Anticancer Peptides: A New Frontier in Cancer Treatment and Drug Innovation
Abstract
Anticancer peptides (ACPs) are emerging as promising therapeutic agents in oncology, offering a safer and more targeted alternative to conventional cancer treatments. These small bioactive molecules demonstrate remarkable selectivity toward cancer cells, reducing off-target effects and toxicity. ACPs can be derived from natural sources, synthetically produced, or chemically modified to enhance their therapeutic properties. They disrupt cancer cell membranes, induce apoptosis, and inhibit tumor growth and angiogenesis. The development of ACPs is supported by advanced synthesis techniques and computational tools such as machine learning models, which accelerate discovery and optimization. Despite challenges related to cost, stability, and immunogenicity, innovative strategies involving nanotechnology and personalized medicine are paving the way for their broader application. This blog explores the current advancements, challenges, and future prospects in ACP drug development, highlighting their transformative role in modern cancer therapy.
Introduction: The Rise of Anticancer Peptides in Oncology
Cancer remains one of the leading causes of mortality worldwide, with millions of new cases diagnosed annually. Despite advancements in treatment strategies such as surgery, chemotherapy, radiotherapy, and immunotherapy, significant limitations persist, including drug resistance, off-target effects, severe toxicity, and tumor relapse[1]. These challenges necessitate the exploration of novel therapeutic options that can address the shortcomings of conventional cancer treatments.
In recent years, anticancer peptides (ACPs) have emerged as promising alternatives in oncology. These small bioactive molecules, typically composed of 10 to 50 amino acids, demonstrate a unique capacity to selectively target cancer cells while sparing healthy tissues[2,3]. Their specificity stems from interactions with negatively charged components present in cancer cell membranes, a characteristic less common in normal cells. This selectivity contributes to ACPs’ reduced toxicity compared to traditional chemotherapeutics, making them attractive candidates for safer cancer treatments.
The advantages of ACPs extend beyond therapeutic applications. In diagnostics, ACPs have shown potential in imaging and biosensing technologies, allowing for the precise detection and monitoring of cancer biomarkers. Additionally, ACPs can be used as drug delivery agents, enhancing the efficiency of other therapeutic compounds by targeting specific tumor microenvironments.
Given their versatile properties and potential in both treatment and diagnostic applications, ACPs are gaining momentum as powerful tools in the fight against cancer. Continued research efforts focus on optimizing their design, enhancing their stability, and leveraging computational techniques for their development. The ongoing advancements in ACP research are expected to further establish their role as essential components of the next generation of anticancer therapies.
Diverse Origins of Anticancer Peptides: Nature, Innovation, and Beyond
Anticancer peptides (ACPs) are diverse bioactive molecules that can be classified based on their sources into natural, synthetic, and modified categories. Natural ACPs are derived from various organisms, including animals, plants, and microorganisms. These peptides often originate as part of innate immune systems, providing natural defenses against pathogens and exhibiting cytotoxic activity against cancer cells. For example, Magainins were first isolated from the skin secretions of the African clawed frog (Xenopus laevis), while Melittin is a potent anticancer peptide found in bee venom[4].
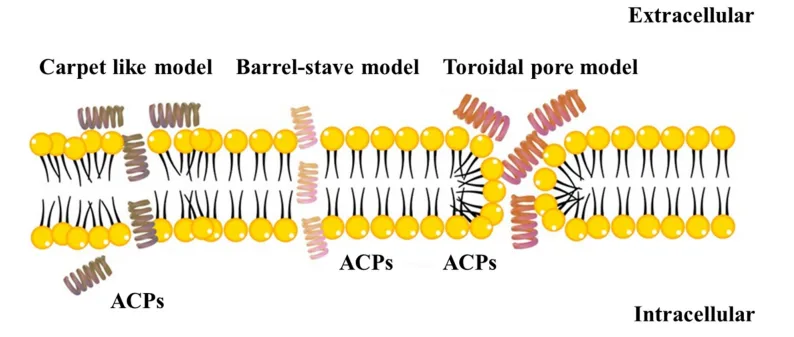
In addition to animal sources, plants and microbial organisms also produce ACPs with significant therapeutic potential. Defensins and thionins from plants have demonstrated activity against cancer cells by disrupting their membrane integrity[5]. Similarly, microbial-derived peptides like bacteriocins have shown promise as selective anticancer agents due to their unique structural features and low toxicity to normal cells.
Synthetic ACPs are designed using advanced chemical techniques to improve stability, enhance therapeutic efficacy, and reduce immunogenicity. Examples include hybrid peptides combining domains from different natural peptides to increase their anticancer activity. Modifications such as PEGylation, lipidation, or incorporation of non-natural amino acids further boost the potency and selectivity of synthetic ACPs[6].
These versatile peptides target cancer cells through diverse mechanisms, including membrane disruption, apoptosis induction, and inhibition of angiogenesis. With their broad range of origins and structural properties, ACPs offer immense potential as safer alternatives to conventional cancer treatments, paving the way for innovative diagnostic and therapeutic solutions.
Innovative Techniques for ACP Synthesis and Chemical Modifications
The development of anticancer peptides (ACPs) relies heavily on efficient synthesis and strategic chemical modifications to enhance their therapeutic potential. One of the most widely used methods for synthesizing peptides is Solid Phase Peptide Synthesis (SPPS). This technique enables the sequential addition of amino acids to a growing peptide chain attached to a solid resin, facilitating rapid and efficient synthesis of peptides with high purity[3]. SPPS is particularly advantageous for producing peptides with complex sequences and allows the incorporation of non-natural amino acids. Despite its efficiency, the technique faces challenges related to solvent consumption and hazardous waste production, motivating efforts toward greener alternatives.
Chemical modifications are essential for optimizing the stability, selectivity, and bioavailability of ACPs. Cholesterol coupling enhances the ability of peptides to penetrate cancer cell membranes by promoting self-assembly and lipid interactions. PEGylation, the attachment of polyethylene glycol (PEG) chains, extends the half-life of ACPs in the bloodstream by increasing their hydrodynamic size, reducing proteolysis, and minimizing immunogenicity. Additionally, phosphorylation of threonine, serine, or tyrosine residues can modulate the activity and toxicity of ACPs, further improving their safety profile.
These synthesis methods and chemical enhancements collectively contribute to improving the pharmacological properties of ACPs, making them more effective and reliable candidates for cancer therapy. As research advances, new strategies are being developed to further refine their therapeutic profiles and overcome limitations in large-scale production.
Overcoming Barriers: Challenges in Anticancer Peptide Development
Despite their promising therapeutic potential, the large-scale development and clinical application of anticancer peptides (ACPs) face several significant challenges. One of the primary issues is the cost and scalability of synthesis. The production of peptides, especially longer and structurally complex ones, often requires sophisticated equipment and expensive reagents, which limits their affordability for widespread clinical use. Techniques such as Solid Phase Peptide Synthesis (SPPS) have streamlined production processes but remain resource-intensive and environmentally burdensome.
Another critical challenge is proteolytic degradation. Once administered, ACPs are prone to enzymatic degradation by peptidases present in the human body, which rapidly reduces their bioavailability and therapeutic efficacy. This susceptibility significantly limits their effectiveness as systemic treatments for cancer. Strategies to counteract this problem include chemical modifications such as cyclization, incorporation of non-natural amino acids, and PEGylation to enhance peptide stability.
Immunogenicity concerns present a further obstacle. Since ACPs are often derived from natural sources, their sequences may resemble endogenous peptides, raising concerns about their potential to trigger undesired immune responses. While synthetic and modified peptides can be designed to minimize immunogenicity, the trade-off is often reduced biological activity.
Efforts are underway to address these challenges by exploring novel synthesis techniques, enhancing peptide stability through chemical modifications, and leveraging computational tools to optimize ACP design for improved therapeutic outcomes. The continued development of these strategies is crucial for unlocking the full clinical potential of ACPs in oncology.
Overcoming Barriers: Challenges in Anticancer Peptide Development
The future of anticancer peptide (ACP) research holds exciting opportunities for transforming cancer treatment strategies. One key area of innovation is the integration of ACPs with nanotechnology. By conjugating ACPs with nanoparticles, researchers are developing targeted drug delivery systems that can enhance therapeutic efficacy and reduce off-target toxicity. Nanocarriers, such as liposomes and gold nanoparticles, improve ACP stability in the bloodstream and facilitate their accumulation in tumor tissues[7].
Another promising trend is the advancement of computational methods to accelerate ACP discovery and optimization. Artificial intelligence (AI) and machine learning (ML) models are being increasingly employed to predict peptide sequences with anticancer properties, minimizing the need for labor-intensive laboratory experiments. These computational approaches streamline the development process, making it faster and more cost-effective[8].
The emergence of personalized cancer therapies further underscores the transformative potential of ACPs. By analyzing a patient’s genetic profile, clinicians can select specific ACPs that target the unique molecular characteristics of their tumor. This tailored approach aims to improve treatment outcomes and minimize adverse effects, marking a significant shift toward precision oncology.
Future Directions and Emerging Trends in ACP-Based Cancer Therapies
Anticancer peptides represent a significant breakthrough in oncology, offering novel solutions for both therapeutic and diagnostic applications. Their high specificity, reduced toxicity, and diverse mechanisms of action position them as strong candidates for future cancer treatments. However, challenges such as proteolytic degradation and production costs remain barriers to their widespread adoption. Continued research and development, alongside advancements in nanotechnology and computational biology, are essential to overcome these obstacles and unlock the full potential of ACPs. As the landscape of cancer treatment evolves, ACPs are poised to play a central role in shaping innovative and more effective therapeutic strategies.
 LinkPeptide
LinkPeptide