Peptides have emerged as promising tools due to their ability to bind selectively to biological targets, including receptors, enzymes, and intracellular proteins. Yet, linear peptides often suffer from poor stability and rapid degradation.
To address these limitations, researchers have turned to fluorescent cyclic peptides—a class of engineered molecules that combine structural stability with powerful imaging capabilities.
What Are Fluorescent Cyclic Peptides?
Fluorescent cyclic peptides are conformationally constrained peptide structures that are chemically modified with fluorescent labels to enable visualization in biological systems.
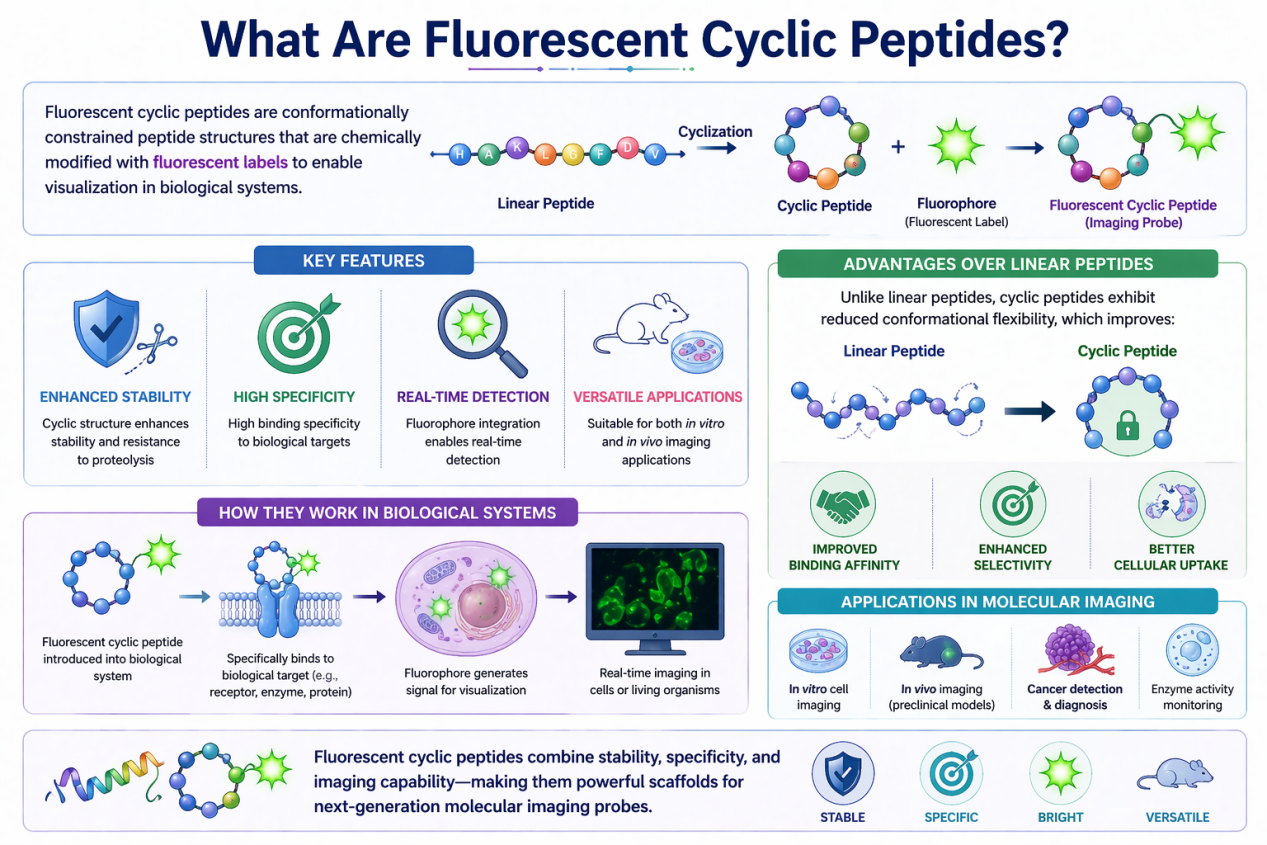
Key Features:
- Cyclic structure enhances stability and resistance to proteolysis
- High binding specificity to biological targets
- Fluorophore integration enables real-time detection
- Suitable for both in vitro and in vivo imaging applications
Unlike linear peptides, cyclic peptides exhibit reduced conformational flexibility, which improves:
- Target binding affinity
- Selectivity
- Cellular uptake
These advantages make them highly effective scaffolds for molecular imaging probes .
Why Cyclization Matters in Peptide Design
Enhanced Stability
Linear peptides are highly susceptible to enzymatic degradation, particularly in disease environments where proteases are abundant. Cyclization significantly improves resistance to these enzymes.
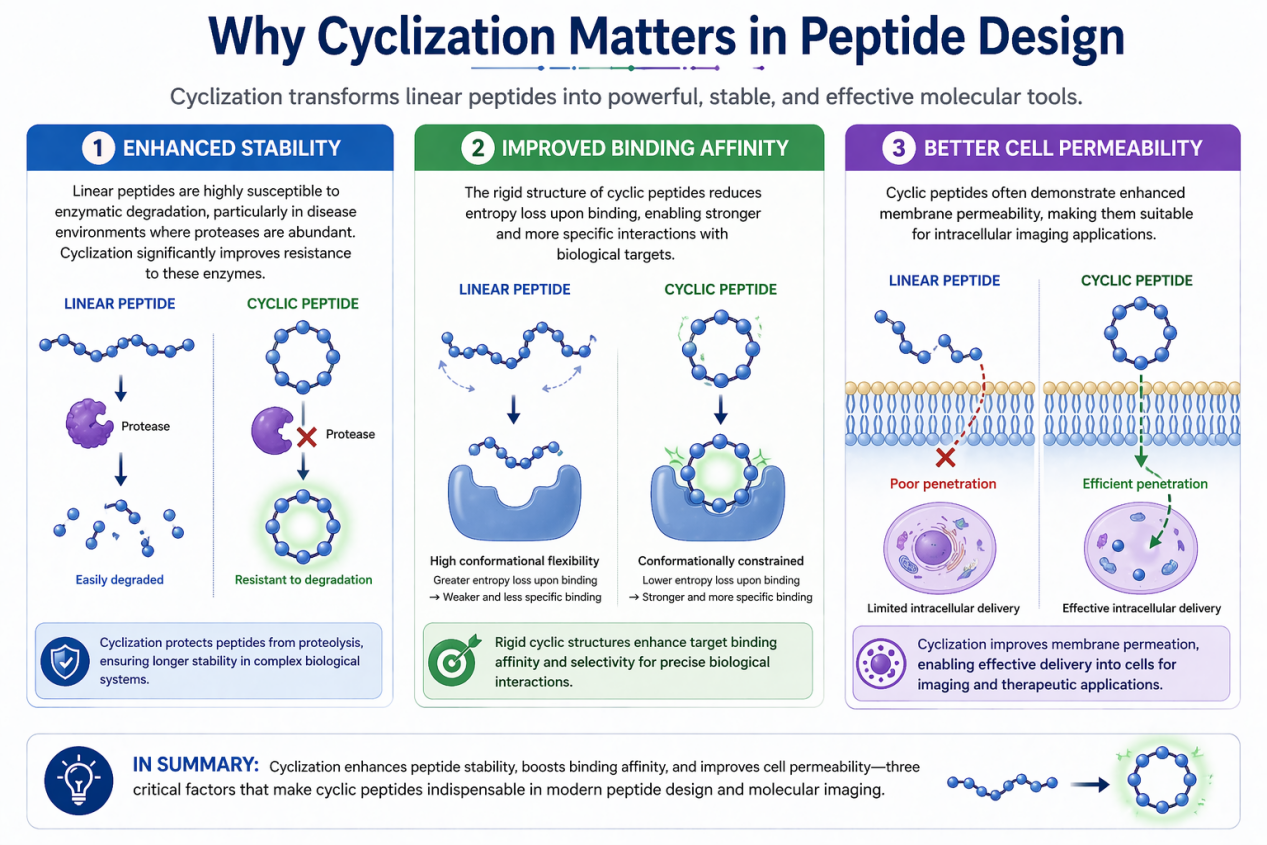
Improved Binding Affinity
The rigid structure of cyclic peptides reduces entropy loss upon binding, enabling stronger and more specific interactions with biological targets.
Better Cell Permeability
Cyclic peptides often demonstrate enhanced membrane permeability, making them suitable for intracellular imaging applications.
Strategies for Designing Fluorescent Cyclic Peptides
Fluorophore Incorporation
Most cyclic peptides do not naturally emit detectable signals. Therefore, external fluorophores are introduced to enable imaging.
Common Fluorophores:
- Fluorescein
- Rhodamine
- Cyanine dyes
- BODIPY derivatives
These fluorophores provide:
- High brightness
- Tunable emission wavelengths
- Compatibility with imaging systems
Labeling Strategies
Pre-Cyclization Labeling
- Fluorophore attached before ring formation
- Targets N- or C-terminal groups
Post-Cyclization Labeling
- Most common approach
- Targets side chains (e.g., Lys, Cys, Ser)
Use of Linkers
- PEG spacers reduce steric hindrance
- Improve solubility and circulation time
Fluorogenic Peptides
An emerging innovation is activatable (fluorogenic) peptides, which:
- Remain non-fluorescent until interacting with targets
- Reduce background noise
- Improve signal-to-noise ratio
Mechanisms include:
- FRET (Förster Resonance Energy Transfer)
- Aggregation-induced emission (AIE)
- Photoinduced electron transfer (PeT)
Structural Diversity of Cyclic Peptides
Cyclic peptides can be engineered in multiple forms:
Cyclization Types
- Head-to-tail cyclization
- Side-chain-to-side-chain linkages
- Disulfide bridges
- Stapled peptides
These variations allow fine-tuning of:
- Stability
- Bioactivity
- Target specificity
Applications in Cell Imaging
Fluorescent cyclic peptides are widely used in biological imaging, particularly for disease-related studies.
Targeting Cell Surface Receptors
One of the most important applications is targeting overexpressed receptors in cancer cells.
Example: RGD Peptides
- Bind integrin receptors (e.g., αvβ3)
- Enable tumor imaging and detection
- Improve targeting accuracy
These peptides are widely used in oncology imaging studies .
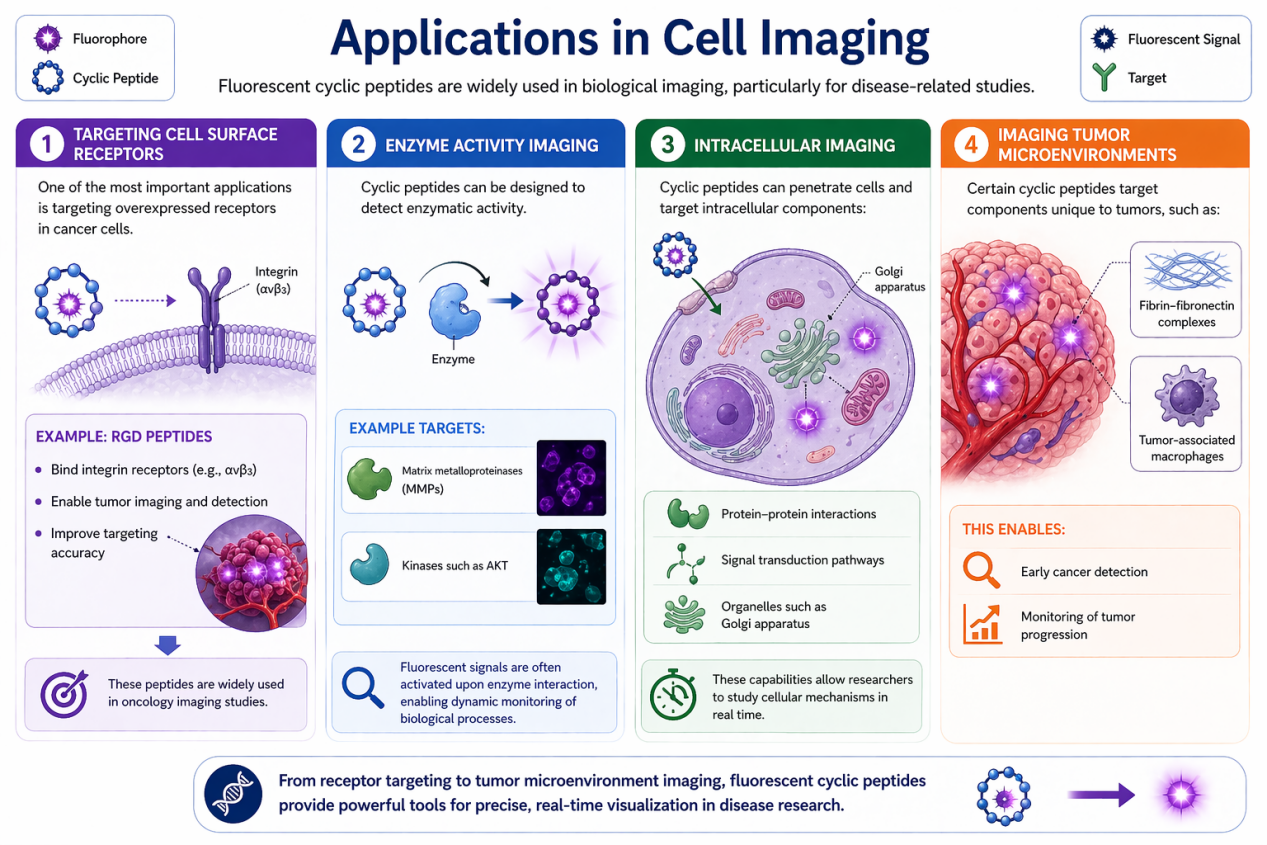
Enzyme Activity Imaging
Cyclic peptides can be designed to detect enzymatic activity.
Example Targets:
- Matrix metalloproteinases (MMPs)
- Kinases such as AKT
Fluorescent signals are often activated upon enzyme interaction, enabling dynamic monitoring of biological processes.
Intracellular Imaging
Cyclic peptides can penetrate cells and target intracellular components:
- Protein–protein interactions
- Signal transduction pathways
- Organelles such as Golgi apparatus
These capabilities allow researchers to study cellular mechanisms in real time.
Imaging Tumor Microenvironments
Certain cyclic peptides target components unique to tumors, such as:
- Fibrin–fibronectin complexes
- Tumor-associated macrophages
This enables:
- Early cancer detection
- Monitoring of tumor progression
In Vivo Imaging Applications
Fluorescent cyclic peptides are not limited to cell-based assays—they are also powerful tools for in vivo imaging.
Advantages:
- Real-time visualization
- High tissue penetration (especially with NIR dyes)
- Low background fluorescence
Near-Infrared (NIR) Imaging
NIR fluorophores (650–900 nm) are particularly valuable because they:
- Penetrate deeper into tissues
- Reduce autofluorescence
- Improve imaging clarity
These properties make NIR-labeled cyclic peptides ideal for:
- Tumor detection
- Fluorescence-guided surgery
Advantages Over Traditional Imaging Probes
Compared to antibodies and small molecules, fluorescent cyclic peptides offer:
Key Benefits
- Smaller size → better tissue penetration
- Faster targeting kinetics
- Lower immunogenicity
- Easier synthesis and modification
Current Challenges in the Field
Despite their advantages, several challenges remain:
Optimization of Fluorophores
- Need for improved brightness and stability
- Development of better NIR dyes
Target Specificity
- Avoiding off-target interactions
Scalability
- Manufacturing complexity for clinical use
Future Outlook: Toward Smart Imaging Probes
The field is rapidly evolving toward next-generation peptide probes, including:
Smart (Activatable) Probes
- Turn-on fluorescence upon target binding
- High diagnostic precision
Theranostic Peptides
- Combine imaging and therapy
- Enable simultaneous detection and treatment
Multimodal Imaging
- Compatible with fluorescence + MRI + PET
These innovations are expected to play a critical role in:
- Personalized medicine
- Early disease diagnosis
- Drug discovery
How LinkPeptide Supports Fluorescent Peptide Research
At LinkPeptide, we support advanced peptide research by offering:
- Custom cyclic peptide synthesis
- Fluorophore conjugation services
- Peptide modification (stapling, cyclization)
- High-quality analytical validation
Our expertise helps researchers develop precision peptide tools for imaging and therapeutic applications.
Conclusion
Fluorescent cyclic peptides represent a powerful intersection of peptide chemistry and molecular imaging, offering unmatched specificity, stability, and versatility.
By enabling precise visualization of biological processes—from receptor binding to intracellular signaling—these molecules are transforming how researchers study disease and develop new therapies.
As synthetic methods and fluorophore technologies continue to advance, fluorescent cyclic peptides are poised to become indispensable tools in modern biomedical research.
Reference
Mendive‐Tapia, L., Wang, J., & Vendrell, M. (2021). Fluorescent cyclic peptides for cell imaging. Peptide Science, 113(1), e24181.
https://doi.org/10.1002/pep2.24181
Guarracino, D. A., Riordan, J. A., Barreto, G. M., Oldfield, A. L., Kouba, C. M., & Agrinsoni, D. (2019). Macrocyclic control in helix mimetics. Chemical reviews, 119(17), 9915-9949.
http://orcid.org/0000-0001-6775-6564
Soriano, A., Vendrell, M., Gonzalez, S., Mallol, J., Albericio, F., Royo, M., … & Casado, V. (2010). A hybrid indoloquinolizidine peptide as allosteric modulator of dopamine D1 receptors. The Journal of pharmacology and experimental therapeutics, 332(3), 876-885.
https://doi.org/10.1124/jpet.109.158824
Cheng, Z., Kuru, E., Sachdeva, A., & Vendrell, M. (2020). Fluorescent amino acids as versatile building blocks for chemical biology. Nature Reviews Chemistry, 4(6), 275-290.
 LinkPeptide
LinkPeptide