Engineering Esc(1-21): Isopeptide Bond Positioning for Optimized Antimicrobial Performance
Abstract
Antimicrobial peptides (AMPs) are promising candidates for combating multidrug-resistant pathogens, yet their therapeutic potential is often limited by host cytotoxicity and proteolytic instability. This study focuses on Esculentin-1a(1–21)NH₂ [Esc(1-21)], a frog-skin–derived AMP with potent Gram-negative activity, and explores a novel optimization strategy: site-specific incorporation of an isopeptide bond at selected lysine residues. Five analogs (ε5, ε9, ε10, ε12, ε20) were synthesized and evaluated for antimicrobial potency, antibiofilm activity, membrane disruption, cytotoxicity, plasma stability, and structural behavior. Results revealed a clear position-dependent effect: the C-terminal analog, Esc(1-21)ε20, preserved or enhanced antimicrobial activity, displayed strong antibiofilm effects, reduced cytotoxicity, and improved proteolytic resistance, while central substitutions impaired performance. These findings demonstrate that precise backbone engineering can fine-tune AMP properties, offering a promising pathway for developing stable, selective, and effective peptide-based therapeutics against resistant bacterial infections.
Rethinking Antimicrobials: Peptide Innovation in the Age of Resistance
The escalating threat of antimicrobial resistance has become a defining challenge in modern medicine, with multidrug-resistant pathogens increasingly outpacing the discovery of effective treatments. This crisis has intensified the search for alternatives to conventional antibiotics, and antimicrobial peptides (AMPs) have emerged as particularly promising candidates. These short, cationic, amphipathic molecules are key components of innate immunity across diverse organisms, exerting their activity primarily through physical disruption of microbial membranes—a mechanism inherently less susceptible to resistance development.
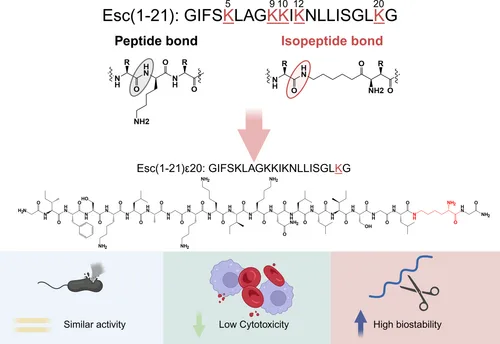
Among the most studied AMPs is Esculentin-1a(1–21)NH₂ (Esc(1-21)), a frog-skin–derived peptide with potent activity against Gram-negative bacteria, notably Pseudomonas aeruginosa. Previous optimization strategies have included incorporation of D-amino acids, introduction of noncanonical residues such as α-aminoisobutyric acid, and targeted single-residue substitutions—each aimed at expanding antimicrobial spectrum, reducing host cytotoxicity, and enhancing proteolytic stability.
The study discussed in this blog adopts a different and innovative approach: site-specific replacement of a conventional peptide bond with an isopeptide bond at selected lysine residues within Esc(1-21). Though chemically subtle, this modification can alter peptide folding, hydrophobicity, and resistance to enzymatic degradation, ultimately reshaping biological activity. By systematically evaluating the impact of isopeptide bond placement, the authors provide new insights into rational AMP design and highlight a pathway toward more stable, selective, and clinically viable antimicrobial agents.
From Concept to Chemistry: Crafting Isopeptide-Modified Esc(1-21)
To investigate the structural and functional consequences of isopeptide bond incorporation, the authors designed five Esc(1-21) analogs in which a single conventional peptide bond was replaced with an isopeptide linkage at one of the peptide’s five lysine residues: K5, K9, K10, K12, or K20. These analogs are designated Esc(1-21)ε5, ε9, ε10, ε12, and ε20, with the subscript denoting the lysine position involved. Isopeptide bond formation was achieved by coupling through the ε-amino group of lysine, rather than the canonical α-amino group—an approach known to enhance proteolytic stability and influence conformational dynamics.

(A) Esc(1-21); (B) Esc(1-21)ε5; (C) Esc(1-21)ε9; (D) Esc(1-21)ε10; (E) Esc(1-21)ε12; (F) Esc(1-21)ε20
All peptides were synthesized using automated microwave-assisted solid-phase peptide synthesis, purified by reverse-phase high-performance liquid chromatography (RP-HPLC), and confirmed for purity (>95%) and identity via time-of-flight mass spectrometry (TOF-MS). The analogs were then subjected to a comprehensive biological and biophysical evaluation to assess antimicrobial potential, safety, stability, and structural properties.
Antimicrobial activity was quantified by determining the minimum inhibitory concentrations (MICs) against a panel of Gram-negative (Escherichia coli, Pseudomonas aeruginosa) and Gram-positive (Staphylococcus epidermidis, Staphylococcus aureus) bacteria. Antibiofilm activity was tested on pre-formed biofilms of P. aeruginosa and S. epidermidis, quantifying reductions in biofilm viability.
To explore mechanism of action, the peptides’ ability to permeabilize bacterial membranes was measured using the Sytox Green uptake assay, while hemolytic potential was assessed by monitoring hemoglobin release from sheep red blood cells. Cytotoxicity toward human keratinocytes (HaCaT) was evaluated using the CCK-8 viability assay after 24 hours of exposure at high peptide concentrations.
Finally, plasma stability was determined by incubating each peptide in human plasma and monitoring degradation over time via RP-HPLC. Structural analysis employed circular dichroism (CD) spectroscopy in aqueous buffer and in the presence of large unilamellar vesicles mimicking bacterial membranes, allowing the authors to assess membrane-induced folding and secondary structure content.
This multi-pronged approach enabled a precise comparison of how isopeptide bond position influences Esc(1-21)’s antimicrobial potency, selectivity, structural integrity, and stability under physiologically relevant conditions.
Decoding the Data: How Bond Position Shapes Function
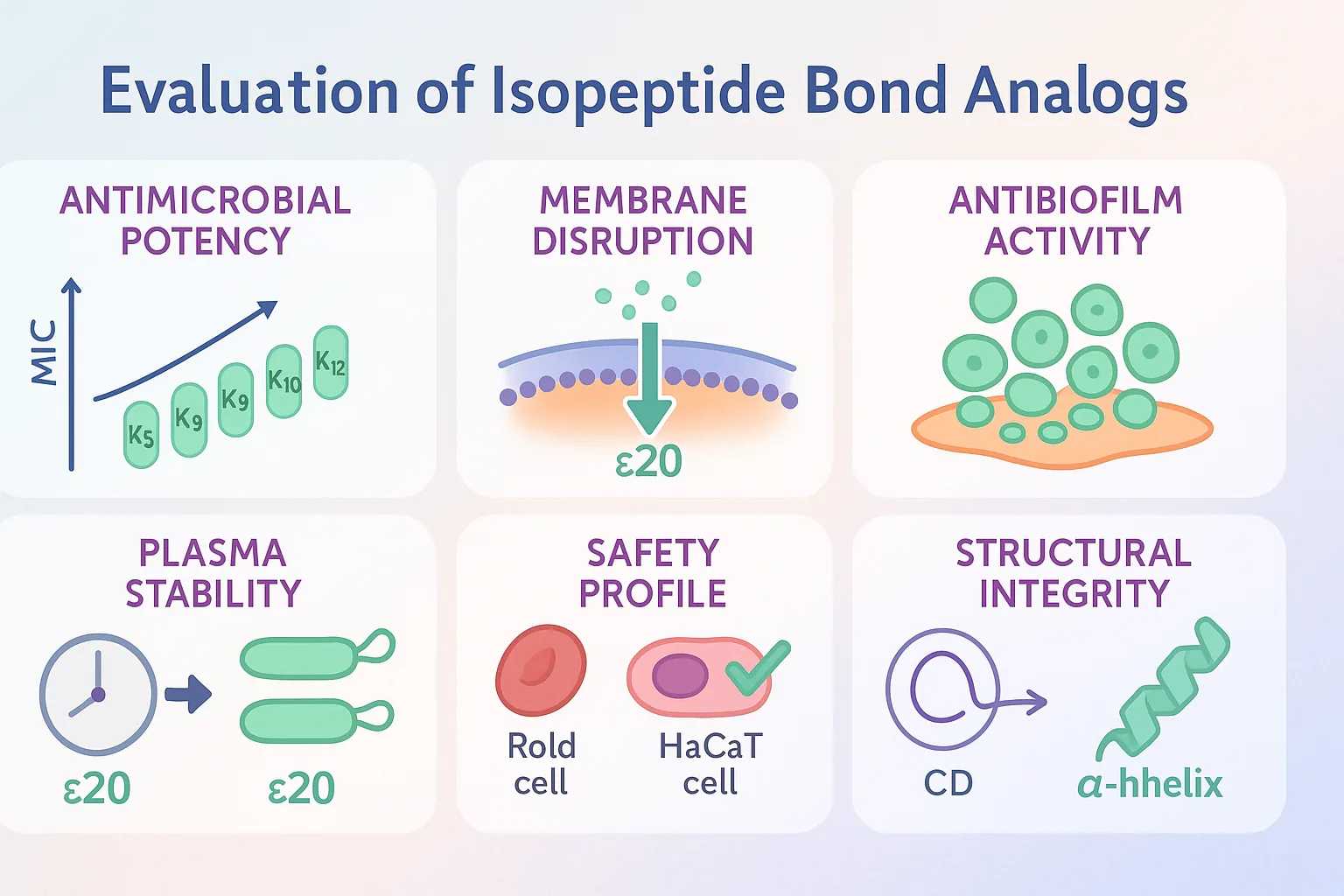
The comparative evaluation of the five isopeptide bond analogs revealed a clear position-dependent effect on the biological activity, structural behavior, and safety of Esc(1-21).
1. Antimicrobial potency varies with substitution site
Minimum inhibitory concentration (MIC) assays showed that analogs with substitutions at K5, K9, and K10 retained antimicrobial activity close to the parent peptide, although with slight increases in MIC values. In contrast, K12 substitution (Esc(1-21)ε12) caused a marked loss of potency across all tested strains, with MICs rising to 25–>50 μM. By comparison, K20 substitution (Esc(1-21)ε20) maintained or slightly improved activity; for example, its MIC against E. coli matched the parent peptide at 1.56 μM and showed enhanced potency against P. aeruginosa strains.
2. Membrane-disruptive ability aligns with antimicrobial results
Sytox Green uptake assays indicated that ε5, ε9, and ε10 produced only modest membrane permeabilization, while ε12 failed to significantly disrupt bacterial membranes. In contrast, ε20 induced rapid, dose-dependent membrane perturbation similar to the wild-type Esc(1-21), confirming that C-terminal substitution preserved its primary mode of action.
3. Antibiofilm activity highlights ε20 as the lead analog
In biofilm eradication assays, ε20 displayed substantial efficacy. Against P. aeruginosa, it reduced biofilm viability by ~60% at 25 μM and ~80% at 50 μM; against S. epidermidis, it achieved >90% reduction across 6.25–50 μM. The ε12 analog, however, remained inactive in all biofilm conditions.
4. Safety profile improved without loss of function in ε20
All analogs exhibited low hemolytic activity (<20% at 50 μM), well below levels typically associated with AMP toxicity. In HaCaT cytotoxicity tests, ε20 reduced cell viability by ~50% at 100 μM but caused significantly less cell death than the parent peptide at 200 μM (~65% vs. ~95% reduction). ε12 was non-toxic at both high concentrations, consistent with its loss of antimicrobial potency.
5. Isopeptide bonds enhance plasma stability
Incubation in human plasma revealed rapid degradation of the parent Esc(1-21), with only ~16% remaining after 24 hours. In contrast, ε20 and ε12 retained ~40% of their initial amount over the same period, indicating that backbone modification substantially improves resistance to proteolytic enzymes.
6. Structural integrity depends on modification site
Circular dichroism analysis showed that in aqueous solution, all peptides were largely unstructured. Upon exposure to membrane-mimicking lipid vesicles, only the parent peptide and ε20 adopted significant α-helical content—an essential structural transition for membrane interaction. Central substitutions (ε9, ε10, ε12) impaired this folding response, correlating with their reduced antimicrobial activity.
Together, these findings demonstrate that C-terminal isopeptide bond incorporation (ε20) uniquely preserves Esc(1-21)’s antimicrobial and antibiofilm properties while enhancing stability and reducing cytotoxicity—qualities that position it as a promising lead for therapeutic development.
Design Lessons: The Structural Logic Behind ε20’s Success
The findings underscore a central principle in antimicrobial peptide engineering: the position of a backbone modification can determine whether function is preserved or lost. Isopeptide bonds, formed through the ε-amino group of lysine, are known to enhance proteolytic stability, but this study shows that their placement also influences folding, hydrophobicity, and membrane interaction.
Substitution at K20, near the C-terminus, appears optimal because it minimally disrupts the α-helical folding required for membrane binding, while subtly increasing hydrophobicity—an attribute that may strengthen bacterial membrane interaction. In contrast, modification at K12, positioned centrally within the sequence, likely interferes with key structural elements, leading to impaired folding, reduced membrane disruption, and loss of antimicrobial and antibiofilm activity.
Compared with prior AMP optimization strategies—such as D-amino acid incorporation, noncanonical residue substitution, or nanoparticle conjugation—this work offers a backbone-level modification strategy that does not require altering side-chain chemistry. This subtle yet impactful change allows ε20 to match or exceed the parent peptide’s potency while delivering tangible benefits: lower cytotoxicity toward eukaryotic cells and markedly improved plasma stability.
Importantly, these results align with earlier reports on isopeptide-modified AMPs, which found that peripheral bond placement can preserve or enhance function, whereas central placement often destabilizes helical structure. By validating this principle in a clinically relevant peptide like Esc(1-21), the study strengthens the case for position-specific backbone engineering as a versatile design tool for balancing potency, selectivity, and stability in next-generation antimicrobial therapeutics.
Beyond the Bench: Pathways Toward Therapeutic Translation
The performance of Esc(1-21)ε20 positions it as a compelling lead for the next generation of antimicrobial therapeutics. Its combination of potent antibacterial and antibiofilm activity, reduced cytotoxicity, and improved stability addresses several of the most common barriers to AMP clinical translation. These features suggest potential applications not only in treating acute infections caused by Gram-negative pathogens but also in tackling chronic, biofilm-associated conditions—such as those found in cystic fibrosis lung infections or infected medical devices.
Future work should include in vivo efficacy studies to validate therapeutic potential, pharmacokinetic profiling to assess distribution and clearance, and formulation strategies that leverage its enhanced stability. Moreover, the demonstrated success of C-terminal isopeptide bond incorporation invites exploration of this modification in other AMP families, potentially yielding a broader platform for rational peptide optimization.
Ultimately, this study exemplifies how precise backbone engineering can fine-tune the delicate balance between antimicrobial potency, host safety, and stability—offering a blueprint for designing more effective and clinically viable peptide-based antibiotics in the fight against resistant pathogens.
Reference
Casciaro, B., Hur, D. B., Roversi, D., Vetrano, C., Kiper, E., Cappella, G., … & Mangoni, M. L. (2025). Modulating Antimicrobial Activity and Structure of the Peptide Esc (1‐21) via Site‐Specific Isopeptide Bond Formation. Journal of Peptide Science, 31(9), e70048.
https://doi.org/10.1002/psc.70048
Brüssow, H. (2024). The antibiotic resistance crisis and the development of new antibiotics. Microbial Biotechnology, 17(7), e14510.
https://doi.org/10.1111/1751-7915.14510
Avci, F. G., Sariyar Akbulut, B., & Ozkirimli, E. (2018). Membrane active peptides and their biophysical characterization. Biomolecules, 8(3), 77.
https://doi.org/10.3390/biom8030077
Luca, V., Stringaro, A., Colone, M., Pini, A., & Mangoni, M. L. (2013). Esculentin (1-21), an amphibian skin membrane-active peptide with potent activity on both planktonic and biofilm cells of the bacterial pathogen Pseudomonas aeruginosa. Cellular and molecular life sciences, 70(15), 2773-2786.
 LinkPeptide
LinkPeptide