When Environment Becomes the Architect: Context Dependence in Peptide Self-Assembly
Abstract
Peptide self-assembly has traditionally been understood through the framework of an “assembly code,” in which the primary sequence is thought to dictate supramolecular architecture. While this concept has enabled rational design of predictable nanostructures, growing evidence demonstrates that peptide assembly is profoundly context-dependent. Environmental variables—including pH, ionic strength, solvent conditions, and molecular crowding—reshape assembly pathways, leading to diverse and sometimes unexpected morphologies. Rather than converging on a single structural state, peptide assemblies navigate dynamic energy landscapes, producing multistable and adaptive outcomes. This shift from deterministic to context-aware perspectives transforms peptides from static building blocks into versatile platforms for engineering responsive materials. Harnessing context dependence opens new opportunities in nanotechnology, regenerative medicine, and drug delivery, while also presenting challenges of reproducibility and predictability. Future progress will depend on integrating experimental, computational, and interdisciplinary approaches to master the contextual interpretation of peptide assemblies.
From Sequence to System: Rethinking Peptide Assembly
Peptides, owing to their modular structures and chemical versatility, have become indispensable building blocks in the design of supramolecular systems. Through a rich repertoire of non-covalent interactions—including hydrogen bonding, π–π stacking, electrostatics, and hydrophobic effects—short peptide sequences can spontaneously organize into ordered nanostructures such as fibrils, sheets, vesicles, and hydrogels. This intrinsic ability to self-assemble forms the foundation for a wide spectrum of applications in nanotechnology, biomedicine, and advanced materials science, ranging from regenerative scaffolds to intelligent drug delivery platforms.
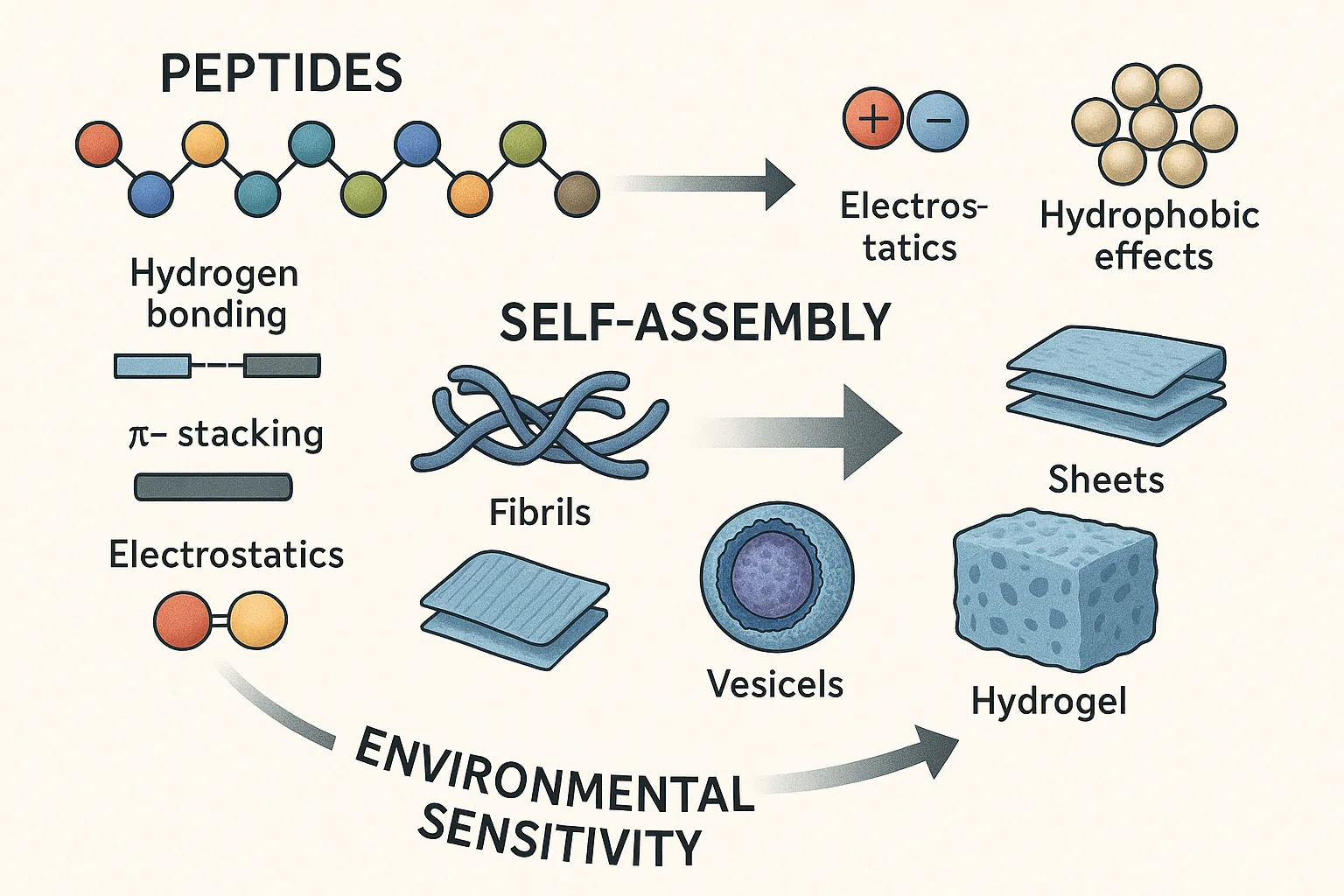
For many years, the concept of an “assembly code” has guided peptide design. According to this view, the primary sequence alone encodes sufficient information to predetermine supramolecular architecture, much like the genetic code governs protein folding. This analogy has offered a powerful design framework, enabling the rational construction of predictable structural motifs by embedding assembly-promoting residues into peptide backbones.
However, emerging evidence reveals that this deterministic model is incomplete. The same peptide sequence can yield strikingly different morphologies depending on its context—whether defined by pH, ionic composition, solvent environment, co-assembly partners, or molecular crowding. Such observations highlight a critical paradigm shift: peptide assembly must be understood not as a static consequence of sequence, but as a dynamic and context-dependent process.
This blog will examine how environmental and molecular variables reinterpret the assembly code, and how this contextual sensitivity opens new avenues for engineering adaptive, functional, and responsive peptide-based materials.
The Assembly Code: Promise and Limitations
The notion of an “assembly code” in peptide science is rooted in the idea that sequence dictates structure. Just as specific amino acid patterns direct the folding of proteins, short peptides often contain minimal motifs that predispose them toward particular supramolecular architectures. For instance, β-sheet–forming sequences frequently yield extended fibrils stabilized by hydrogen bonding, while amphiphilic arrangements promote segregation into bilayers, micelles, or vesicular assemblies. These predictable outcomes have enabled researchers to employ sequence design as a blueprint for generating functional nanomaterials.
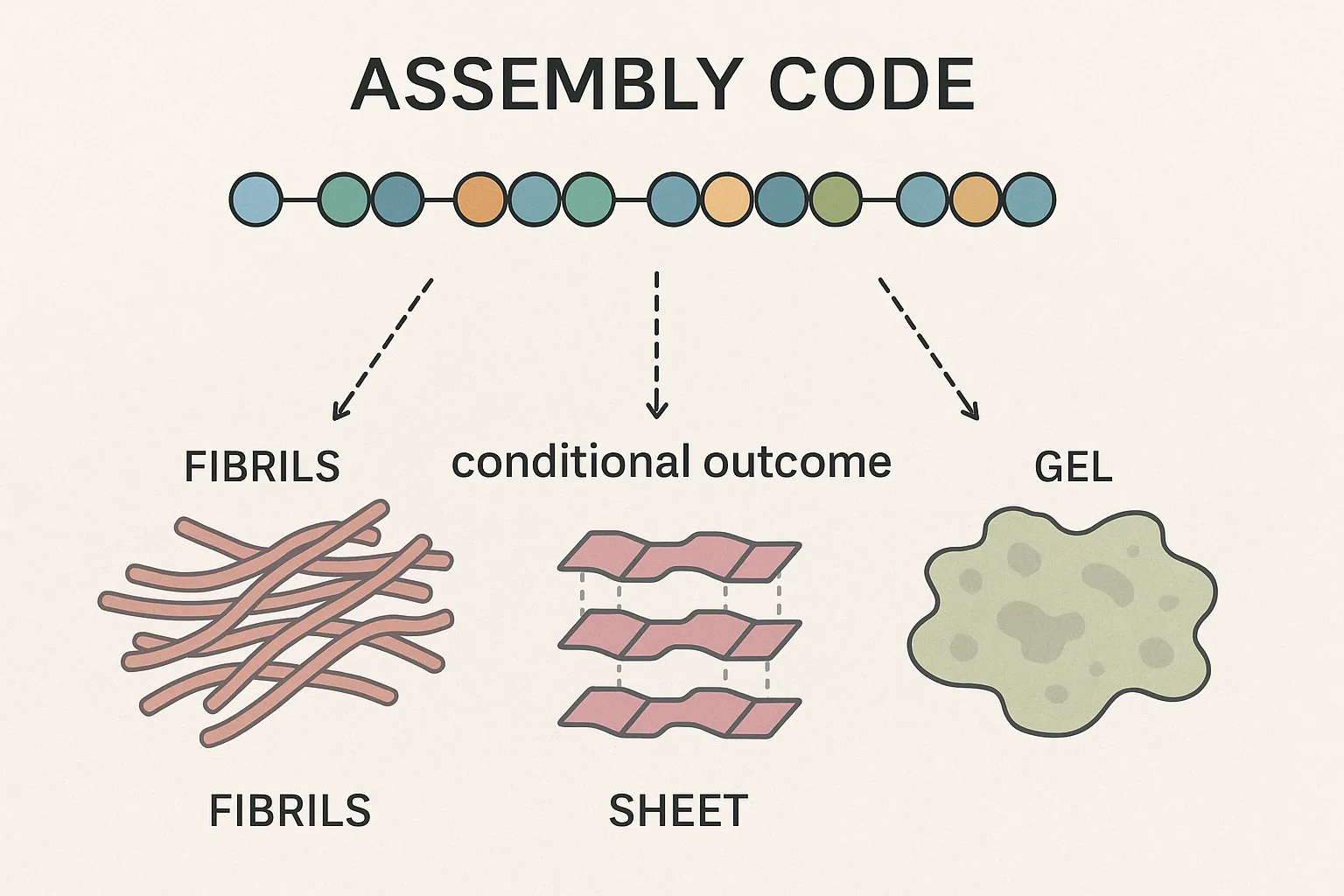
Yet, the analogy to a genetic code has limitations. Unlike protein folding, which typically converges on a unique and thermodynamically favored state, peptide self-assembly is inherently multivalent and plastic. A single sequence may encode the potential for multiple morphologies, with the dominant structure determined by external conditions. In this sense, the “code” is less a deterministic script than a set of possibilities awaiting contextual interpretation.
This nuance becomes particularly evident when comparing sequences with subtle modifications. A single amino acid substitution can shift the balance between hydrophobic and hydrophilic residues, thereby altering the assembly pathway. Similarly, charge distribution along the backbone can tune intermolecular interactions, making the same sequence capable of producing fibrils in one environment and sheets or gels in another.
Thus, while the assembly code remains a valuable guiding principle, it must be understood as conditional rather than absolute. Peptide design, therefore, requires not only sequence-level considerations but also an appreciation of how the surrounding environment influences structural outcomes.
When Context Rewrites the Code
While peptide sequence provides the foundation for self-assembly, it is the context in which assembly occurs that ultimately determines structural outcomes. Environmental factors such as pH, ionic strength, solvent polarity, and temperature exert profound influence on the balance of intermolecular forces. Even subtle shifts in these variables can redirect assembly pathways, yielding morphologies that differ dramatically from those predicted by sequence alone.
A well-documented example lies in β-sheet–forming peptides. In neutral aqueous conditions, these sequences often assemble into fibrillar architectures stabilized by backbone hydrogen bonds. However, under acidic or high-salt conditions, the same peptides may form flat ribbons or sheet-like aggregates, highlighting the role of electrostatics and solvation in modulating supramolecular order. Similarly, amphiphilic peptides—with alternating hydrophobic and hydrophilic residues—illustrate the plasticity of assembly outcomes. Depending on concentration, ionic composition, and co-solvents, they can generate spherical micelles, lamellar vesicles, or extended hydrogels, each with distinct physical and biological properties.
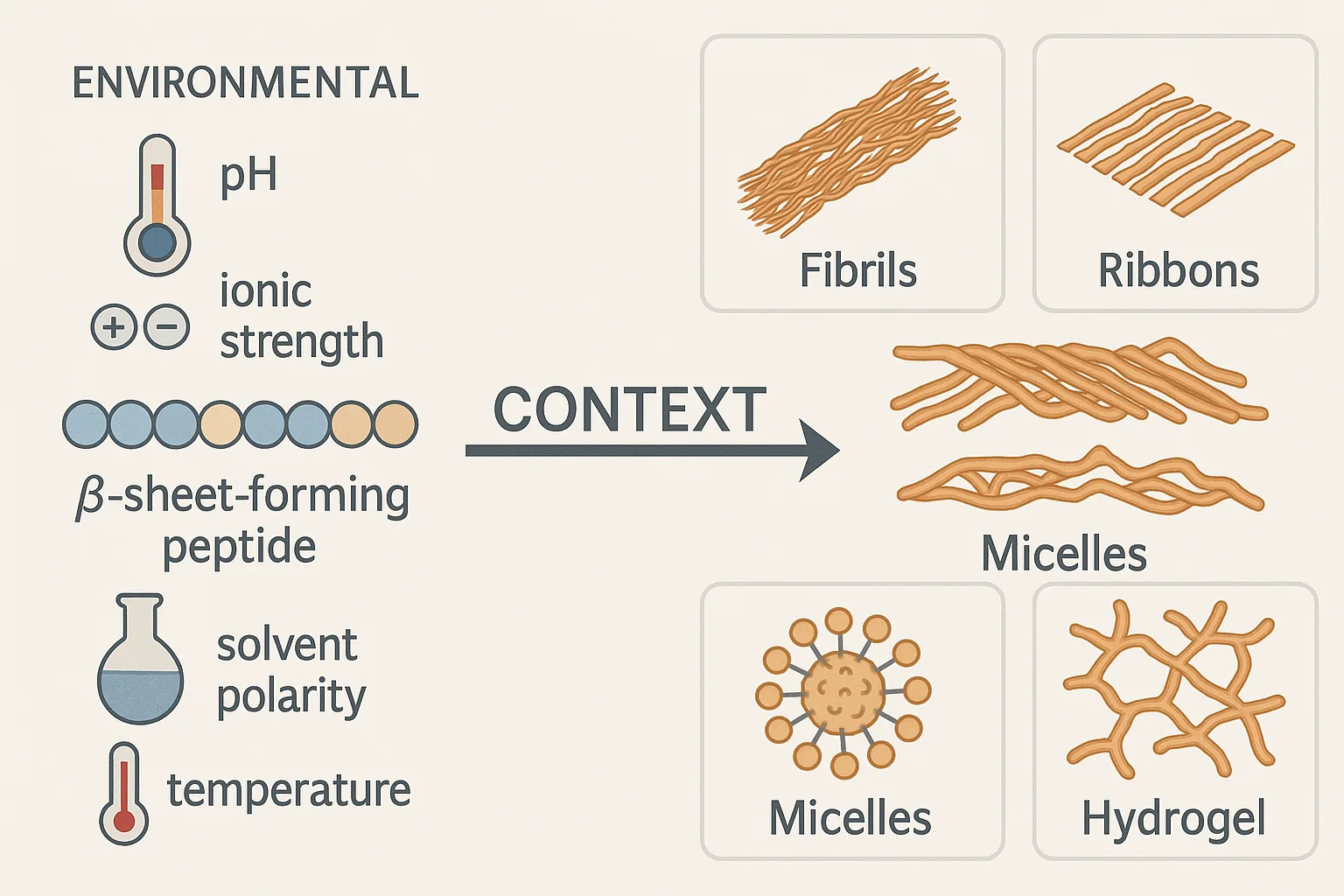
Context dependence extends beyond solution conditions to include molecular crowding and co-assembly partners. The introduction of nucleic acids, proteins, or synthetic polymers can template or stabilize alternative structures, creating hybrid materials with emergent functionalities. For instance, peptide–polymer conjugates can produce hierarchical assemblies not accessible to peptides alone, while interactions with biomacromolecules can mimic or even modulate biological processes such as amyloid formation.
Crucially, this context sensitivity introduces a multistable character to peptide assemblies. Rather than converging on a single lowest-energy structure, peptides navigate an energy landscape shaped by external cues, making them inherently adaptable. This dynamic responsiveness not only complicates prediction but also offers opportunities: the same sequence can serve as a versatile platform, producing different materials depending on the intended application.
Designing with Variability: Applications of Context Dependence
The recognition that peptide self-assembly is profoundly context-dependent has far-reaching consequences for the design of functional biomaterials. Whereas the deterministic “assembly code” once encouraged the pursuit of fixed and predictable outcomes, the modern view embraces variability and responsiveness as assets. By understanding how environmental factors modulate supramolecular organization, researchers can intentionally design adaptive systems that harness context dependence rather than attempt to suppress it.
One major opportunity lies in the development of stimuli-responsive materials. Because peptides readily reorganize in response to changes in pH, ionic strength, redox conditions, or the presence of cofactors, they can be engineered into smart scaffolds that dynamically adapt to biological environments. For example, a hydrogel scaffold designed for tissue engineering might remain stable under physiological conditions yet disassemble in the presence of a specific enzymatic trigger, releasing bioactive agents in a controlled manner. Similarly, peptide nanostructures that switch morphology with pH or temperature can serve as precision-controlled drug delivery vehicles, releasing therapeutics only under targeted pathological conditions.
Another implication is the potential for co-assembly strategies. By pairing peptides with polymers, nucleic acids, or proteins, researchers can generate hybrid materials with emergent properties not attainable by peptides alone. Such systems enable hierarchical architectures, improved mechanical robustness, and even biofunctional mimicry. For instance, peptide–polymer conjugates can yield mechanically tunable scaffolds for regenerative medicine, while peptide–DNA hybrids may create programmable nanostructures for sensing and diagnostics.
Yet, the promise of context dependence also introduces challenges. Predictability remains limited, as small variations in conditions can produce divergent outcomes, complicating reproducibility across laboratories or clinical settings. This unpredictability underscores the need for computational modeling and integrative experimental approaches to better map the multistable energy landscapes of peptide assemblies. Furthermore, scalable production requires careful control of assembly conditions to ensure material consistency.
Ultimately, the shift toward context-aware design transforms peptides from static building blocks into dynamic, programmable platforms. Rather than asking, “What structure does this sequence form?” the more powerful question becomes, “Under which conditions will this sequence produce the structure we need?” This reframing opens the door to highly versatile materials capable of adapting to both biomedical and technological demands.
Toward Adaptive Peptide Materials
The study of supramolecular peptide materials is undergoing a conceptual transformation. Once framed primarily through the lens of a deterministic “assembly code,” peptide self-assembly is now recognized as a context-dependent process in which sequence information interacts with environmental variables to yield diverse structural and functional outcomes. This shift in perspective challenges the notion of predictability, but it also reveals a powerful design space—one in which adaptability and responsiveness are not limitations but advantages.
By embracing context dependence, researchers can engineer peptide-based systems that are not only structurally sophisticated but also functionally dynamic, capable of responding to the complex and fluctuating conditions of biological and technological environments. Such materials promise innovations ranging from adaptive scaffolds in regenerative medicine to programmable nanostructures for targeted therapies and diagnostics.
Looking forward, the integration of computational modeling, high-throughput experimentation, and interdisciplinary design strategies will be essential to fully harness this paradigm. Ultimately, the future of peptide science lies not in treating assembly as a fixed outcome, but in mastering the art of contextual interpretation, enabling the next generation of smart, multifunctional biomaterials.
Reference
Kaygisiz, K., Sementa, D., Athiyarath, V., Chen, X., & Ulijn, R. V. (2025). Context dependence in assembly code for supramolecular peptide materials and systems. Nature Reviews Materials, 1-24.
https://doi.org/10.1038/s41578-025-00782-6
Kojima, K., & Sudo, Y. (2023). Convergent evolution of animal and microbial rhodopsins. RSC advances, 13(8), 5367-5381.
https://doi.org/10.1039/D2RA07073A
de Greef, T. F., & Meijer, E. W. (2008). Supramolecular polymers. Nature, 453(7192), 171-173.
https://doi.org/10.1038/453171a
Yuan, C., Fan, W., Zhou, P., Xing, R., Cao, S., & Yan, X. (2024). High-entropy non-covalent cyclic peptide glass. Nature Nanotechnology, 19(12), 1840-1848.
 LinkPeptide
LinkPeptide