Is Semaglutide the Master Key for MASH? Mechanistic Insights and Clinical Evidence
Abstract
Metabolic dysfunction–associated steatohepatitis (MASH) represents the progressive and clinically significant form of metabolic dysfunction–associated steatotic liver disease (MASLD), a condition now recognized as the most common cause of chronic liver disease worldwide. Closely linked to obesity, type 2 diabetes mellitus, and cardiovascular morbidity, MASH carries a substantial risk of cirrhosis, hepatocellular carcinoma, and mortality, yet effective therapies remain limited. Semaglutide, a glucagon-like peptide-1 receptor agonist (GLP-1 RA), originally developed for diabetes and obesity management, has demonstrated significant benefits in weight reduction, glycemic control, and cardiovascular outcomes. Emerging evidence suggests that semaglutide also improves hepatic steatosis, insulin resistance, and inflammation in MASH, though its effects on fibrosis remain modest. This review synthesizes mechanistic insights, preclinical findings, and recent clinical trial data, including Phase III results, to evaluate semaglutide’s role in MASH management. While not the ultimate solution, semaglutide may represent a cornerstone in future combination therapies targeting this complex disease.
Introduction: The Rising Burden of MASH
Metabolic dysfunction–associated steatohepatitis (MASH), the progressive inflammatory form of metabolic dysfunction–associated steatotic liver disease (MASLD), has emerged as a critical global health challenge. Currently affecting hundreds of millions of people worldwide, MASH is now recognized as the most common cause of chronic liver disease and is projected to become the leading indication for liver transplantation within the next decade. The disease represents the transition from simple steatosis to a more severe state marked by hepatocellular injury, chronic inflammation, and varying degrees of fibrosis—pathological changes that can culminate in cirrhosis and hepatocellular carcinoma (HCC).
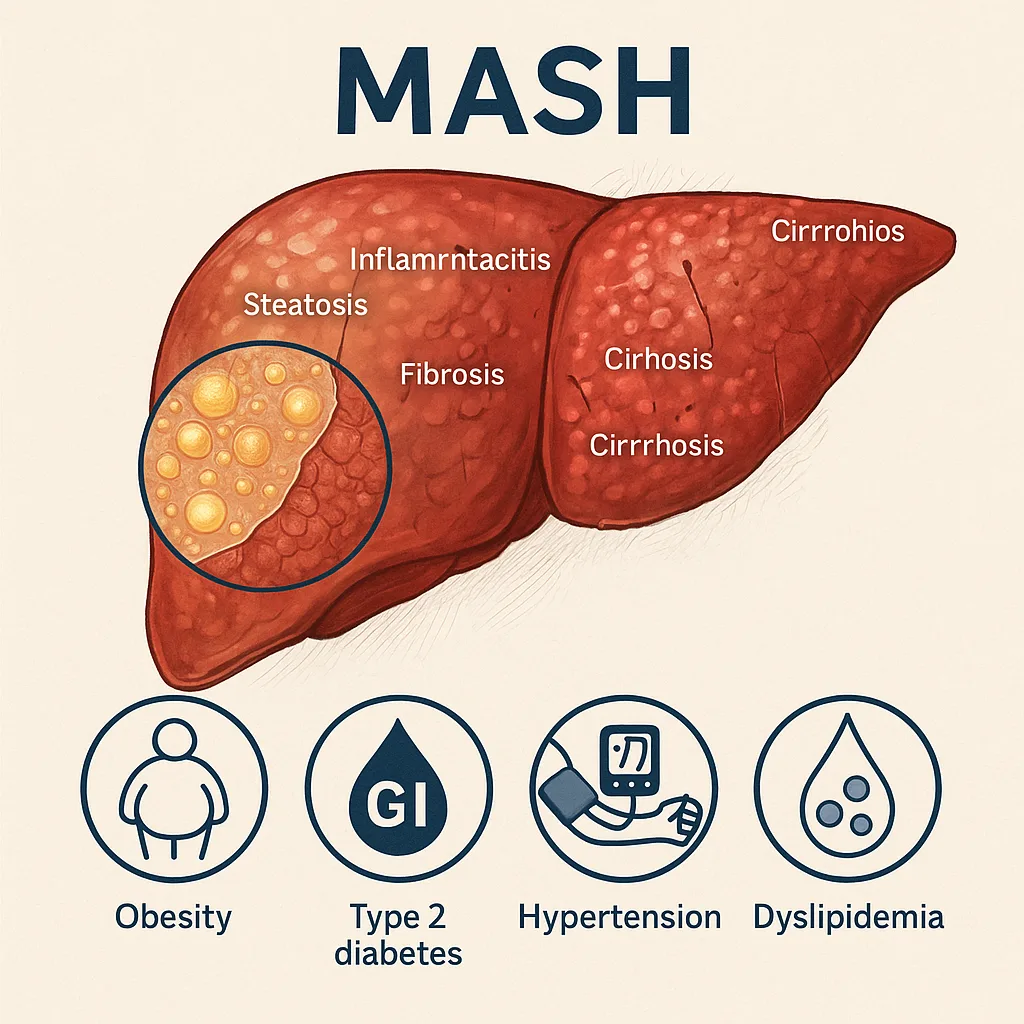
MASH rarely develops in isolation. Instead, it is strongly associated with obesity, type 2 diabetes mellitus (T2DM), insulin resistance, hypertension, and dyslipidemia—conditions that collectively amplify disease severity and accelerate progression. Epidemiological studies illustrate this overlap: nearly two-thirds of individuals with T2DM also present with MASLD or MASH, and when these conditions coexist, the risks of cardiovascular disease, chronic kidney disease, and malignancy rise sharply. Importantly, cardiovascular complications remain the predominant cause of mortality in patients with MASH, underscoring the systemic nature of this disease.
Despite its rising prevalence and serious clinical implications, effective therapies for MASH remain scarce. Lifestyle modification through dietary intervention, weight reduction, and increased physical activity is still the cornerstone of treatment, yet long-term adherence is difficult to achieve. Bariatric surgery offers benefits in selected patients but is not broadly applicable. The approval of resmetirom, a thyroid hormone receptor-β agonist, marks a milestone as the first targeted pharmacological treatment for MASH, but its arrival also highlights the need for additional agents capable of addressing the disease’s complex metabolic and inflammatory underpinnings.
Against this backdrop, semaglutide, a glucagon-like peptide-1 receptor agonist (GLP-1 RA), has generated considerable interest. Initially developed for the management of T2DM and obesity, semaglutide has demonstrated profound benefits in weight reduction, glycemic control, cardiovascular protection, and systemic inflammation. These effects align closely with the pathophysiological drivers of MASH, raising an important question: Could semaglutide represent a transformative therapy for MASH and serve as the long-sought “master key” to halting disease progression?
Semaglutide Beyond Diabetes: Expanding Therapeutic Horizons
Semaglutide is a glucagon-like peptide-1 receptor agonist (GLP-1 RA), a class of incretin-based therapies that has reshaped the management of type 2 diabetes mellitus (T2DM) and obesity. By mimicking the action of endogenous GLP-1, semaglutide stimulates insulin secretion in a glucose-dependent manner, suppresses glucagon release, delays gastric emptying, and reduces appetite. Collectively, these actions lead to improved glycemic control, significant weight reduction, and favorable cardiometabolic outcomes, making semaglutide one of the most effective GLP-1 RAs developed to date.
From a regulatory standpoint, semaglutide is available in three proprietary formulations approved by the U.S. Food and Drug Administration (FDA):
- Ozempic® – a once-weekly injectable indicated for T2DM, also shown to reduce cardiovascular risk.
- Wegovy® – a higher-dose injectable approved specifically for long-term weight management in individuals with obesity or overweight.
- Rybelsus® – the first oral GLP-1 RA, providing a daily alternative for glycemic management.
Large clinical trial programs have established semaglutide’s broad therapeutic impact. The SUSTAIN trials demonstrated robust reductions in glycated hemoglobin (HbA1c) and body weight, while the landmark SUSTAIN-6 study confirmed cardiovascular risk reduction in high-risk T2DM patients. Similarly, the PIONEER trials established the efficacy and safety of oral semaglutide across diverse populations, and the FLOW trial demonstrated renal protection in patients with T2DM and kidney disease. These findings highlight semaglutide’s role as a multisystem therapy extending well beyond glucose regulation.
The relevance of these outcomes for MASH is clear. The disease is tightly linked with insulin resistance, obesity, and cardiovascular complications—domains in which semaglutide has already proven effective. Moreover, emerging evidence points to its anti-inflammatory properties and favorable modulation of lipid metabolism, both of which may provide indirect hepatoprotective effects. Thus, semaglutide is uniquely positioned as a candidate capable of addressing the metabolic, inflammatory, and cardiovascular dimensions of MASH.
Mechanistic Pathways: How Semaglutide Shapes MASH Biology
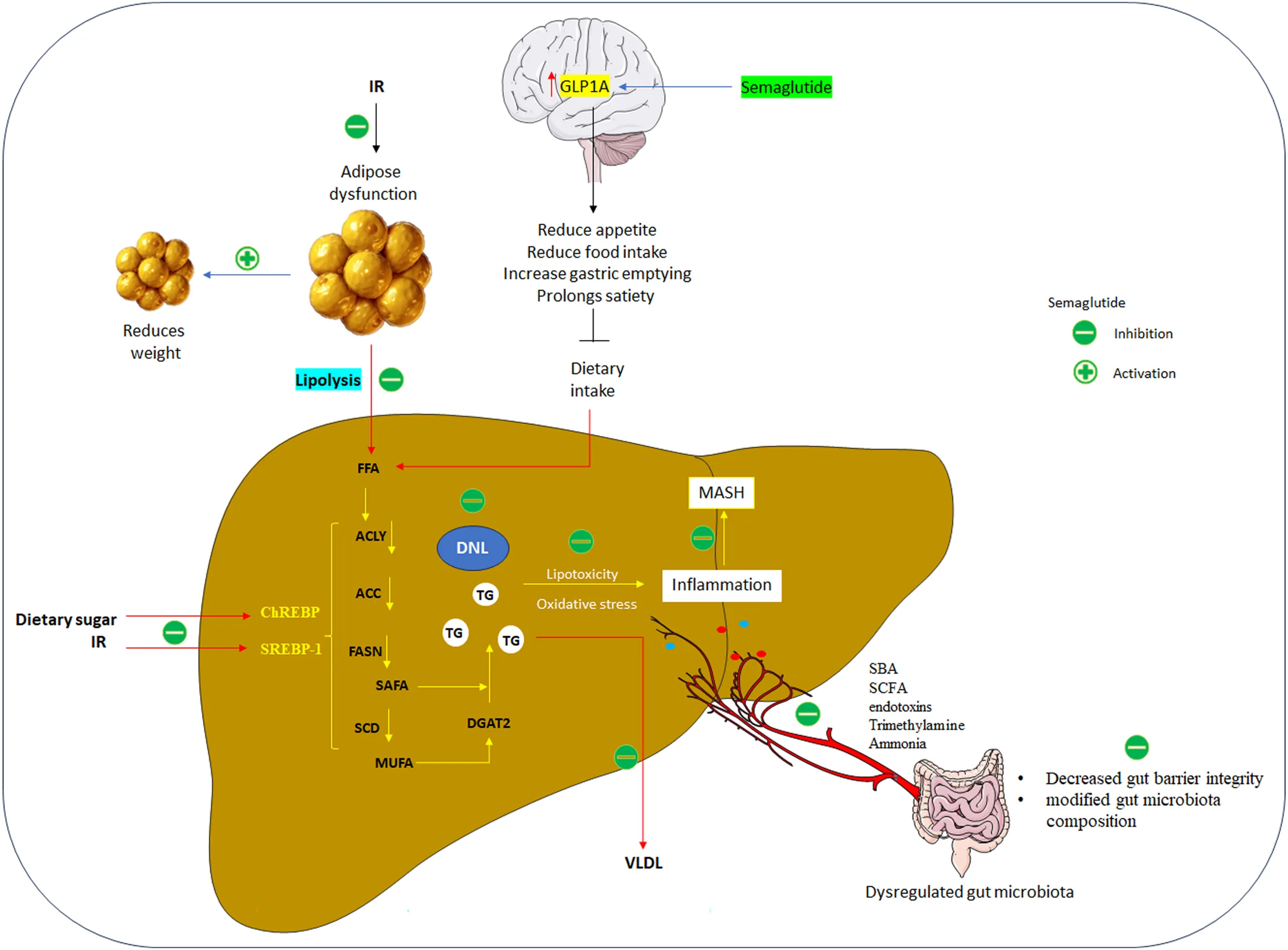
The therapeutic potential of semaglutide in metabolic dysfunction–associated steatohepatitis (MASH) lies in its ability to influence multiple metabolic and inflammatory pathways. Importantly, both human and murine studies confirm that GLP-1 receptors are not expressed in hepatocytes, suggesting that semaglutide does not act directly on the liver. Instead, its benefits are mediated through indirect systemic effects, which converge to reduce hepatic steatosis, inflammation, and associated injury.
3.1 Appetite Suppression and Free Fatty Acid Flux
Semaglutide activates GLP-1 receptors in the brain, suppressing appetite and prolonging satiety. This leads to reduced caloric intake and diminished delivery of dietary free fatty acids (FFAs) to the liver. Given that dietary fat contributes significantly to hepatic triglyceride accumulation, this reduction plays a critical role in alleviating steatosis.
3.2 Insulin Sensitivity and Adipose Tissue Regulation
By improving systemic insulin sensitivity, semaglutide curtails inappropriate lipolysis in adipose tissue, thereby lowering circulating FFA levels and reducing hepatic lipid burden. Additionally, semaglutide reduces the mass of both white and brown adipose tissue, further limiting the release of lipotoxic substrates that drive hepatocellular injury.
3.3 Inhibition of De Novo Lipogenesis (DNL)
Semaglutide also interferes with de novo lipogenesis, a major contributor to MASH pathogenesis. It downregulates transcription factors ChREBP and SREBP-1c, along with lipogenic enzymes such as acetyl-CoA carboxylase (ACC), fatty acid synthase (FAS), and stearoyl-CoA desaturase-1 (SCD-1). This suppression of DNL reduces hepatic triglyceride synthesis and lipid droplet accumulation, thereby mitigating hepatocellular ballooning and steatosis.
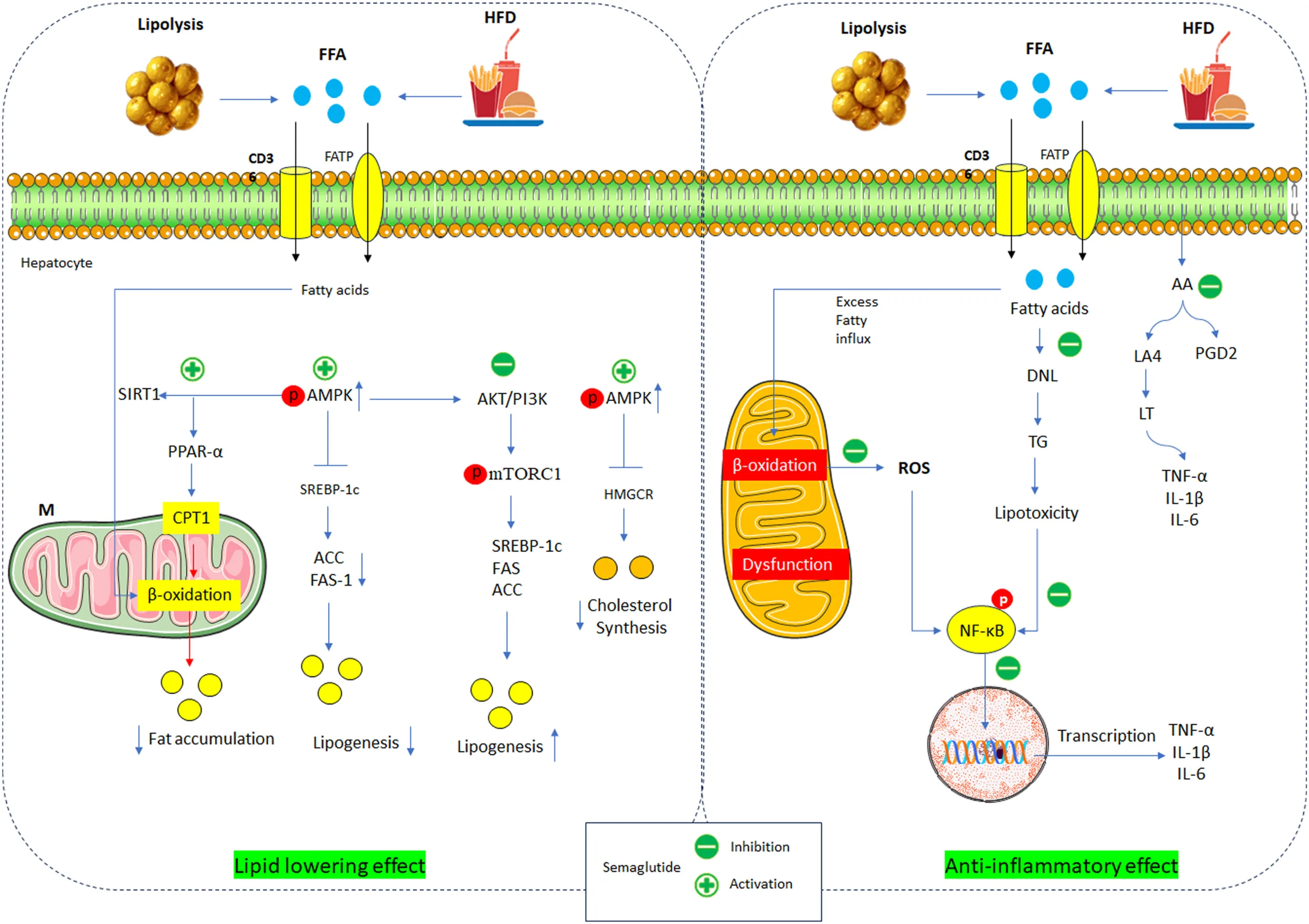
3.4 Anti-Inflammatory and Antioxidant Effects
MASH is characterized by persistent hepatic inflammation and oxidative stress. Semaglutide reduces the expression of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β) and lipid mediators such as prostaglandins and leukotrienes. It also decreases hepatic galectin-3, a protein implicated in both inflammation and fibrosis. At the same time, semaglutide enhances antioxidant defenses, lowering reactive oxygen species (ROS) and endoplasmic reticulum stress, thereby protecting hepatocytes from ongoing injury.
3.5 Modulation of the Gut–Liver Axis
Emerging evidence highlights semaglutide’s ability to alter the gut microbiome, improving gut barrier integrity and reshaping microbial composition. In experimental models, semaglutide increased beneficial genera such as Lactobacillus and Alloprevotella, while reducing dysbiosis-associated species. These changes improve insulin sensitivity and reduce hepatic inflammation, underscoring the importance of the gut–liver axis in MASH therapy.
3.6 Integration of Mechanisms
Taken together, semaglutide acts on multiple interconnected systems—reducing caloric intake, lowering FFA flux, improving insulin sensitivity, suppressing DNL, attenuating inflammation, and modulating the gut microbiota. Although indirect, these systemic effects converge on the liver to promote MASH resolution, explaining the consistent improvements in steatosis and inflammation observed in clinical studies.
Translational Evidence: From Bench to Bedside
The mechanistic rationale for semaglutide in metabolic dysfunction–associated steatohepatitis (MASH) has been tested extensively in both preclinical models and clinical trials. Collectively, the evidence demonstrates that semaglutide improves steatosis, inflammation, and metabolic parameters, but its effect on fibrosis remains limited.
4.1 Preclinical Evidence
In vitro studies using hepatocyte–macrophage co-cultures showed that semaglutide reduces apoptosis, alleviates endoplasmic reticulum stress, and decreases lipid accumulation. In multiple murine models of diet- or obesity-induced MASH, semaglutide consistently improved NAFLD activity scores, reduced triglyceride accumulation, and downregulated inflammatory markers such as TNF-α and IL-6. Importantly, semaglutide also improved insulin signaling through AMPK and PPAR-α activation.
However, its effects on fibrosis were modest. While semaglutide decreased markers of hepatic stellate cell activation (e.g., α-SMA, collagen), it did not reliably reverse established fibrosis. Combination therapies have shown promise: pairing semaglutide with lysophosphatidic acid receptor antagonists or fibroblast growth factor 21 analogues yielded greater reductions in fibrosis than semaglutide alone.
4.2 Clinical Evidence
Human studies mirror preclinical findings. In patients with T2DM and MASLD, oral semaglutide (up to 14 mg daily) improved glycemic control, liver enzymes, insulin resistance, and hepatic steatosis, alongside weight loss. Injectable semaglutide (0.4–2.4 mg weekly) produced dose-dependent improvements in steatosis, transaminases, and inflammatory markers, with benefits closely tied to the degree of weight reduction.
A pivotal Phase II trial demonstrated that daily subcutaneous semaglutide (0.1–0.4 mg for 72 weeks) achieved MASH resolution in a significant proportion of patients, but without parallel improvement in fibrosis stage. More recently, interim results from the Phase III ESSENCE trial (2.4 mg weekly for 72 weeks) showed that:
- 62.9% achieved MASH resolution without worsening fibrosis.
- 36.8% achieved fibrosis improvement without worsening steatohepatitis.
- 32.7% achieved both endpoints.
These results are encouraging, though they confirm that fibrosis remains the “missing master key.” Ongoing long-term ESSENCE data (240 weeks) will be critical in determining whether semaglutide can alter the natural history of MASH-related fibrosis.
The Road Ahead: Challenges, Combinations, and Clinical Outlook
While semaglutide has shown remarkable promise in resolving steatohepatitis, several limitations and challenges remain. Most importantly, its antifibrotic efficacy is modest. Both preclinical and clinical studies consistently demonstrate improvements in steatosis, inflammation, and metabolic parameters, yet fibrosis—arguably the strongest predictor of long-term outcomes—remains largely unchanged. This limitation suggests that semaglutide alone may be insufficient for patients with advanced disease.
Another concern is patient safety in special populations. Rapid weight loss induced by semaglutide may exacerbate complications in individuals with cirrhosis, as illustrated by reports of ascites and hepatic decompensation following aggressive weight reduction. Additionally, some studies highlight the potential risk of sarcopenia, particularly in elderly or diabetic patients, as semaglutide can reduce lean body mass alongside fat loss. These issues underscore the need for careful patient selection, monitoring, and possibly dose adjustments in high-risk groups.
Looking forward, combination therapy represents the most promising avenue. Trials pairing semaglutide with agents such as cilofexor, firsocostat, and fibroblast growth factor 21 analogues have already shown synergistic improvements in hepatic and metabolic biomarkers. Such strategies may ultimately provide the comprehensive approach needed to target both inflammation and fibrosis in MASH.
In conclusion, semaglutide stands as one of the most compelling pharmacological candidates for MASH resolution, offering profound benefits in weight reduction, glycemic control, and hepatic inflammation. However, fibrosis remains the “missing master key.” Future success will likely depend on multi-drug regimens that integrate semaglutide’s systemic benefits with direct antifibrotic therapies, thereby addressing the full spectrum of MASH pathogenesis.
Reference
Ezhilarasan, D. (2025). Mechanism of Semaglutide in MASLD Treatment: Where Is the Master Key?. Journal of Gastroenterology and Hepatology.
https://doi.org/10.1111/jgh.17037
Alkhouri, N., Charlton, M., Gray, M., & Noureddin, M. (2025). The pleiotropic effects of glucagon-like peptide-1 receptor agonists in patients with metabolic dysfunction-associated steatohepatitis: a review for gastroenterologists. Expert Opinion on Investigational Drugs, 34(3), 169-195.
https://doi.org/10.1080/13543784.2025.2473062
Marso, S. P., Bain, S. C., Consoli, A., Eliaschewitz, F. G., Jódar, E., Leiter, L. A., … & Vilsbøll, T. (2016). Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. New England Journal of Medicine, 375(19), 1834-1844.
https://doi.org/10.1056/NEJMoa1607141
Friedman, S. L., Neuschwander-Tetri, B. A., Rinella, M., & Sanyal, A. J. (2018). Mechanisms of NAFLD development and therapeutic strategies. Nature medicine, 24(7), 908-922.
 LinkPeptide
LinkPeptide