Revealing the Vulnerable Moment: Fusion Peptide Dynamics in HIV-1 Entry
Abstract
Recent structural studies have illuminated a critical aspect of HIV-1 entry: the dynamic trajectory of the fusion peptide (FP) during CD4-induced Env opening. While the FP is a known site of vulnerability to broadly neutralizing antibodies (bnAbs) in the closed Env conformation, its fate during receptor engagement was previously unclear. A 2025 Nature Communications study reveals a stepwise process in which the FP remains transiently accessible even after substantial Env rearrangement. Using cryo-EM, surface plasmon resonance, and single-molecule FRET, the authors characterize a functional intermediate—Population 1—where the FP is still targetable. These findings uncover a structural window during which bnAbs like VRC34.01 can bind and neutralize the virus, reshaping strategies for immunogen design. This mechanistic clarity highlights how HIV-1 balances immune evasion and fusion readiness and offers actionable insights for vaccine and therapeutic development.
Unmasking HIV-1’s Fusion Peptide: A Hidden Opportunity in Viral Entry
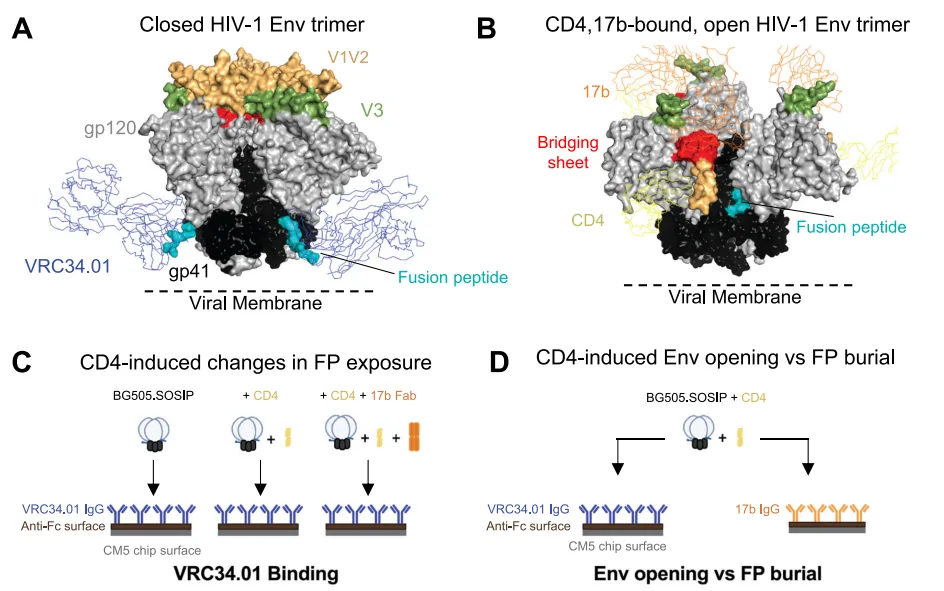
The HIV-1 envelope glycoprotein (Env), a trimer of gp120-gp41 heterodimers, is central to viral entry and immune evasion. A key feature of this complex is the fusion peptide (FP)—a short, hydrophobic segment at the N-terminus of gp41—that inserts into the host membrane to initiate fusion. Notably, in its prefusion-closed conformation, the FP is solvent-exposed and accessible to broadly neutralizing antibodies (bnAbs), such as VRC34.01. This exposure defines it as a critical site of viral vulnerability and a target of vaccine interest.
However, upon receptor binding—specifically to CD4—Env undergoes a series of conformational transitions that culminate in FP burial within a gp120-gp41 pocket, rendering it inaccessible to antibody binding. While structures of the FP in its accessible and buried forms have been resolved, the mechanistic steps connecting these states have remained elusive.
A recent study published in Nature Communications (2025) addresses this gap. Using high-resolution cryo-electron microscopy, supported by surface plasmon resonance and single-molecule FRET, the authors delineate a stepwise trajectory of FP reorganization during CD4-induced Env opening. Their findings reveal not only intermediate states where FP remains antibody-accessible despite substantial Env opening, but also the structural determinants that drive its eventual sequestration.
This work refines our understanding of the HIV-1 entry process and reveals a previously underappreciated window during which the FP remains targetable—an insight with direct implications for immunogen design and bnAb-based therapeutics.
Dissecting Env Opening: Structural Intermediates Along the Entry Pathway
HIV-1 entry begins when the Env trimer engages the CD4 receptor on host cells. This interaction triggers a cascade of allosteric rearrangements within the Env complex, collectively referred to as “Env opening.” The gp120 subunits undergo rigid-body displacement and rotation, leading to the disruption of inter-protomer interactions—particularly within the V1V2 and V3 loop regions at the trimer apex—and the formation of the bridging sheet, a hallmark of CD4-induced opening.
During this process, gp41 is reorganized to initiate membrane fusion. Critically, these structural changes reposition the fusion peptide (FP) from its exposed location at the base of the trimer into a newly formed, hydrophobic pocket at the gp120-gp41 interface. This pocket formation is key to rendering the FP antibody-inaccessible. However, the sequence and dynamics of this transition were not well understood until now.
In this study, cryo-EM analysis of BG505 SOSIP Env trimers incubated with CD4 and the coreceptor-mimicking antibody 17b revealed three conformational states (Populations 1–3), each defined by distinct stoichiometries of FP-directed antibody (VRC34.01) binding.
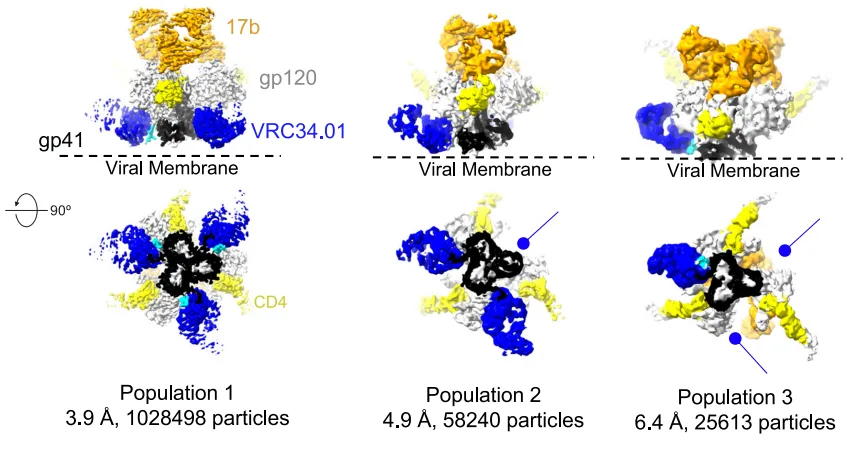
- Population 1: All three protomers are bound by CD4, 17b, and VRC34.01—indicating an open Env conformation with accessible FP.
- Population 2: Only two FP sites remain bound by VRC34.01; one FP is buried.
- Population 3: Just one accessible FP remains.
These structures demonstrate that Env opening does not immediately occlude the FP. Instead, there is a transitional window where the virus is increasingly primed for fusion but remains partially vulnerable to neutralization. This nuanced view of Env dynamics provides a refined framework for interpreting antibody-Env interactions and designing immunogens that exploit intermediate conformational states.
Transient Exposure, Lasting Impact: Fusion Peptide Accessibility and Neutralization Potential
One of the central insights from this study is that the fusion peptide (FP) remains accessible to antibody binding well into the Env opening process. This defies the prevailing assumption that FP burial coincides directly with early receptor engagement. Instead, the data reveal a stepwise transition, where FP accessibility is gradually lost as the conformational landscape evolves.
The authors used the FP-specific broadly neutralizing antibody VRC34.01 as a structural probe. In Population 1, the FP remains exposed in all three protomers, even though Env is fully bound by CD4 and 17b—both markers of advanced opening. This state is not only structurally symmetrical but also functionally consistent with prior cryo-ET observations of CD4-bound Env on virions, supporting its physiological relevance.
As the Env progresses to Populations 2 and 3, the number of VRC34.01-bound sites decreases to two and one, respectively. Detailed structural analysis shows that in unbound protomers, the FP is sequestered into a gp120/gp41 hydrophobic pocket, becoming sterically inaccessible. This burial correlates with increased lateral displacement of gp120, a movement orthogonal to the trimer axis, and reorganization of gp41, including straightening of the fusion peptide proximal region (FPPR) and alterations in HR1/HR2 helices. These rearrangements enlarge the FP-binding cavity and stabilize FP occlusion.
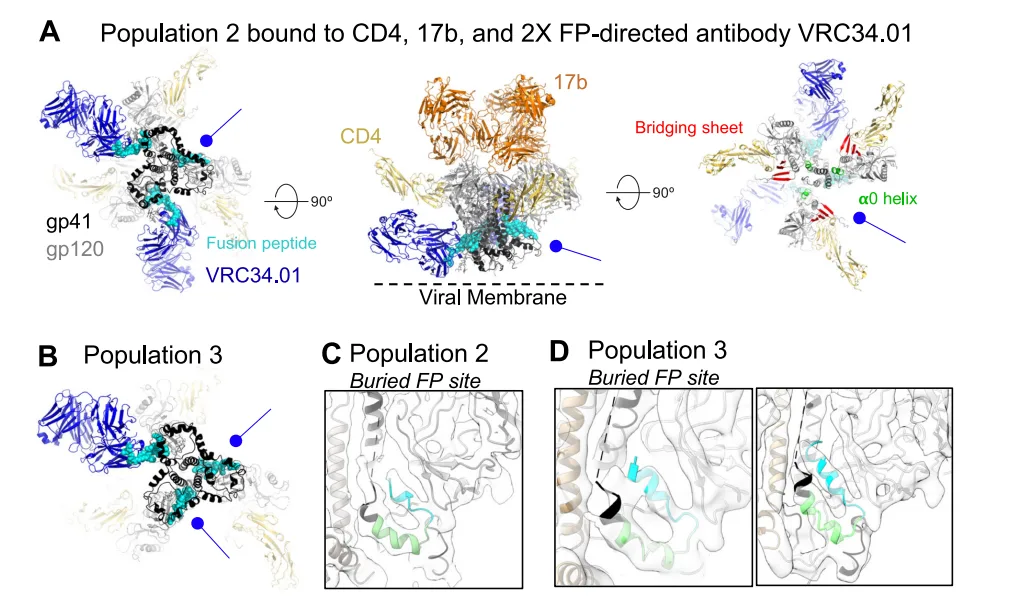
Importantly, this conformational spectrum suggests that FP targeting antibodies may neutralize HIV-1 during a specific intermediate stage—after CD4 engagement but before full FP burial. The identification of this intermediate, characterized by both CD4-triggered gp120 reorientation and persistent FP accessibility, provides a structural rationale for how VRC34.01 and similar antibodies achieve broad neutralization.
In essence, the study uncovers a previously unappreciated “neutralization window”—a fleeting structural state that could be exploited by vaccines or therapeutics targeting the FP.
Bridging Structure and Dynamics: Validating Env Intermediates on Native Virions
To validate the structural intermediates observed by cryo-EM and to assess their relevance on intact virions, the authors turned to single-molecule Förster resonance energy transfer (smFRET). This approach enables real-time monitoring of Env conformational dynamics by measuring distance-dependent energy transfer between fluorophores strategically positioned on gp120 and gp41.
Using dual-labeled HIV-1_BG505 Env embedded in virions, smFRET analysis identified three primary conformational states:
- Pre-triggered (PT)
- Prefusion-closed (PC)
- CD4-bound open (CO)
In the ligand-free state, Env predominantly resided in the PT conformation. Upon addition of CD4 and 17b, it shifted toward the fully open CO state. However, when VRC34.01 was present—alone or in combination with CD4 and 17b—the conformational landscape shifted toward intermediate populations, consistent with the Partially Open Population 1 structure from cryo-EM.
This convergence of cryo-EM and smFRET data strengthens the conclusion that intermediate states with antibody-accessible FP exist transiently on the viral surface, not just in engineered soluble Env constructs. Moreover, the smFRET profiles illustrate that VRC34.01 can stabilize Env in an intermediate state, delaying or preventing full FP burial.
Together, these techniques present a cohesive picture: the HIV-1 Env does not flip instantaneously from closed to open but instead samples a spectrum of conformations, some of which remain amenable to antibody targeting. Capturing this continuum of structural states is crucial for guiding the design of immunogens that mimic or stabilize functional entry intermediates.
Designing for Vulnerability: Translating Structural Insights into Vaccine Innovation
The discovery of antibody-accessible fusion peptide (FP) intermediates reshapes how we think about targeting HIV-1 Env. Unlike fusion peptides in other class I viral fusion proteins—such as SARS-CoV-2 or paramyxoviruses—which are buried in the pre-fusion state and revealed only upon receptor activation, the HIV-1 FP begins exposed and is only sequestered after CD4-induced Env opening. This unusual trajectory has dual consequences: it provides a window of vulnerability, yet also raises the question—why would such a conserved epitope be left unshielded?
The authors suggest an evolutionary compromise. Full FP burial requires partial Env opening, which itself exposes other neutralization-sensitive sites. Thus, HIV-1 may favor a compact, closed trimer where the FP remains solvent-exposed but shielded by a limited glycan cloak—minimizing immunogenic exposure while avoiding broader conformational unmasking.
From a vaccine design perspective, this insight is critical. Previous immunogen strategies have focused on mimicking the prefusion-closed Env, where FP is exposed and structurally well-defined. This study now establishes that the FP retains accessibility in early entry intermediates, expanding the targetable window. It also explains why FP-based immunogens have been able to elicit bnAbs with over 50% neutralization breadth, as shown in prior clinical studies.
Furthermore, knowledge of the conformational transitions that lead to FP burial opens new avenues for immunogen engineering. Vaccines may be designed to stabilize intermediate states like Population 1, optimizing the presentation of FP in a context recognized by neutralizing antibodies such as VRC34.01 or 8ANC195.
Ultimately, this work redefines the FP not just as a static target but as a transient epitope, accessible during a critical stage of viral entry. This understanding provides a precise structural roadmap for designing next-generation HIV vaccines and antibody-based interventions.
Reference
Thakur, B., Katte, R. H., Xu, W., Janowska, K., Sammour, S., Henderson, R., … & Acharya, P. (2025). Conformational trajectory of the HIV-1 fusion peptide during CD4-induced envelope opening. Nature Communications, 16(1), 1-14.
https://doi.org/10.1038/s41467-025-59721-2
Pancera, M., Zhou, T., Druz, A., Georgiev, I. S., Soto, C., Gorman, J., … & Kwong, P. D. (2014). Structure and immune recognition of trimeric pre-fusion HIV-1 Env. Nature, 514(7523), 455-461.
https://doi.org/10.1038/nature13808
Hoffman, T. L., LaBranche, C. C., Zhang, W., Canziani, G., Robinson, J., Chaiken, I., … & Doms, R. W. (1999). Stable exposure of the coreceptor-binding site in a CD4-independent HIV-1 envelope protein. Proceedings of the National Academy of Sciences, 96(11), 6359-6364.
https://doi.org/10.1073/pnas.96.11.6359
Jackson, C. B., Farzan, M., Chen, B., & Choe, H. (2022). Mechanisms of SARS-CoV-2 entry into cells. Nature reviews Molecular cell biology, 23(1), 3-20.
 LinkPeptide
LinkPeptide