Peptide-Based Nanoparticles for Targeted Drug Delivery, Imaging, and Cancer Immunotherapy
Abstract
As cancer therapies evolve, precision and adaptability have become critical. Peptide-functionalized nanoparticles are emerging as next-generation tools that combine the targeting accuracy of peptides with the delivery efficiency of nanomaterials. These smart systems enable selective drug, gene, and natural product delivery to tumors while minimizing off-target effects. Peptides serve not only as targeting ligands but also as therapeutic agents and structural components in self-assembled nanocarriers. Beyond treatment, peptide-based nanoparticles play vital roles in immunotherapy, cancer vaccines, and real-time diagnostic imaging—paving the way for truly integrated, personalized oncology. Although challenges remain in clinical translation and system complexity, the future of cancer therapy is increasingly defined by multifunctional platforms that can treat, track, and adapt. This blog explores the mechanisms, applications, and future directions of peptide-based nanoparticles and their transformative impact on cancer care.
From Toxic to Targeted: Why Cancer Therapy Needs a Smarter Delivery System
Despite decades of advancement, cancer treatment still faces significant challenges. Chemotherapy, radiation, and surgery—the pillars of conventional therapy—are often hampered by limited specificity, treatment resistance, and collateral damage to healthy tissues. Many patients endure severe side effects, while some cancers persist or return, shielded by complex tumor microenvironments and adaptive survival mechanisms.
In recent years, nanomedicine has emerged as a transformative solution. By engineering nanoparticles—ultrafine carriers measured in billionths of a meter—scientists can encapsulate therapeutic agents and direct them precisely to tumor sites. These nanostructures can improve drug solubility, prolong circulation time, and release their cargo in response to specific biological triggers. Yet, even the most sophisticated nanoparticles require accurate guidance systems to achieve true precision.
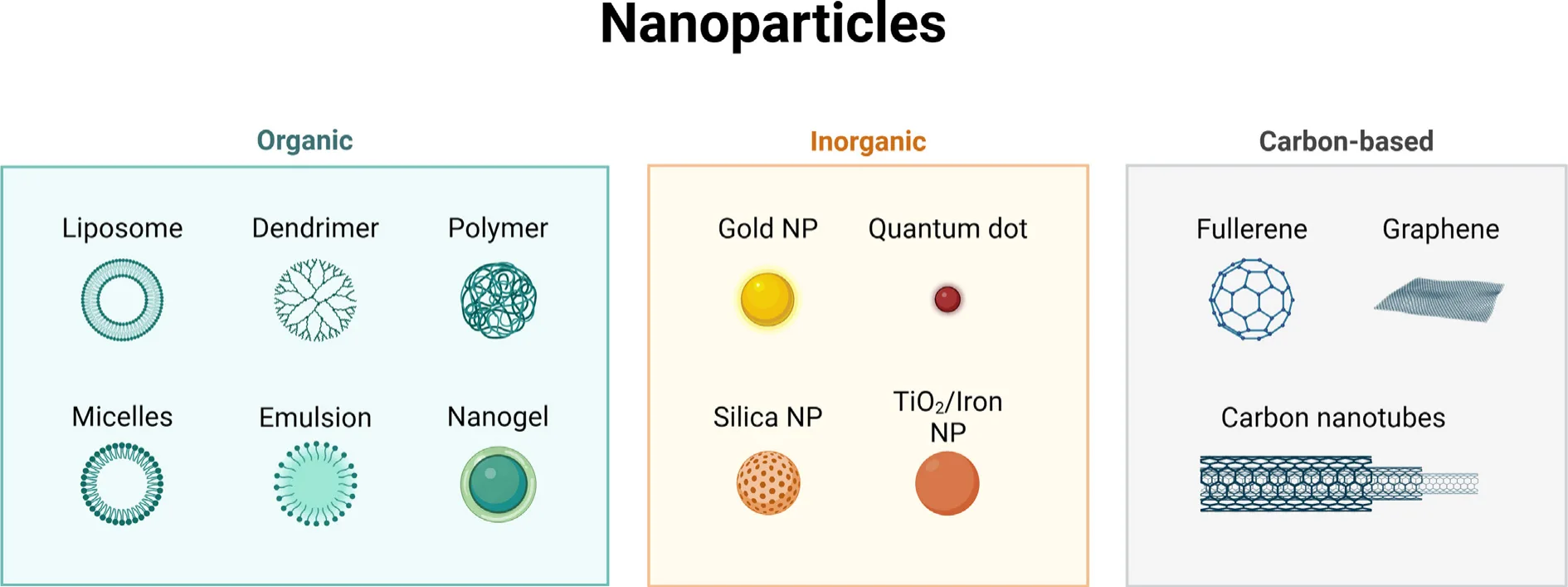
This is where peptides come in. These short chains of amino acids offer remarkable benefits: they are highly biocompatible, structurally versatile, and capable of binding selectively to receptors that are overexpressed on cancer cells. Peptides can penetrate cell membranes, respond to stimuli like pH or redox gradients, and even self-assemble into functional nanostructures. When integrated with nanoparticles—either as targeting ligands, structural components, or therapeutic agents—peptides transform these carriers into smart, multifunctional systems.
The convergence of peptide technology and nanomedicine is ushering in a new era of cancer therapy. These hybrid systems promise not only more accurate drug delivery but also enhanced diagnostic imaging, immunomodulation, and reduced systemic toxicity. In the sections that follow, we’ll explore how peptide-functionalized nanoparticles are reshaping the landscape of oncology—one molecular innovation at a time.
Peptides in Control: Guiding, Building, and Powering Nanomedicine
Peptides aren’t just passive passengers in cancer therapy—they’re active engineers, shaping how nanoparticles behave, where they go, and what they do once they arrive. When thoughtfully designed and integrated, peptides enable nanocarriers to overcome some of the most persistent barriers in cancer treatment: poor tumor penetration, lack of cellular specificity, and inefficient internalization.
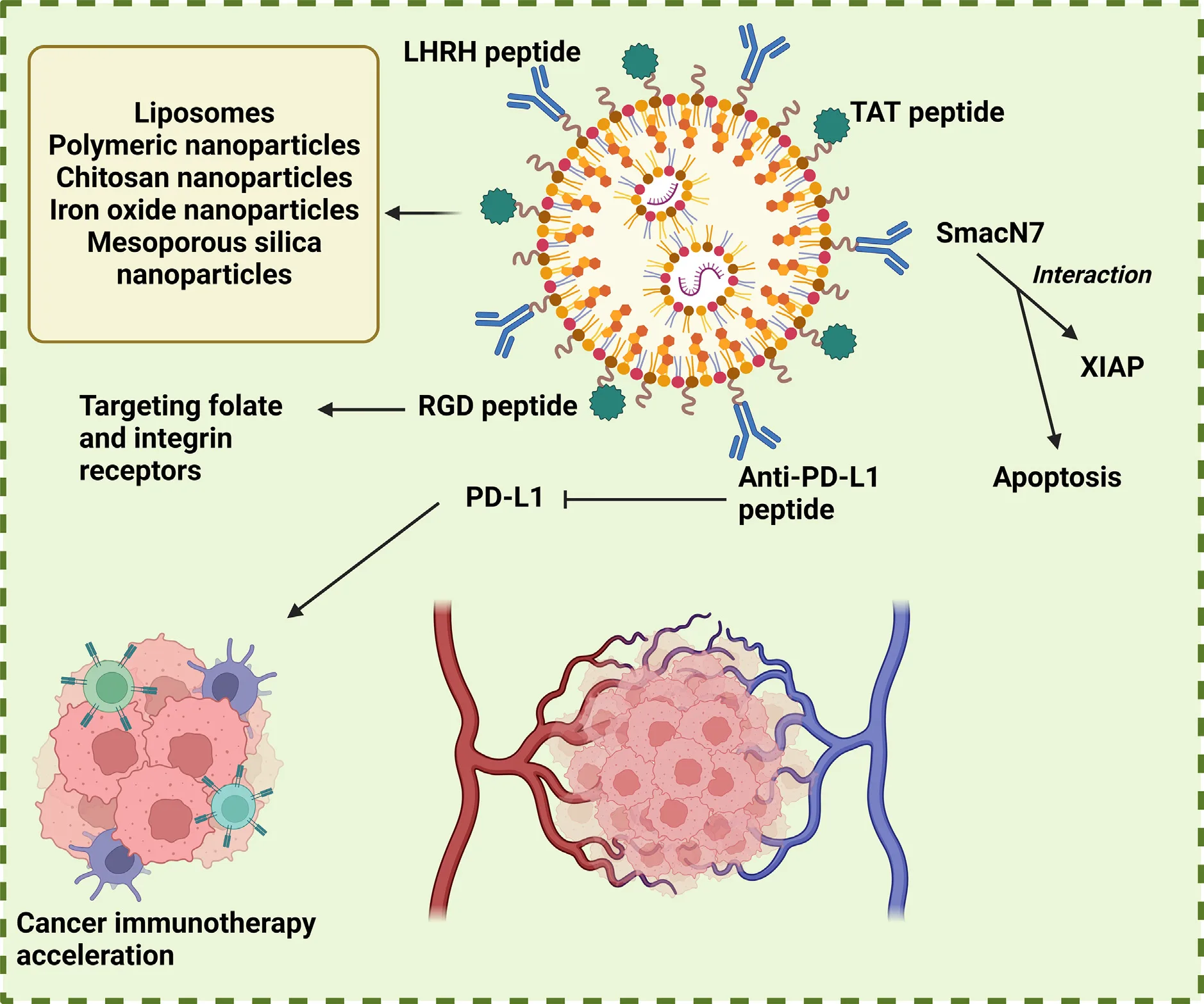
One of the most important roles peptides play is as targeting ligands. Certain peptides can bind selectively to receptors that are overexpressed on cancer cells or the surrounding vasculature. For example, RGD peptides are well-known for their ability to target integrin receptors involved in tumor angiogenesis and metastasis. When these ligands are conjugated to nanoparticles, they guide the therapeutic payload directly to cancerous tissues, reducing the risk of harming healthy cells.
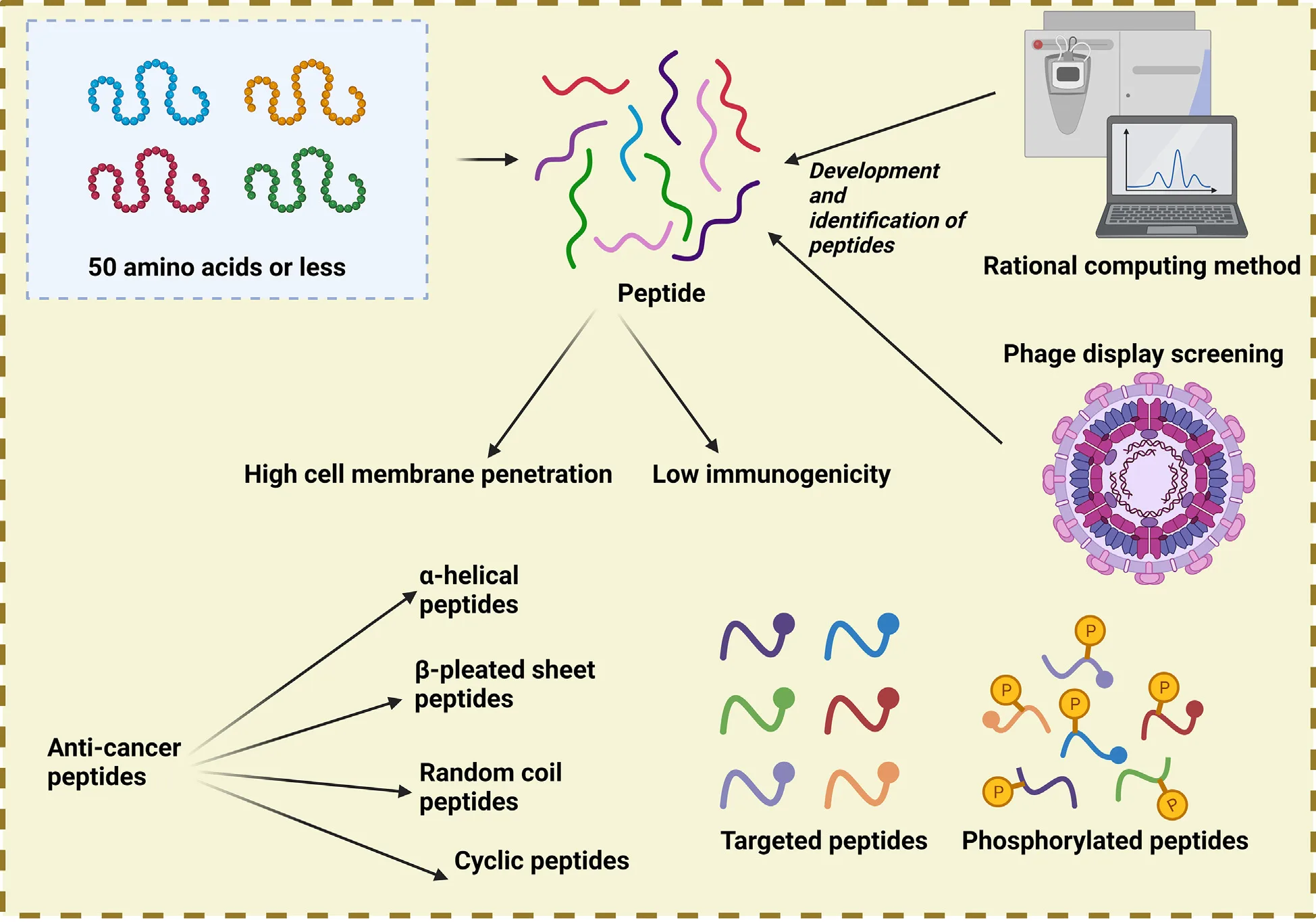
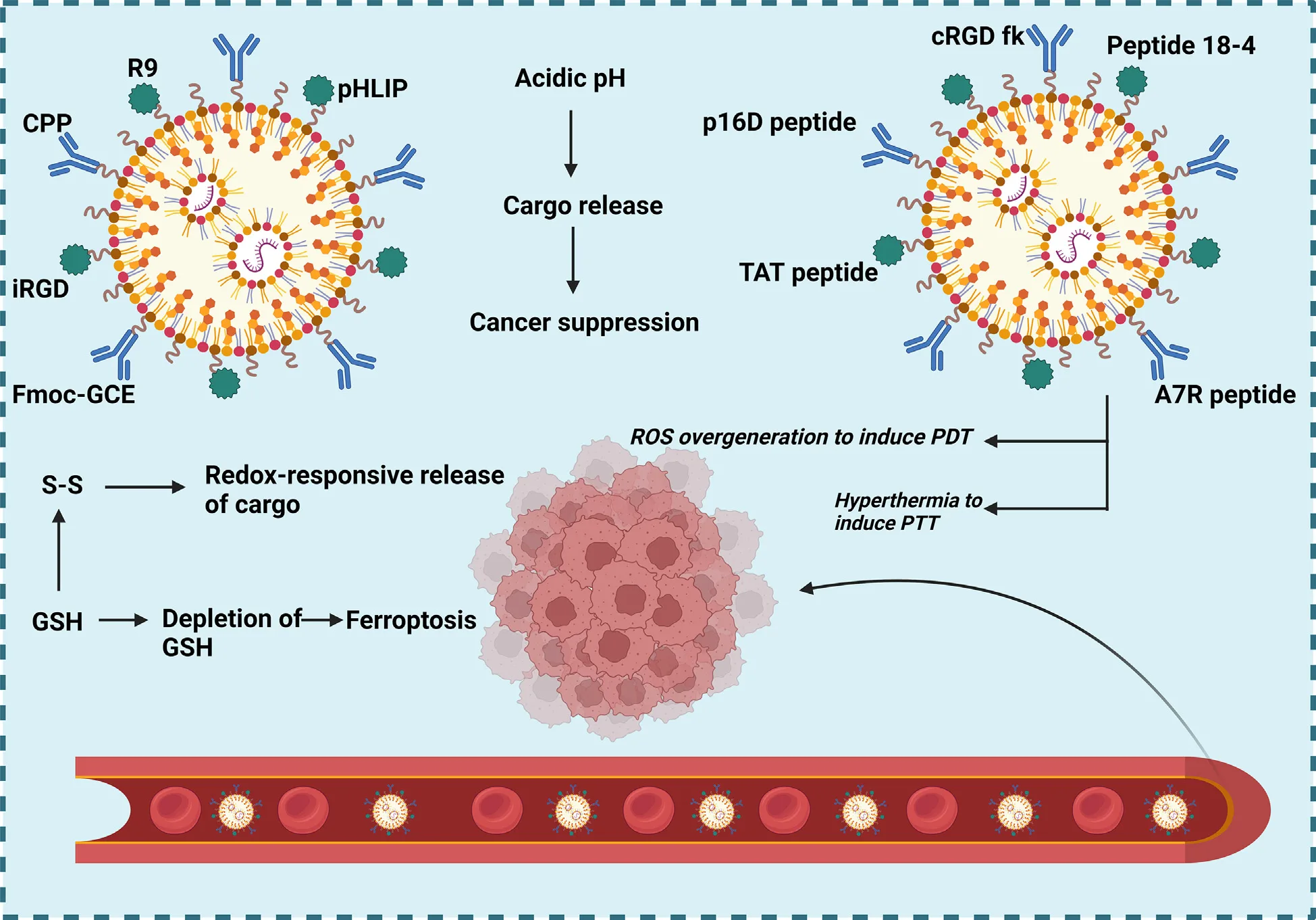
Peptides also serve as building blocks for self-assembly. Their sequences can be designed to form stable nanostructures—such as micelles, nanofibers, or hydrogels—through non-covalent interactions like hydrogen bonding, hydrophobic forces, or electrostatic attraction. These self-assembled systems not only enhance drug loading and stability but can also respond dynamically to the tumor microenvironment, such as releasing drugs in acidic or redox-sensitive conditions.
Beyond targeting and assembly, peptides can act as active therapeutic agents themselves. Many anticancer peptides (ACPs) directly induce apoptosis or disrupt cancer cell membranes. Some peptides modulate immune checkpoints, inhibit oncogenic signaling pathways, or interfere with transcription factors critical to tumor survival.
To optimize performance, researchers often modify peptides by cyclization, PEGylation, or the substitution of L-amino acids with their D-forms. These adjustments improve stability, circulation time, and resistance to enzymatic degradation, ensuring that peptides maintain functionality in the bloodstream and reach their intended targets intact.
In short, peptides are not merely embellishments on the surface of nanoparticles—they are precision tools that transform nanocarriers into intelligent systems capable of seeking, sensing, and striking cancer cells with unprecedented accuracy.
Precision Delivery in Action: Drugs, Genes, and Natural Compounds
In cancer therapy, delivery is everything. It’s not just about what drug you use—it’s how, where, and when it reaches the tumor. This is where peptide-based nanoparticles excel, offering unprecedented control over therapeutic delivery. Whether it’s a chemotherapy agent, a gene-silencing molecule, or a natural compound, peptides help guide these payloads past biological defenses and directly into the heart of the tumor.
Precision Chemotherapy
Traditional chemotherapy drugs like doxorubicin (DOX), paclitaxel, and cisplatin are powerful—but also indiscriminate. By loading them into peptide-functionalized nanoparticles, researchers can concentrate their effects where they’re needed most. For example, nanoparticles modified with VATANST peptides target vimentin, a protein overexpressed on colon tumor cells, while RGD-functionalized carriers home in on integrin receptors in metastatic sites.
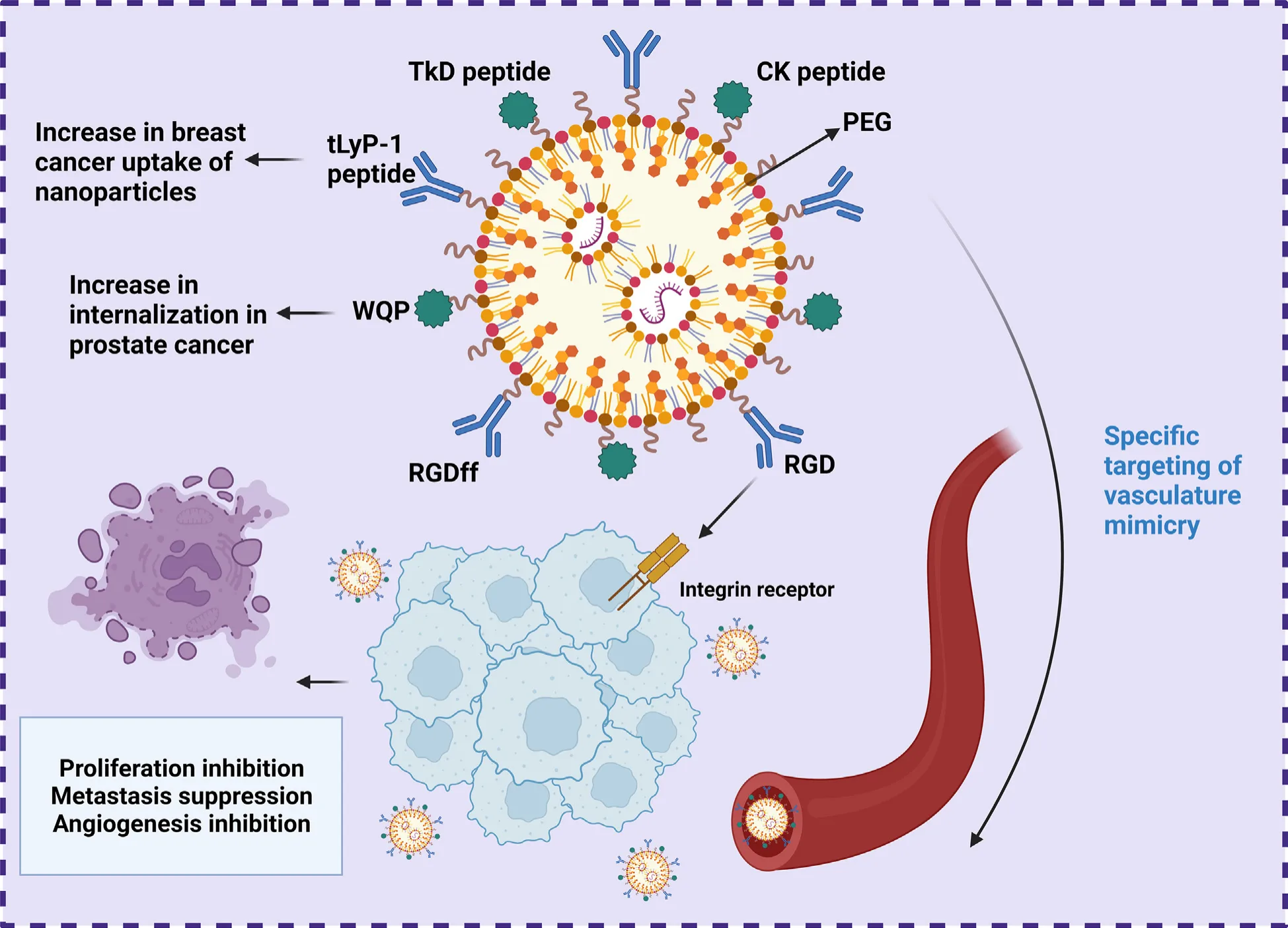
These peptide-guided systems don’t just find their target—they respond to it. Many are engineered to release their cargo in response to tumor-specific cues, such as elevated glutathione (GSH) levels or acidic pH. This kind of stimuli-responsive release ensures that the drugs stay inactive in healthy tissues and activate only in the tumor microenvironment, minimizing collateral damage.
Gene Therapy, Reloaded
Peptide-based nanocarriers are also redefining gene delivery in oncology. They can transport small interfering RNA (siRNA), short hairpin RNA (shRNA), and plasmids with high specificity and cellular uptake. For example, combining LL37 peptides with VAV1-targeting siRNA in PLGA nanoparticles has been shown to inhibit pancreatic cancer metastasis. Other formulations use fusogenic peptides like GALA to promote endosomal escape, ensuring that the genetic cargo reaches the cytoplasm intact and functional.
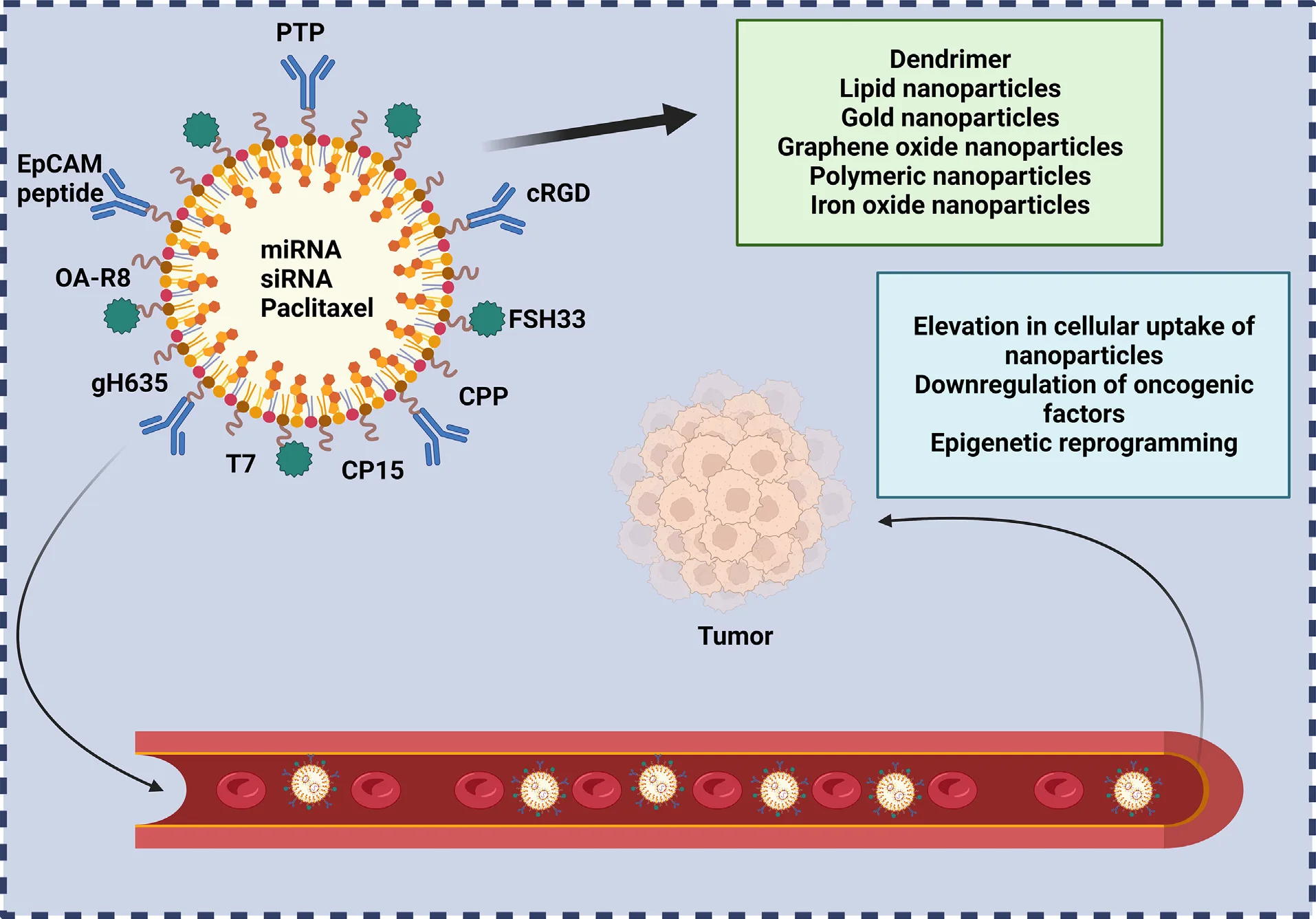
What makes peptide-functionalized nanoparticles especially powerful is their ability to co-deliver genes and drugs, amplifying therapeutic impact. In one example, glycol chitosan nanostructures co-delivered DOX and anti-PD-L1 peptides to induce immune activation while simultaneously killing cancer cells—an elegant synergy of chemotherapy and immunotherapy.
Natural Products, Supercharged
Many plant-derived compounds like curcumin and resveratrol hold great promise in cancer therapy but suffer from poor solubility and fast metabolism. Nanoparticles solve these problems, and peptides bring them home. For instance, PD-L1-binding peptides have been used to direct curcumin-loaded serum albumin nanoparticles to breast tumors, enhancing their apoptotic effects. In other cases, peptides guide resveratrol to EGFR-overexpressing head and neck cancers or facilitate bone-targeted delivery in metastatic disease.
Smarter Combinations, Better Outcomes
The real power of peptide-based nanomedicine lies in its combinatorial capacity. Researchers are developing systems that simultaneously deliver multiple drugs, pair chemotherapy with immunotherapy, or combine phototherapy with gene silencing. These platforms enable personalized treatment strategies, offering flexibility and adaptability in tackling the complex biology of cancer.
In all these applications, peptides transform nanoparticles from simple carriers into intelligent delivery systems—capable of navigating the bloodstream, homing in on tumors, responding to microenvironmental triggers, and unleashing multi-pronged attacks on cancer cells.
Beyond the Tumor: Immune Activation, Imaging, and Theranostic Potential
Peptide-based nanoparticles aren’t limited to drug delivery—they’re also unlocking powerful tools for immunotherapy, cancer vaccines, and diagnostic imaging. This shift from treatment to multi-functionality represents a leap toward integrated, precision oncology.
Nanovaccines and Immune Activation
One of the most exciting applications lies in cancer immunotherapy. Peptides can guide nanoparticles to immune cells, helping activate the body’s natural defenses. For instance, TY peptides can target dendritic cells and promote the maturation of CD8+ cytotoxic T cells, essential for attacking tumors. Other systems carry PD-L1-blocking peptides, disrupting cancer’s ability to evade immune detection. These nanovaccines help “teach” the immune system to recognize and attack tumor cells, potentially offering long-lasting protection.
Even B cells can be engaged. In ovarian cancer models, Sp17-derived peptide vaccines altered immunoglobulin profiles, enhancing antibody responses against tumors. This represents a promising step toward personalized immunization strategies.
Imaging and Theranostics
On the diagnostic front, peptide-functionalized nanoparticles offer high specificity for tumor visualization. Gold nanoparticles modified with triptorelin peptides or chlorotoxin can enhance CT and SPECT imaging of brain tumors. In magnetic resonance imaging (MRI), iron oxide nanoparticles conjugated with peptides show enhanced tumor accumulation and contrast.
The combination of therapy and imaging—known as theranostics—is already in motion. For example, PEG-PPa nanoparticles with the TRP2 peptide enable PET imaging while simultaneously delivering immune-stimulating payloads to treat melanoma.
These innovations represent more than convenience—they offer real-time monitoring, improved targeting, and integrated treatment in a single platform, helping doctors adapt strategies and track outcomes with greater precision.
What’s Next: Challenges, Gaps, and the Future of Precision Oncology
Peptide-functionalized nanoparticles are rapidly reshaping the future of cancer therapy—offering a fusion of precision, adaptability, and multifunctionality. They can target tumors, evade healthy tissue, deliver drugs and genes, stimulate the immune system, and even track disease progression through advanced imaging techniques. But despite their enormous promise, several challenges remain.
Current research has only scratched the surface of what’s possible. Magnetic-, enzyme-, and temperature-responsive systems are still underexplored. Integrating nanoparticles with exosomes or real-time tumor biomarkers could offer smarter delivery and personalized response tracking. Furthermore, clinical translation remains a hurdle, with many promising systems still confined to lab models due to scalability, manufacturing complexity, or safety validation.
Still, the momentum is undeniable. As materials science, immunology, and nanotechnology converge, peptide-based nanomedicine is poised to become a cornerstone of precision oncology. It offers a path toward not just treating cancer—but understanding it, visualizing it, and ultimately defeating it with minimal harm and maximum efficacy.
The age of smart therapeutics has arrived—and peptides are leading the way.
Reference
Dai, J., Ashrafizadeh, M., Aref, A. R., Sethi, G., & Ertas, Y. N. (2024). Peptide-functionalized,-assembled and-loaded nanoparticles in cancer therapy. Drug Discovery Today, 29(7), 103981.
https://doi.org/10.1016/j.drudis.2024.103981
Ferrari, M. (2005). Cancer nanotechnology: opportunities and challenges. Nature reviews cancer, 5(3), 161-171.
https://doi.org/10.1038/nrc1566
Shi, J., Kantoff, P. W., Wooster, R., & Farokhzad, O. C. (2017). Cancer nanomedicine: progress, challenges and opportunities. Nature reviews cancer, 17(1), 20-37.
https://doi.org/10.1038/nrc.2016.108
Shi, J., Liu, S., Yu, Y., He, C., Tan, L., & Shen, Y. M. (2019). RGD peptide-decorated micelles assembled from polymer–paclitaxel conjugates towards gastric cancer therapy. Colloids and Surfaces B: Biointerfaces, 180, 58-67.
 LinkPeptide
LinkPeptide