Antimicrobial Peptides: Pioneering the Future of Infection Control and Combating Antibiotic Resistance
Abstract
The global rise in antibiotic resistance has intensified the search for alternative antimicrobial strategies. Antimicrobial peptides (AMPs) have emerged as promising candidates due to their broad-spectrum activity and unique mechanisms of action, including membrane disruption and intracellular targeting. Unlike traditional antibiotics, AMPs exhibit low resistance potential and effectively combat multidrug-resistant pathogens. Their structural diversity—ranging from α-helices to β-sheets—enhances their adaptability in targeting various microorganisms. Advances in genetic engineering, synthetic peptide design, and nanotechnology have further optimized AMP stability, bioavailability, and efficacy. Additionally, AMPs demonstrate therapeutic potential beyond antibacterial applications, showing activity against viral, fungal, and parasitic infections. Despite challenges in clinical translation, ongoing research continues to refine AMP properties for enhanced safety and effectiveness. This blog explores the structural diversity, mechanisms of action, and future directions of AMPs, highlighting their potential as next-generation therapeutics in the fight against infectious diseases.
Rising Antibiotic Resistance and the Need for Innovative Solutions
The discovery and widespread use of antibiotics in the early 20th century revolutionized modern medicine, significantly reducing mortality from bacterial infections and transforming healthcare practices. However, the irrational and excessive use of antibiotics has led to the emergence of multidrug-resistant (MDR) bacteria, posing a substantial threat to global health. According to recent reports[1], if current trends continue unchecked, antibiotic-resistant infections could result in approximately 10 million deaths annually by 2050, surpassing deaths caused by cancer and other major diseases.
Traditional antibiotics are becoming less effective due to several mechanisms of bacterial resistance, including mutations in target sites, enzymatic degradation, and efflux pumps that actively expel antibiotics from bacterial cells[2]. Moreover, the environmental impact of antibiotic misuse has exacerbated the issue. Antibiotics excreted by animals and humans enter the ecosystem, disrupting microbial communities and promoting resistance among environmental bacteria.
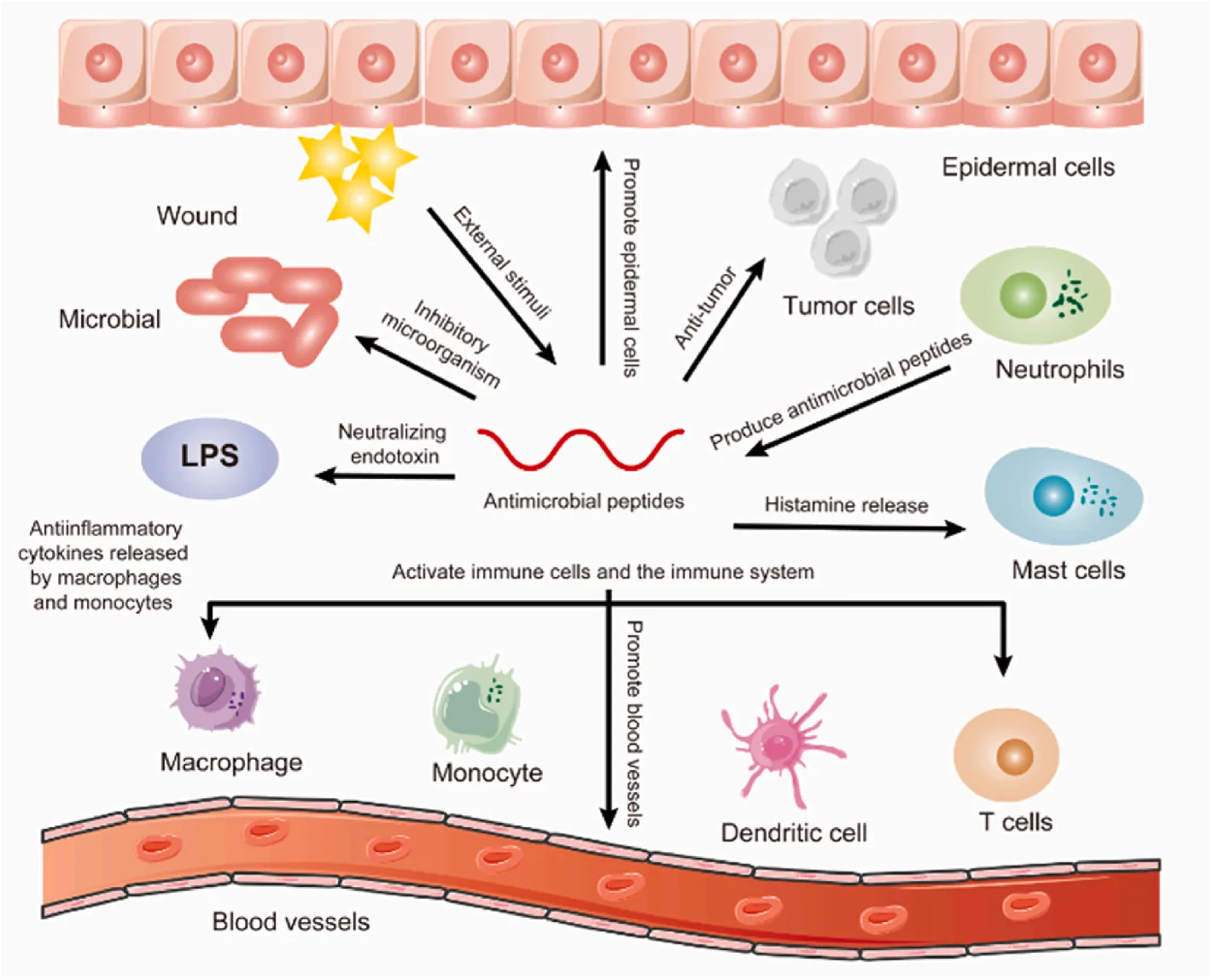
In response to this growing crisis, antimicrobial peptides (AMPs) have garnered significant attention as promising alternatives to conventional antibiotics. AMPs are naturally occurring molecules present in various organisms, including humans, animals, and plants, as part of their innate immune defense[3]. Unlike traditional antibiotics that often target specific bacterial pathways, AMPs typically disrupt bacterial membranes, leading to rapid bacterial cell death. This multitarget mode of action reduces the likelihood of resistance development and offers broad-spectrum antimicrobial properties.
Given their potential, AMPs are being explored for therapeutic applications against drug-resistant bacteria, as well as in combination therapies to enhance the efficacy of existing antibiotics. The growing body of research underscores the critical need for continued investment in the development of AMPs as viable solutions to combat antibiotic resistance and safeguard global health.
Structural Diversity and Functional Properties of Antimicrobial Peptides
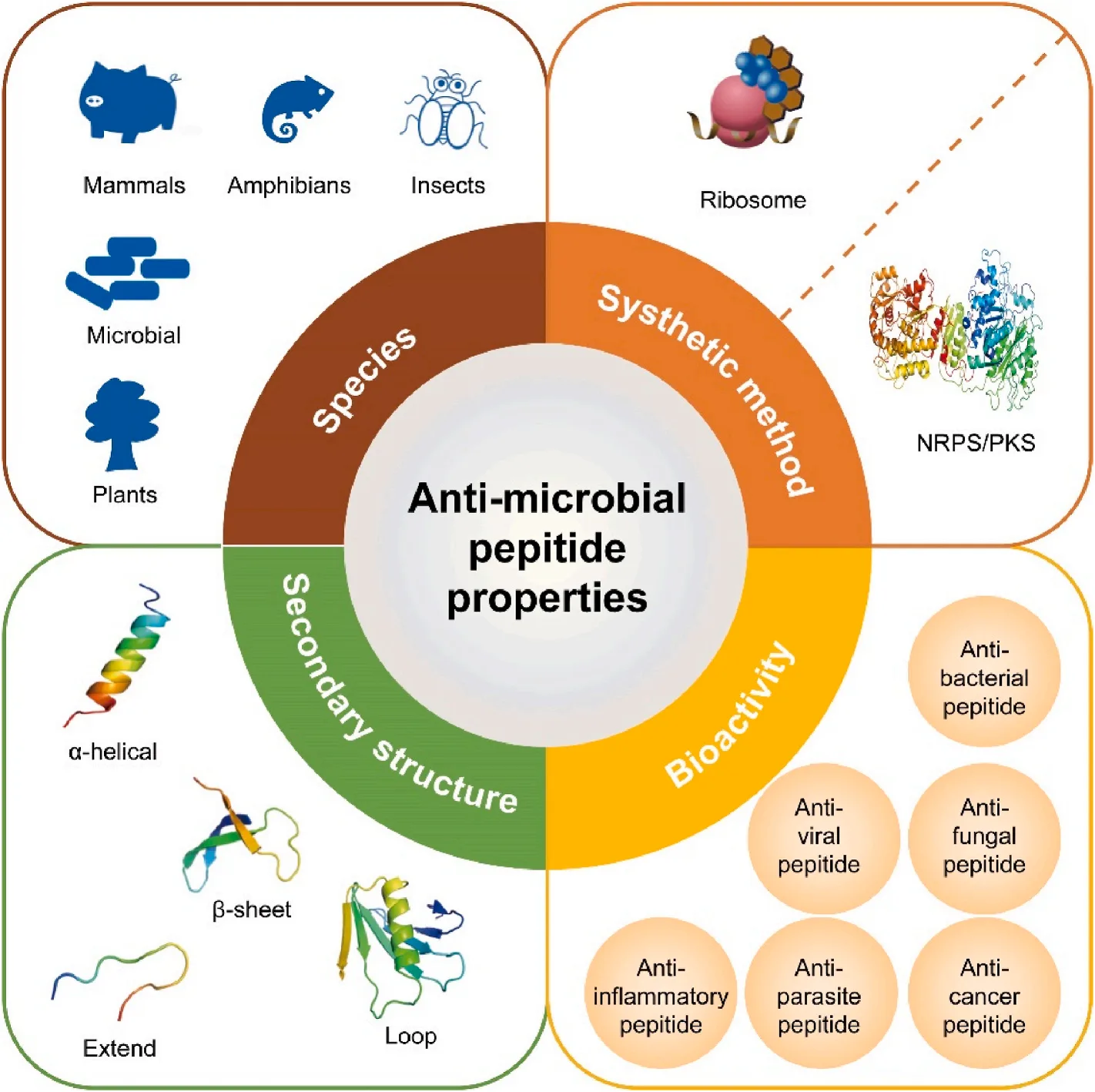
Antimicrobial peptides (AMPs) are a diverse group of molecules with significant structural and functional variability. They can be broadly classified based on their secondary structures into four major categories: α-helical peptides, β-sheet peptides, extended peptides, and looped peptides. α-helical peptides, such as cecropins, adopt amphipathic helices that interact with microbial membranes, leading to membrane disruption[4]. β-sheet peptides, exemplified by defensins, are stabilized by disulfide bonds, providing structural rigidity and antimicrobial efficacy. Extended peptides, like indolicidin, are rich in specific amino acids (e.g., proline, glycine) and exhibit flexible linear conformations, while looped peptides, such as theta-defensins, form cyclic structures that enhance their stability and resistance to proteolysis.
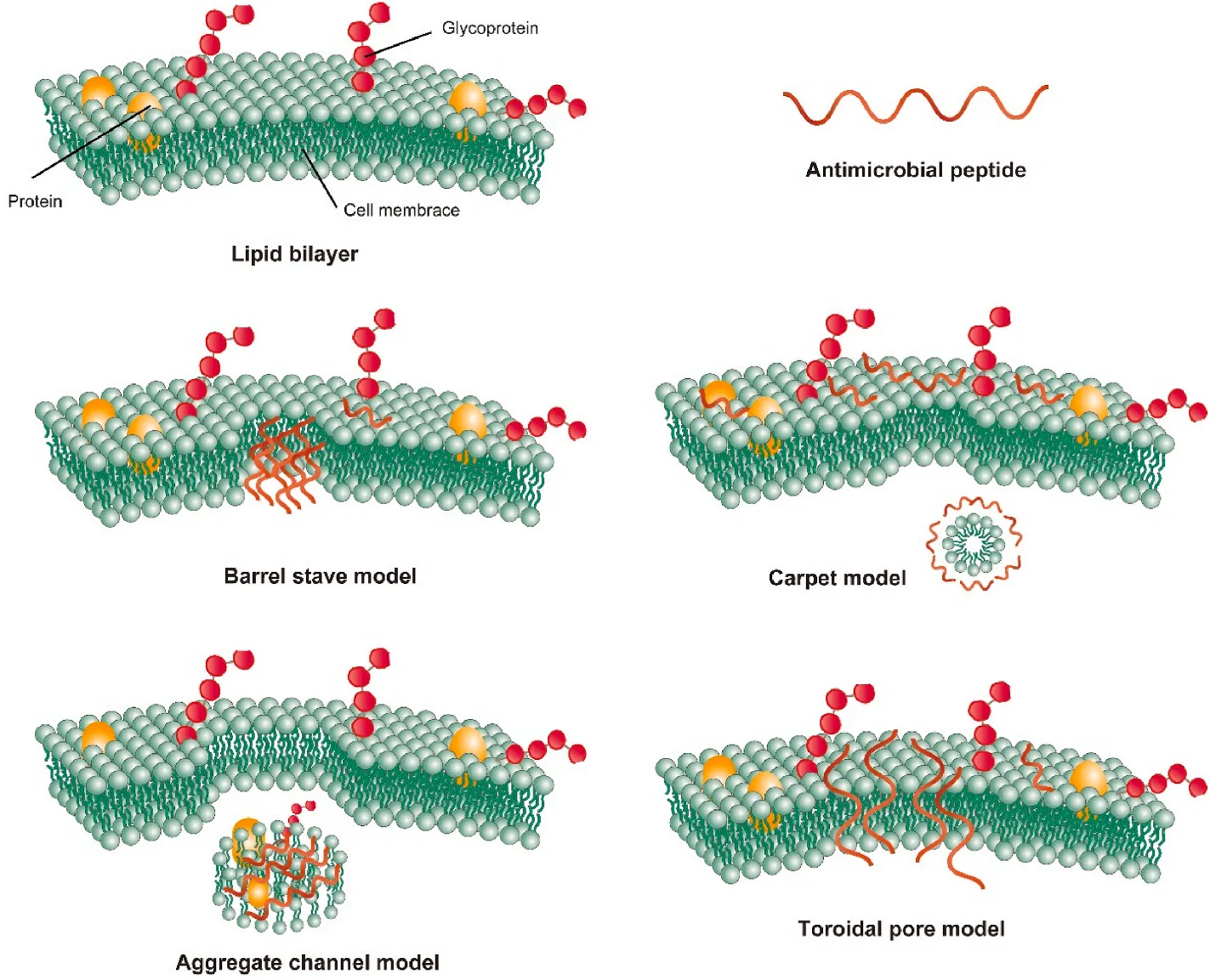
The physicochemical properties of AMPs are critical for their antimicrobial activity. Most AMPs are cationic and amphipathic, allowing them to interact electrostatically with negatively charged bacterial membranes and integrate into the lipid bilayer. This interaction is further influenced by the peptides’ hydrophobicity, charge distribution, and structural conformation. Once bound to the membrane, AMPs can employ various mechanisms to exert their antimicrobial effects, including membrane disruption through the barrel-stave, carpet, and toroidal pore models, as well as intracellular targeting that interferes with nucleic acid and protein synthesis.
Several well-studied AMPs illustrate the breadth of this structural and functional diversity[5]. Defensins, found in mammals and plants, exhibit broad-spectrum activity due to their β-sheet structures stabilized by disulfide bonds. Cathelicidins, such as LL-37, are α-helical peptides known for their immunomodulatory roles and membrane-disruptive capabilities. Protegrins, isolated from porcine leukocytes, combine β-sheet structures with potent antimicrobial action against bacteria and viruses.
This diversity in structure and function enables AMPs to serve as versatile agents against a wide range of pathogens, including multidrug-resistant bacteria. Their ability to target microbial membranes and intracellular processes makes them promising candidates for novel antimicrobial therapies.
Targeting Pathogens: Advanced Mechanisms of Antimicrobial Peptides
Antimicrobial peptides (AMPs) exhibit a broad spectrum of activity against various pathogens through distinct and multifaceted mechanisms. The primary mode of action involves membrane permeabilization, which leads to the disruption of microbial membrane integrity. This disruption can occur through several models, including the barrel-stave, carpet, and toroidal pore models. In the barrel-stave model, AMPs align perpendicularly within the bacterial membrane, forming pore-like structures that compromise membrane integrity. Conversely, the carpet model involves AMPs covering the bacterial membrane surface like a carpet, causing disintegration without forming distinct pores[6]. The toroidal pore model combines features of both, where AMPs induce membrane curvature, leading to the formation of continuous pores lined by both peptides and lipid head groups.
Beyond membrane disruption, AMPs can penetrate bacterial cells and target intracellular components. Once inside the cell, AMPs inhibit the synthesis of vital macromolecules such as DNA, RNA, and proteins. For instance, buforin II is known to cross bacterial membranes and bind to nucleic acids, disrupting genetic replication and transcription processes[7]. Other AMPs, such as Bac5 and Bac7, inhibit protein synthesis and cellular respiration by binding to ribosomal components, leading to ATP depletion and metabolic collapse.
One of the significant advantages of AMPs over traditional antibiotics is their ability to minimize the development of bacterial resistance. Their multimodal action, particularly targeting conserved microbial structures like membranes and essential intracellular pathways, reduces the chances of resistance emergence. Moreover, scientific studies have demonstrated that certain AMPs, such as synthetic cationic peptide D11, can disrupt the proton motive force (PMF) in bacteria, thereby impairing the respiratory chain and generating lethal reactive oxygen species (ROS).
The efficacy of AMPs against multidrug-resistant (MDR) bacteria has been well-documented. Research indicates that AMPs such as magainins, derived from amphibians, exhibit potent activity against gram-negative bacteria by increasing outer membrane permeability and inducing ion leakage. Additionally, peptides like TO17 have shown effectiveness against Staphylococcus aureus by entering cells and degrading genetic material, leading to rapid bacterial death.
These diverse and robust mechanisms highlight AMPs’ potential as next-generation therapeutics in combating antibiotic-resistant infections.
Bridging the Gap: Challenges and Innovations in AMP Clinical Development
The potential of antimicrobial peptides (AMPs) extends far beyond their current applications, offering numerous avenues for future research and therapeutic development. Genetic engineering presents a promising strategy to enhance AMP production by enabling the large-scale synthesis of peptides with optimized structures and enhanced antimicrobial properties. Techniques such as recombinant DNA technology and CRISPR-Cas9 gene editing allow for the modification of native AMPs to improve their stability, potency, and specificity against multidrug-resistant pathogens.
Another exciting direction is the design of synthetic AMPs and peptidomimetics, which aim to replicate or enhance the functional properties of natural peptides while overcoming limitations such as susceptibility to proteolytic degradation. These synthetic analogs can be engineered to increase membrane selectivity, reduce toxicity, and improve pharmacokinetic profiles, making them more viable for clinical use.
Additionally, combining AMPs with conventional antibiotics and nanotechnology holds significant potential for synergistic effects. Nanocarriers, such as liposomes and polymeric nanoparticles, can be utilized to deliver AMPs more effectively, enhancing their stability and targeted delivery while minimizing systemic toxicity. This integration can also reduce the required doses of conventional antibiotics, mitigating the risk of resistance development.
Moreover, ongoing research is expanding the role of AMPs beyond antibacterial applications. Studies have shown promising activity of AMPs against viral, fungal, and parasitic infections, highlighting their versatility as broad-spectrum therapeutics. This emerging evidence underscores the need for continued research to unlock the full therapeutic potential of AMPs in combating infectious diseases globally.
Shaping the Future: Expanding the Therapeutic Potential of AMPs
Antimicrobial peptides (AMPs) represent a revolutionary class of therapeutics with immense potential to address the growing challenge of antibiotic resistance. Their unique mechanisms of action, including membrane disruption and intracellular targeting, make them formidable agents against multidrug-resistant pathogens. Advances in genetic engineering, synthetic peptide design, and nanotechnology are paving the way for the development of more stable, potent, and clinically viable AMP-based therapies.
As research continues to uncover their broad-spectrum capabilities—including efficacy against viral, fungal, and parasitic infections—AMPs are poised to become indispensable tools in modern medicine. Sustained investment in AMP research and development is crucial to fully realize their potential and combat the escalating global health threat posed by antimicrobial resistance.
 LinkPeptide
LinkPeptide