Apraglutide: Revolutionizing Short Bowel Syndrome Treatment with Long-Acting GLP-2 Therapy
Abstract
Apraglutide, a novel glucagon-like peptide-2 (GLP-2) analog, represents a significant advancement in the treatment of short bowel syndrome-associated intestinal failure (SBS-IF). Engineered for once-weekly dosing, Apraglutide offers extended therapeutic effects by enhancing fluid absorption and reducing reliance on parenteral support. Its mechanism of action, targeting the GLP-2 receptor, promotes intestinal growth, increases nutrient absorption, and stabilizes fluid balance. Clinical trials have demonstrated Apraglutide’s safety and efficacy, with patients experiencing increased urine output and reduced parenteral nutrition needs. The longer half-life of Apraglutide positions it as a more patient-friendly alternative to existing GLP-2 analogs, such as teduglutide, which requires daily injections. Ongoing phase 3 trials aim to further explore its long-term benefits and broader applications in gastrointestinal disorders. Apraglutide offers a promising future for improving the quality of life for SBS-IF patients.
Unveiling Apraglutide: A Next-Generation GLP-2 Analog for Gastrointestinal Health
Glucagon-like peptide-2 (GLP-2) plays a vital role in regulating intestinal growth, nutrient absorption, and gastrointestinal homeostasis. Produced by enteroendocrine L-cells in the distal gut, GLP-2 is released postprandially and stimulates intestinal adaptation, promoting mucosal growth, enhancing barrier function, and increasing nutrient and fluid absorption. This has made GLP-2 analogs an essential therapeutic approach for treating intestinal failure and malabsorption disorders, such as short bowel syndrome-associated intestinal failure (SBS-IF)[1].
Apraglutide, a novel long-acting GLP-2 analog, is engineered to provide sustained exposure and extended therapeutic effects compared to its predecessors. Its key advantage is its once-weekly dosing, offering significant improvements in patient compliance and convenience over teduglutide, the currently approved GLP-2 analog that requires daily injections[2]. Apraglutide has shown promising results in increasing fluid absorption and reducing the need for parenteral support in patients with SBS-IF, positioning it as a valuable option for long-term management of this complex condition[3]. This innovation represents a significant advancement in gastrointestinal therapeutics, with ongoing studies further validating its clinical utility.
How Apraglutide Works: A Deeper Look at Its Mechanism and Clinical Impact
Apraglutide, like other GLP-2 analogs, targets the glucagon-like peptide-2 receptor (GLP-2R), primarily located in the gastrointestinal tract. Activation of this receptor stimulates several key physiological processes, including increased intestinal epithelial growth, enhanced barrier function, and improved fluid and nutrient absorption[4]. This is particularly beneficial for patients with short bowel syndrome-associated intestinal failure (SBS-IF), where extensive surgical resection of the small intestine leads to significant malabsorption and dependency on parenteral support for fluid and nutrient intake.
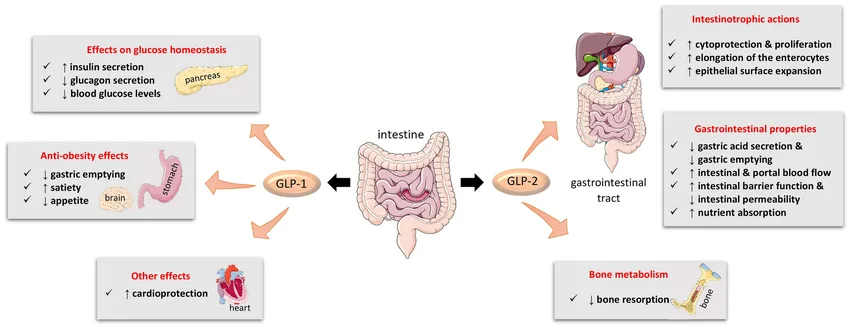
One of Apraglutide’s critical benefits is its longer half-life, enabling less frequent administration compared to its predecessor, teduglutide. While teduglutide requires daily injections due to its relatively short half-life, Apraglutide’s resistance to enzymatic degradation allows for a more convenient once-weekly dosing regimen[5]. Additionally, Apraglutide not only promotes the growth and adaptation of the remaining intestine but also enhances fluid retention by increasing sodium and water absorption, crucial for reducing the need for parenteral nutrition in SBS-IF patients. This advancement in the GLP-2 class offers improved patient compliance and quality of life, marking a significant step forward in the management of SBS-IF.
Efficacy and Safety: Insights from Clinical Trials of Apraglutide
The clinical efficacy and safety of Apraglutide have been evaluated through a randomized, placebo-controlled phase 2 trial focusing on patients with short bowel syndrome-associated intestinal failure (SBS-IF). The trial demonstrated that Apraglutide significantly increased intestinal fluid absorption, as evidenced by increased urine volume output in patients receiving once-weekly doses of 5 mg and 10 mg compared to placebo[6]. This increase in urine volume suggests enhanced fluid absorption, reducing the need for parenteral support, which is crucial for patients suffering from SBS-IF.
Regarding safety, Apraglutide was generally well tolerated, with most treatment-related adverse events (AEs) being mild to moderate in severity. Common AEs included polyuria, decreased stoma output, and edema. Importantly, no serious AEs were considered directly related to Apraglutide treatment. The safety profile of Apraglutide was similar for both the 5 mg and 10 mg doses, indicating that higher doses did not significantly increase the risk of adverse events. Furthermore, the extended half-life of Apraglutide allows for a more patient-friendly, once-weekly dosing regimen, contributing to improved patient compliance and quality of life compared to daily treatments like teduglutide.
Future Prospects: Expanding the Role of Apraglutide in Gastrointestinal Therapy
Apraglutide’s success in improving fluid absorption and reducing the need for parenteral support presents promising opportunities for future research and clinical applications. As a long-acting GLP-2 analog, its once-weekly dosing offers a significant advantage over teduglutide, providing not only clinical benefits but also greater convenience for patients with short bowel syndrome-associated intestinal failure (SBS-IF). This improvement could lead to enhanced patient adherence, fewer treatment interruptions, and a better overall quality of life.
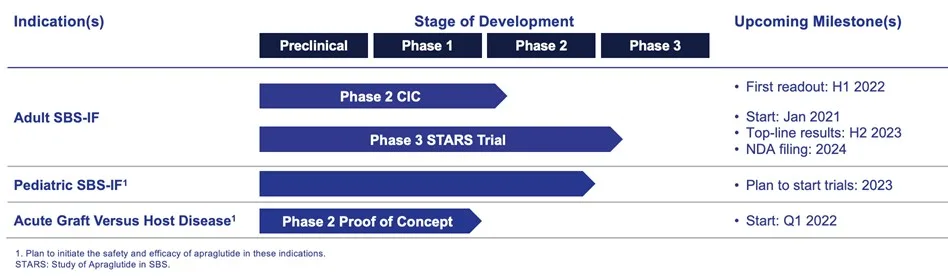
The ongoing phase 3 trials are expected to provide further insights into Apraglutide’s long-term safety, efficacy, and its potential to further reduce parenteral nutrition requirements in a broader patient population. Additionally, future studies may explore the possibility of Apraglutide being used in other gastrointestinal conditions that benefit from enhanced nutrient and fluid absorption, such as Crohn’s disease or radiation enteritis.
Furthermore, Apraglutide’s ability to reduce fluid and electrolyte imbalances, which are major causes of hospitalizations in SBS-IF patients, could translate to a reduction in healthcare costs and improved long-term outcomes[7]. Its unique pharmacokinetic profile sets the stage for expanded applications of GLP-2 analogs in managing chronic gastrointestinal conditions.
Apraglutide’s Promise: Revolutionizing Care for Patients with Short Bowel Syndrome
In conclusion, Apraglutide represents a significant advancement in the treatment of short bowel syndrome-associated intestinal failure (SBS-IF). Its long-acting, once-weekly dosing provides a more patient-friendly alternative to the currently available daily treatment, teduglutide. By improving intestinal fluid absorption and reducing the reliance on parenteral support, Apraglutide not only enhances patient outcomes but also improves quality of life through easier management and increased compliance.
The ongoing phase 3 trials will further solidify Apraglutide’s place in the therapeutic landscape for SBS-IF and potentially broaden its application to other conditions requiring improved gastrointestinal absorption. Its robust safety profile, combined with its efficacy in increasing urine volume output and sodium absorption, offers promising long-term benefits for patients. This innovation in GLP-2 analog therapy marks a pivotal step forward in gastrointestinal care, positioning Apraglutide as a crucial tool in managing SBS-IF and possibly other related disorders.
As the field of gastrointestinal therapeutics continues to evolve, Apraglutide’s development highlights the potential of peptide-based therapies to address complex conditions like SBS-IF, offering new hope for patients and clinicians alike.
Reference
- Jeppesen, P. B. (2014). Spectrum of short bowel syndrome in adults: intestinal insufficiency to intestinal failure. Journal of Parenteral and Enteral Nutrition, 38, 8S-13S.
- Wiśniewski, K., Sueiras-Diaz, J., Jiang, G., Galyean, R., Lu, M., Thompson, D., … & Schteingart, C. D. (2016). Synthesis and pharmacological characterization of novel glucagon-like peptide-2 (GLP-2) analogues with low systemic clearance. Journal of Medicinal Chemistry, 59(7), 3129-3139.
- Hargrove, D. M., Alagarsamy, S., Croston, G., Laporte, R., Qi, S., Srinivasan, K., … & Dimitriadou, V. (2020). Pharmacological characterization of apraglutide, a novel long-acting peptidic glucagon-like peptide-2 agonist, for the treatment of short bowel syndrome. Journal of Pharmacology and Experimental Therapeutics, 373(2), 193-203.
- Jeppesen, P. B., Hartmann, B., Hansen, B. S., Thulesen, J., Holst, J. J., & Mortensen, P. B. (1999). Impaired meal stimulated glucagon-like peptide 2 response in ileal resected short bowel patients with intestinal failure. Gut, 45(4), 559-563.
- Marier, J. F., Beliveau, M., Mouksassi, M. S., Shaw, P., Cyran, J., Kesavan, J., … & Caminis, J. (2008). Pharmacokinetics, safety, and tolerability of teduglutide, a glucagon‐like peptide‐2 (GLP‐2) analog, following multiple ascending subcutaneous administrations in healthy subjects. The Journal of Clinical Pharmacology, 48(11), 1289-1299.
- Eliasson, J., Hvistendahl, M. K., Freund, N., Bolognani, F., Meyer, C., & Jeppesen, P. B. (2022). Apraglutide, a novel glucagon‐like peptide‐2 analog, improves fluid absorption in patients with short bowel syndrome intestinal failure: findings from a placebo‐controlled, randomized phase 2 trial. Journal of Parenteral and Enteral Nutrition, 46(4), 896-904.
- Fuglsang, K. A., Brandt, C. F., Scheike, T., & Jeppesen, P. B. (2020). Hospitalizations in patients with nonmalignant short‐bowel syndrome receiving home parenteral support. Nutrition in Clinical Practice, 35(5), 894-902.
 LinkPeptide
LinkPeptide