Stronger by Design: Peptide Nanorods Transform GelMA Hydrogels for Regenerative Medicine
Abstract
Hydrogels such as gelatin methacryloyl (GelMA) hold great promise for regenerative medicine, offering biocompatibility, photocurability, and adaptability for in situ repair and 3D bioprinting. Yet, their inherent brittleness limits performance in load-bearing applications like cartilage and bone regeneration. Inspired by the mechanical resilience of intervertebral discs, researchers have developed a reinforcement strategy using peptide-based rigid nanorods (PRNs). Covalently integrated into the GelMA network, PRNs act as nanoscale beams that dissipate energy, prevent crack propagation, and markedly enhance strength, toughness, and fatigue resistance—without compromising cell compatibility. This innovation enables GelMA to support osteochondral regeneration and complex additive manufacturing, expanding its utility for personalized implants. By combining molecular-level design with bioinspired mechanics, PRN-reinforced GelMA represents a new direction for multifunctional biomaterials, bridging the gap between biological performance and mechanical durability.
From Promise to Problem: Why Hydrogels Struggle Under Pressure
Hydrogels are among the most promising materials in regenerative medicine, celebrated for their soft, water-rich networks that closely resemble the body’s own extracellular matrix. They can host living cells, deliver therapeutic molecules, and adapt to a wide range of biomedical applications—from wound dressings to engineered tissue scaffolds. Among these, gelatin methacryloyl (GelMA) has earned particular prominence. Derived from natural gelatin, it inherits intrinsic biocompatibility and biodegradability, while its methacryloyl groups enable rapid, light-triggered crosslinking. This unique combination makes GelMA a top choice for in situ tissue repair and the 3D printing of patient-specific implants.
However, in load-bearing environments such as cartilage repair or subchondral bone support, GelMA’s strengths are undermined by a critical weakness: brittleness and low mechanical resilience. Under sustained or repeated stress, its network can fracture, deform permanently, or lose the structural integrity essential for functional regeneration. Conventional toughening strategies—such as incorporating inorganic nanoparticles, reinforcing fibers, or creating double-network architectures—can improve strength, but they often come at a cost. These methods may introduce cytotoxicity, slow degradation, require harsh processing, or limit the precision of fabrication.
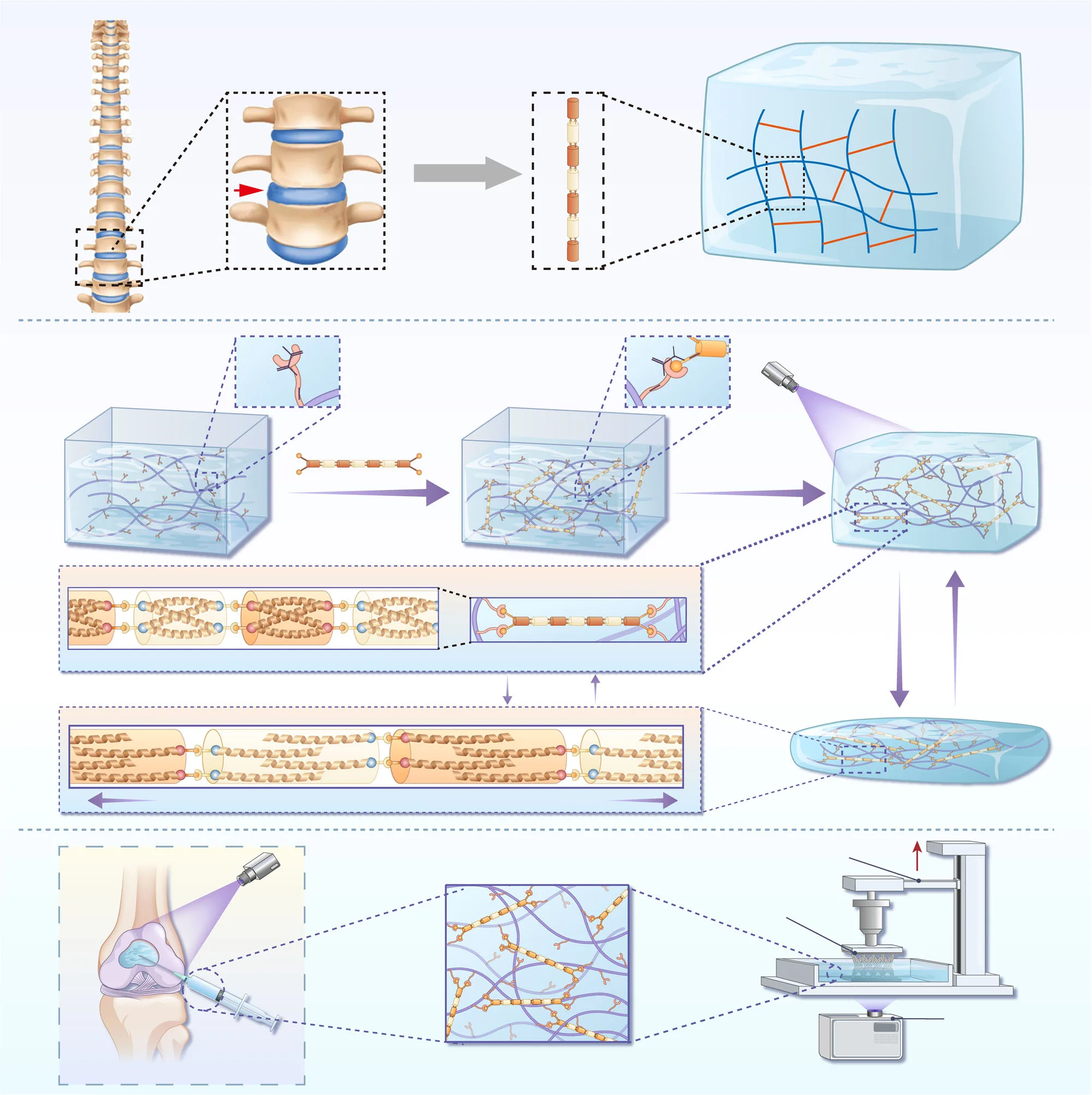
A recent study presents a bioinspired solution, taking cues from the intervertebral disc—a natural structure capable of withstanding immense mechanical loads while maintaining flexibility. The researchers developed peptide-based rigid nanorods (PRNs) that integrate seamlessly into the GelMA network, acting like nanoscale beams that reinforce the hydrogel without compromising its biological advantages. This approach could mark a pivotal advance in creating hydrogels that are not only friendly to cells but also tough enough for the body’s most demanding environments.
Peptide Nanorods: Building Strength at the Molecular Level
At the core of this innovation are peptide-based rigid nanorods (PRNs)—nanoscale, rod-shaped structures assembled from short peptides that form tightly packed, antiparallel bundles. Unlike the flexible polymer chains in typical hydrogels, PRNs possess exceptional rigidity, enabling them to act as miniature structural beams within the network. Each nanorod is capped with thiol groups at both ends, allowing it to covalently bond to the methacryloyl groups in GelMA via thiol–ene chemistry. This means that once the hydrogel is photocrosslinked, the PRNs become an integral, load-bearing part of the matrix rather than free-floating fillers.
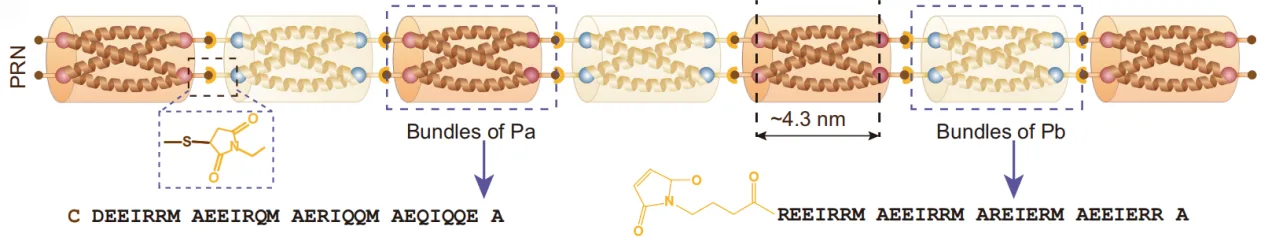
This chemical integration is a crucial departure from traditional reinforcement methods. Many earlier strategies relied on physically mixing nanoparticles, nanofibers, or mineral platelets into hydrogels to increase stiffness. While such additives can improve strength, they often disperse unevenly, clump during processing, and create local weak spots that compromise mechanical reliability. In contrast, covalently anchored PRNs distribute uniformly throughout the GelMA network, providing reinforcement at the molecular level and ensuring consistent performance throughout the material.
The mechanical concept draws directly from the inspiration of the intervertebral disc. Just as discs absorb shocks and redistribute forces between vertebrae, PRNs in GelMA help absorb and dissipate mechanical energy. When the hydrogel is compressed, the PRNs elongate slightly and release energy through the breaking of reversible, non-covalent interactions within their peptide bundles. This sacrificial bond mechanism prevents catastrophic network failure, delays crack propagation, and allows the hydrogel to withstand higher strains without losing integrity.
By uniting molecular-level architecture with bioinspired mechanics, the PRN-based strategy transforms GelMA from a material best suited for soft, low-load applications into one that can realistically support load-bearing tissue engineering and high-precision additive manufacturing.
Stronger, Tougher, and Still Cell-Friendly
The introduction of PRNs into GelMA results in a striking transformation in mechanical behavior. Without delving into exhaustive data, the trend is clear: PRN-reinforced hydrogels are markedly stronger, tougher, and more fatigue-resistant than their unmodified counterparts. They tolerate higher strains before failure, recover shape more reliably after deformation, and resist cracking under repeated compression.
This improvement stems from energy dissipation at the nanoscale. In standard GelMA, stress travels directly along polymer chains, which can snap once stretched beyond their limit. In PRN-supported GelMA, part of the load is diverted into the nanorods. As these PRNs elongate under stress, they absorb energy through the reversible breaking of non-covalent interactions within their peptide bundles—mechanical “shock absorbers” that delay failure of the main network.
Equally important, this toughening does not come at the cost of biological performance. Bone marrow-derived mesenchymal stem cells (BMSCs) cultured with NSG hydrogels adhere well, maintain healthy morphology, and show high viability over time. PRNs themselves exhibit no cytotoxicity and can even support enhanced cell proliferation under certain conditions.
In animal implantation studies, NSG hydrogels also displayed a favorable immune profile. Markers of pro-inflammatory activity were reduced, while anti-inflammatory markers associated with tissue repair increased. This suggests that beyond mechanical benefits, PRN integration may help create a local environment more conducive to healing.
By pairing mechanical resilience with cell-friendly, regenerative potential, PRN-reinforced GelMA advances the field toward hydrogels that can withstand the rigors of load-bearing use without sacrificing the biological advantages that make them so valuable in tissue engineering.
Healing Cartilage, Printing the Future
One of the most compelling targets for PRN-reinforced GelMA is osteochondral repair—the restoration of damaged cartilage and the underlying bone. These junctions endure constant, high-pressure loading, making them a challenging site for conventional hydrogels. In preclinical models, nanorod-supported GelMA (NSG) produced more complete and integrated tissue regeneration than unmodified GelMA. Newly formed cartilage in NSG-treated defects displayed smooth surfaces, robust structure, and biochemical hallmarks such as collagen type II, a key component of healthy articular cartilage. Subchondral bone also showed improved regeneration, with denser, better-organized trabecular architecture.
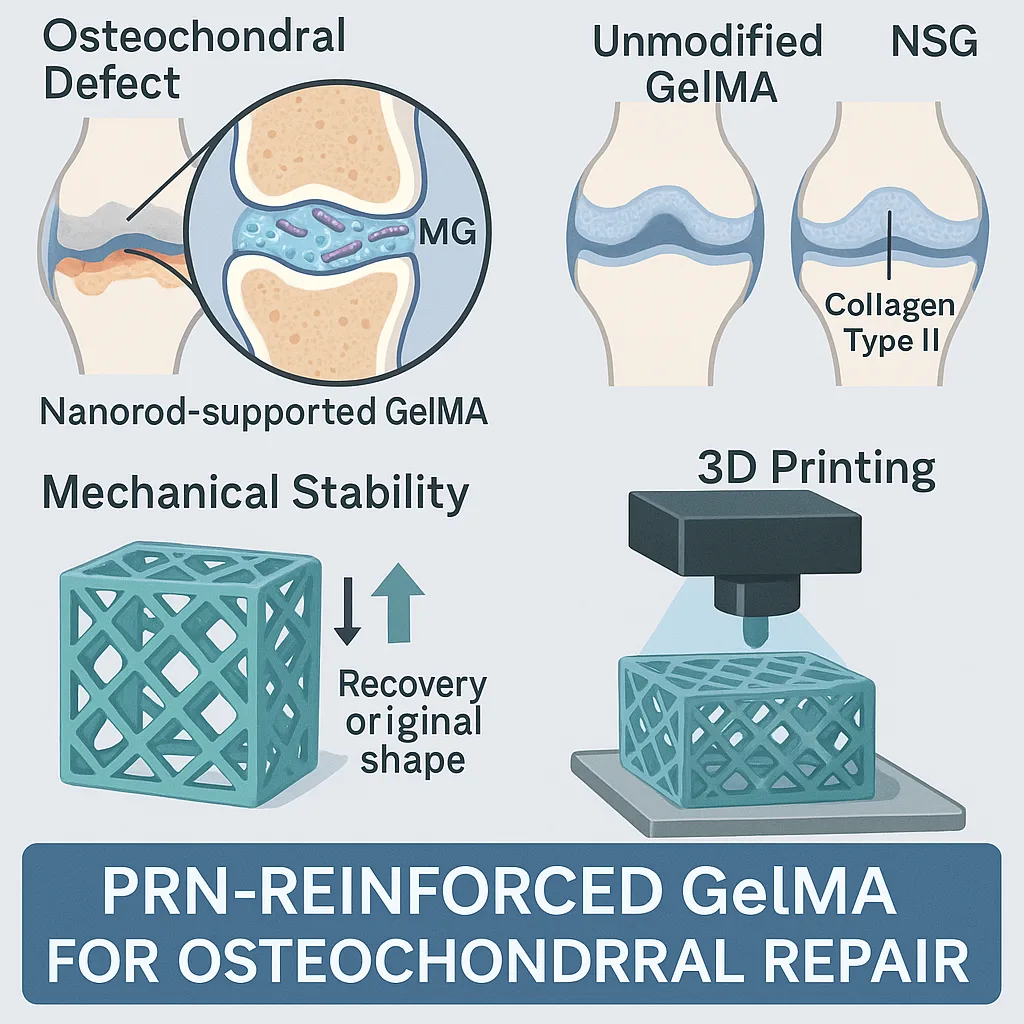
The likely reason is mechanical compatibility. A scaffold that can bear physiological loads without collapsing provides a more stable platform for cell infiltration, extracellular matrix deposition, and long-term tissue maturation. At the same time, the immunomodulatory effects of PRNs may further support a regenerative environment.
Beyond direct repair, PRN reinforcement preserves one of GelMA’s most valuable features—photo-responsiveness—which is central to additive manufacturing applications. Using techniques such as digital light processing (DLP), NSG hydrogels can be printed into complex, patient-specific geometries that remain stable under mechanical stress. In demonstrations, intricate lattice and multi-channel structures withstood more than 50% deformation without structural failure, recovering their original form after release.
This resilience dramatically broadens the design space for personalized implants and biofabricated scaffolds, allowing the creation of shapes optimized for specific anatomical sites and mechanical demands. By bridging the gap between custom fabrication and mechanical reliability, PRN-reinforced GelMA stands out as a versatile platform for both clinical translation and advanced biomedical manufacturing.
Beyond GelMA: Expanding the Horizons of PRN Technology
The integration of peptide-based rigid nanorods into GelMA offers more than just an incremental improvement—it represents a design shift in how hydrogels can be reinforced for demanding biomedical roles. By embedding structural strength at the molecular level while preserving cell-friendly features, this approach points toward a new class of multifunctional, bioinspired materials.
Looking ahead, several challenges will shape its path to clinical use. Long-term studies are needed to confirm stability and controlled biodegradation under varied physiological conditions. Scaling PRN synthesis in a cost-effective, reproducible way will be critical for moving beyond laboratory prototypes. Additionally, regulatory considerations will require comprehensive safety and performance data.
The adaptability of the PRN strategy is especially promising. Its chemistry can be extended to other photocurable hydrogels, opening possibilities in drug delivery, wound healing, organoid culture, and even soft robotics. With its balance of mechanical resilience, biological compatibility, and manufacturing flexibility, PRN-reinforced GelMA could become a cornerstone material for next-generation regenerative therapies—one that moves the field closer to hydrogels capable of performing like the tissues they are meant to replace.
Reference
Zhu, J., Wei, Y., Yang, G., Zhang, J., Liu, J., Zhang, X., … & Wang, J. (2025). Peptide-based rigid nanorod-reinforced gelatin methacryloyl hydrogels for osteochondral regeneration and additive manufacturing. Nature Communications, 16(1), 7090.
https://doi.org/10.1038/s41467-025-62540-0
Fang, Z., Mu, H., Sun, Z., Zhang, K., Zhang, A., Chen, J., … & Xie, T. (2024). 3D printable elastomers with exceptional strength and toughness. Nature, 631(8022), 783-788.
https://doi.org/10.1038/s41586-024-07588-6
Kim, J., Zhang, G., Shi, M., & Suo, Z. (2021). Fracture, fatigue, and friction of polymers in which entanglements greatly outnumber cross-links. Science, 374(6564), 212-216.
https://doi.org/10.1126/science.abg6320
Abramson, J., Adler, J., Dunger, J., Evans, R., Green, T., Pritzel, A., … & Jumper, J. M. (2024). Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature, 630(8016), 493-500.
 LinkPeptide
LinkPeptide