Peptide-Based Therapeutics: Overcoming Challenges to Unlock New Horizons in Drug Development
Abstract
Peptide-based therapeutics are increasingly recognized for their high specificity, low immunogenicity, and ability to mimic biological functions, positioning them as valuable tools in modern drug development. However, despite their potential, these drugs face considerable challenges, including poor stability, limited bioavailability, and complex delivery requirements. This blog examines the major hurdles in peptide drug development, including susceptibility to enzymatic degradation, poor membrane permeability, and non-specific interactions. It also explores innovative solutions such as backbone modifications, cyclization, PEGylation, lipid attachment, and advanced delivery systems like implantable devices and inhalable formulations. These advancements aim to enhance peptide stability, pharmacokinetics, and therapeutic efficacy, broadening their applicability in fields such as oncology, endocrinology, and infectious diseases. As research continues, peptide therapeutics are expected to become increasingly pivotal in targeted and precision medicine, addressing previously unmet medical needs.
Exploring the Potential of Peptide-Based Drugs in Modern Medicine
Peptide-based therapeutics represent a rapidly growing category within pharmaceutical research, celebrated for their high specificity, effectiveness, and relatively low immunogenicity compared to traditional small molecules and larger biologics. Derived from natural amino acid sequences, therapeutic peptides typically range from 500 to 5000 Da in molecular weight, allowing them to mimic or enhance the functions of endogenous molecules, such as hormones, growth factors, and neurotransmitters[1]. The unique structural properties of peptides enable them to interact with various biological targets, including cell surface receptors and intracellular proteins, making them valuable for treating conditions like cancer, diabetes, and infectious diseases[2].
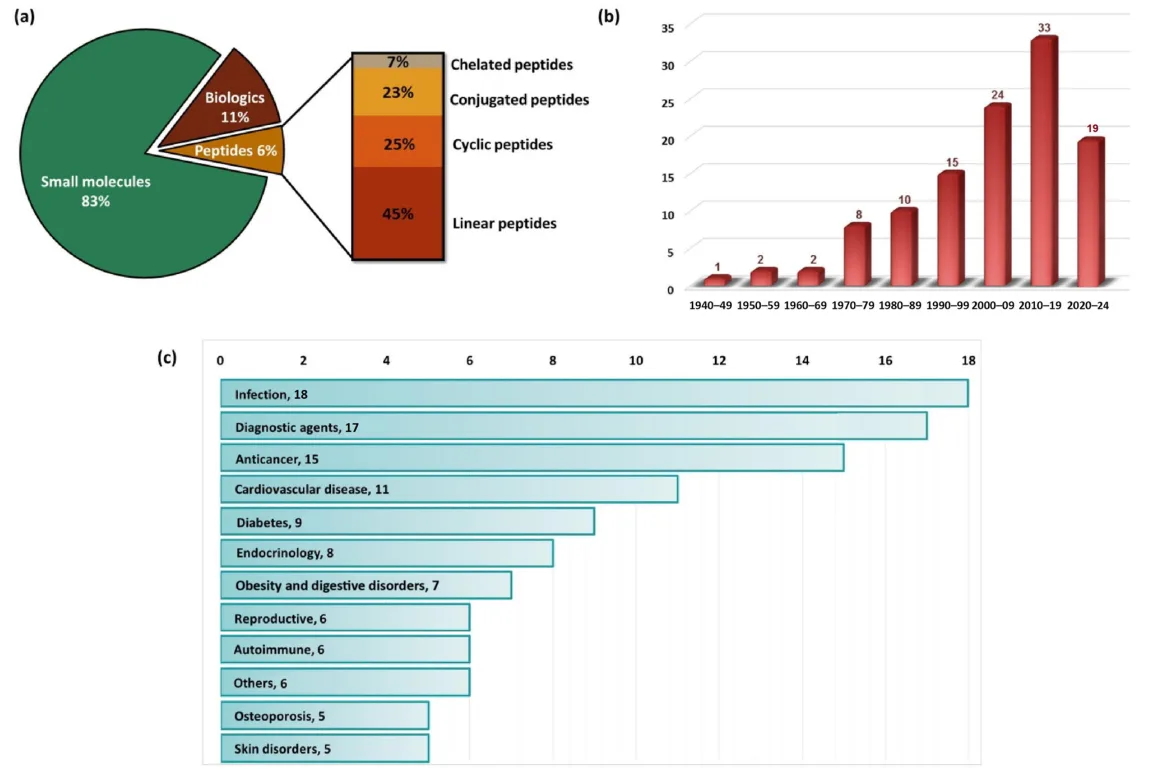
The history of peptide therapeutics began in earnest with the discovery and therapeutic application of insulin in the 1920s. Since then, the field has advanced considerably, now encompassing over 110 approved peptide-based drugs worldwide as of 2024, with a projected market growth to reach $68.7 billion by 2030[3]. Despite these advances, peptide drugs face challenges such as stability, membrane permeability, and limited bioavailability, which have spurred ongoing research to develop innovative stabilization and delivery methods. As pharmaceutical research continues to advance in the fields of structural biology, synthetic chemistry, and drug delivery, peptide therapeutics are expected to play an increasingly significant role in personalized and targeted medicine.
Understanding the Hurdles: Stability, Bioavailability, and Delivery Issues in Peptide Therapeutics
Peptide-based drugs, despite their promising therapeutic potential, face significant challenges that limit their clinical applicability. One primary challenge is their chemical and enzymatic instability, as peptide bonds are particularly susceptible to hydrolysis and degradation by proteolytic enzymes in biological environments. This vulnerability often results in a short half-life and rapid elimination from the body, necessitating frequent dosing or advanced stabilization methods[4].
Another major hurdle for peptide drugs is their poor membrane permeability. Due to their hydrophilic nature and relatively large molecular size, peptides struggle to cross cellular membranes, limiting their ability to reach intracellular targets. This low bioavailability complicates administration routes and restricts therapeutic efficacy, especially in cases where direct cell entry is required[5]. Additionally, peptides may exhibit non-specific interactions with plasma proteins, which can diminish their therapeutic activity and increase the risk of side effects. These interactions, combined with their susceptibility to immune recognition, can lead to rapid clearance from the bloodstream[6].
Finally, unlike larger protein biologics, natural peptides lack the secondary and tertiary structures that provide additional stability. Without these structures, peptides are more prone to losing their functional shape and activity in vivo. These challenges highlight the need for innovative strategies in peptide design, structural modifications, and formulation approaches to improve the stability, selectivity, and therapeutic performance of peptide-based drugs.
Enhancing Peptide Stability: From Backbone Modifications to Cyclization Techniques
To address the inherent instability and limited bioavailability of peptide drugs, several innovative stabilization techniques have been developed to enhance their therapeutic potential. One key approach involves backbone modifications—replacing natural L-amino acids with D-amino acids or incorporating N-methylated amino acids. These alterations enhance proteolytic stability, as the modified backbone resists degradation by enzymes[7]. Additionally, introducing non-natural amino acids like β-amino acids or peptoids in vulnerable sites can significantly prolong peptide half-life.
Cyclization is another effective strategy, where linear peptides are chemically modified to form cyclic structures, such as head-to-tail or side-chain-to-side-chain cyclization. This process not only stabilizes secondary structures, like α-helices and β-sheets, but also improves cell permeability and proteolytic resistance. By constraining the peptide into a cyclic structure, it adopts a more stable conformation, effectively reducing degradation and enhancing its interaction with target receptors.
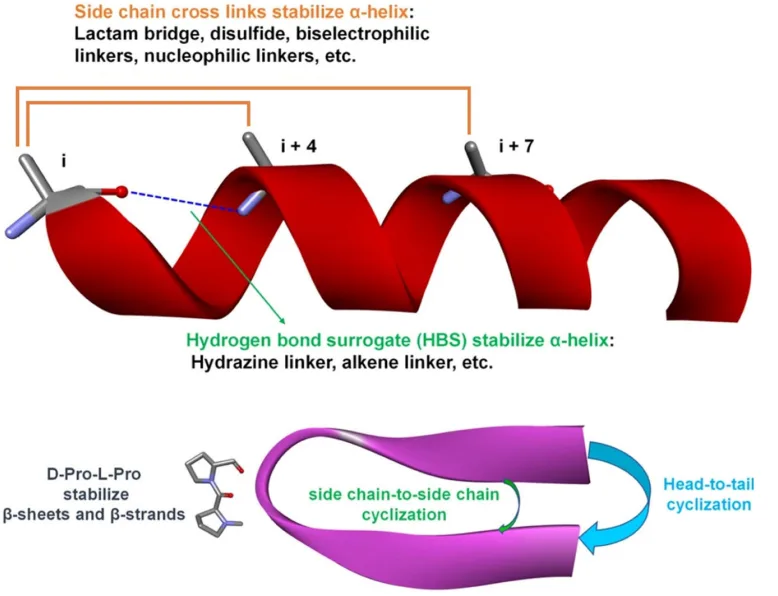
Moreover, cross-linking techniques such as “stapling” are frequently employed to reinforce the α-helical structure of peptides. Stapled peptides introduce covalent bonds between amino acid residues (often at i, i+4 or i, i+7 positions), which stabilize the helical conformation and increase affinity for target proteins. This approach is especially valuable for targeting intracellular protein-protein interactions, where stability and specificity are paramount. Collectively, these innovative modifications are pivotal in advancing peptide therapeutics, allowing for greater durability, targeted action, and reduced dosage frequency in clinical applications.
Improving Peptide Pharmacokinetics: PEGylation, Lipidation, and Beyond
To improve the pharmacokinetics of peptide-based drugs, researchers employ various chemical modifications that enhance their stability, solubility, and circulation time in the body. PEGylation, or the attachment of polyethylene glycol (PEG) chains to peptides, is a widely used technique to increase molecular weight, reduce renal clearance, and protect the peptide from enzymatic degradation. PEGylation also minimizes immunogenicity and prolongs half-life, allowing for extended drug efficacy with less frequent dosing.
Another effective strategy involves the attachment of lipids or larger proteins to peptides. By conjugating peptides with fatty acids or proteins like serum albumin, their molecular size increases, decreasing renal filtration and extending their circulation time. This approach is particularly beneficial in therapies requiring sustained peptide activity, as seen with GLP-1 analogs like liraglutide and semaglutide, which have been modified with fatty acid chains for prolonged release and stability[8].
In addition to PEGylation and lipidation, advanced drug delivery systems such as solid lipid nanoparticles (SLNs), liposomes, and self-emulsifying drug delivery systems (SEDDS) are being developed to enhance peptide bioavailability and stability. These systems enable peptides to overcome biological barriers, improve absorption, and ensure targeted delivery to specific tissues. By encapsulating peptides in such formulations, their therapeutic efficacy and overall bioavailability are significantly increased, thereby addressing some of the key limitations in peptide-based therapeutics.
Pioneering Delivery Systems for Peptide Drugs: Oral, Transdermal, and Implantable Solutions
Overcoming the challenges associated with peptide delivery is essential to fully unlock their therapeutic potential. Traditional administration routes are often unsuitable due to peptides’ low oral bioavailability and susceptibility to enzymatic degradation. Oral administration remains challenging, but advances in formulation, such as co-formulation with permeation enhancers, have shown promise. For instance, semaglutide, a GLP-1 analog, utilizes sodium N-[8-(2-hydroxybenzoyl) amino]caprylate (SNAC) as an absorption enhancer, allowing it to withstand the digestive environment and be effectively absorbed through the gastric membrane.
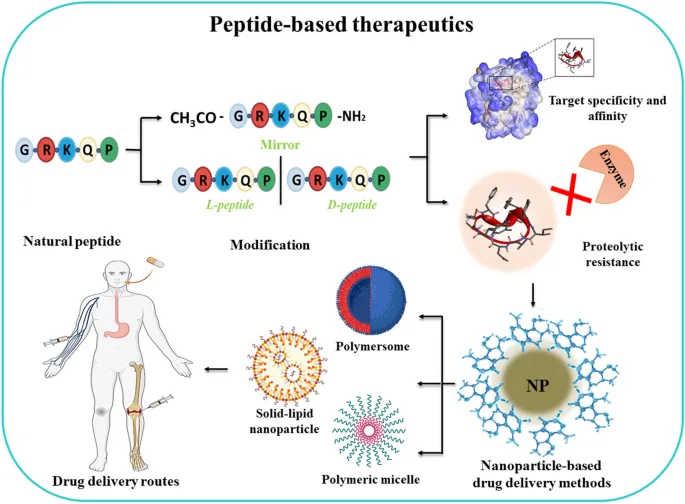
In addition to oral methods, alternative routes like transdermal and pulmonary delivery offer novel pathways for peptide drugs. Transdermal patches and inhalable formulations allow peptides to bypass the digestive system entirely, reducing degradation and potentially enhancing absorption. Inhalable insulin and peptide vaccines are examples of successful pulmonary delivery approaches, where peptides are delivered directly to the bloodstream via lung tissue, achieving rapid onset and avoiding the challenges of gastrointestinal absorption.
Furthermore, implantable devices, such as micro-pumps, are being developed to ensure sustained and controlled peptide release. These devices offer a promising option for conditions requiring continuous peptide dosing, providing precise and programmable administration rates. As these technologies advance, they are expected to expand the range of applications for peptide-based therapeutics, offering improved patient compliance and enhanced efficacy across various therapeutic areas[9].
Peptide Therapeutics: Future Perspectives in Overcoming Challenges for Targeted Drug Development
Peptide-based therapeutics have emerged as a promising class of drugs, offering high specificity, low immunogenicity, and versatility in addressing diverse medical conditions. However, despite these advantages, significant challenges in stability, bioavailability, and delivery limit their broader application. Advances in structural modifications, such as backbone and side-chain alterations, and stabilization techniques like PEGylation and lipidation, have proven effective in enhancing peptide pharmacokinetics and therapeutic efficacy. Additionally, innovative delivery systems, including oral permeation enhancers, transdermal and pulmonary routes, and implantable devices, have opened new avenues for peptide drug administration, improving patient compliance and expanding therapeutic options.
Looking forward, continued research in peptide chemistry, formulation science, and drug delivery technologies will be critical in overcoming the remaining barriers in peptide-based therapeutics. Integration of peptide drugs with precision medicine approaches and personalized delivery systems could further enhance their effectiveness, enabling tailored treatments for individual patients. As new strategies emerge, peptides are anticipated to play an increasingly central role in modern pharmaceuticals, particularly in fields such as oncology, endocrinology, and infectious diseases. The growing interest and investment in peptide therapeutics highlight their potential to shape the future of targeted therapies, potentially leading to breakthroughs in previously unmet clinical needs.
 LinkPeptide
LinkPeptide