Motixafortide: A Breakthrough in Stem Cell Mobilization and Cancer Therapy
Abstract
Motixafortide is a selective CXCR4 antagonist that has recently gained FDA approval for its use in combination with G-CSF to mobilize hematopoietic stem cells (HSCs) in patients with multiple myeloma. This novel agent disrupts the CXCL12/CXCR4 axis, promoting the release of HSCs from the bone marrow into peripheral blood, facilitating autologous stem cell transplantation. In addition to its role in stem cell mobilization, Motixafortide has demonstrated potential in cancer treatment, including pancreatic cancer and acute myeloid leukemia (AML), by inhibiting cancer cell migration, proliferation, and survival. Clinical trials have shown that Motixafortide, in combination with chemotherapy and immunotherapy, can enhance therapeutic outcomes in challenging malignancies. Ongoing research is expanding its application in gene therapy and immunotherapy, positioning Motixafortide as a promising tool in both oncology and regenerative medicine.
Motixafortide: Advancing Stem Cell Mobilization and Cancer Treatment
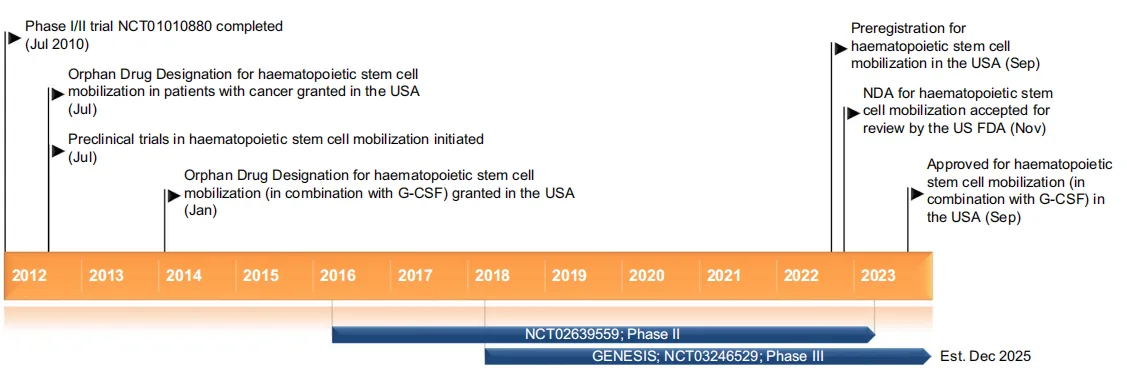
Motixafortide is a novel therapeutic agent that acts as a selective C-X-C motif chemokine receptor 4 (CXCR4) antagonist. It has gained significant attention for its role in the mobilization of hematopoietic stem cells (HSCs) and in cancer treatment, particularly for multiple myeloma and other malignancies. In September 2023, the U.S. Food and Drug Administration (FDA) approved Motixafortide for use in combination with filgrastim (G-CSF) to mobilize HSCs for autologous transplantation in patients with multiple myeloma[1]. This breakthrough highlights the clinical importance of effectively mobilizing CD34+ HSCs, a key component of successful stem cell transplantation.

Beyond its role in multiple myeloma, Motixafortide has shown promise in treating other cancers, including pancreatic cancer and acute myeloid leukemia (AML)[2]. Its ability to disrupt the CXCL12/CXCR4 axis, which plays a crucial role in cancer cell migration, proliferation, and survival, positions Motixafortide as a valuable therapeutic in oncology[3]. The approval of Motixafortide and its ongoing research mark a significant advancement in both cancer treatment and stem cell mobilization, offering new hope for patients with challenging diseases.
Targeting the CXCL12/CXCR4 Axis in Disease Pathology
Motixafortide operates as a highly selective antagonist of the C-X-C motif chemokine receptor 4 (CXCR4), a receptor critical for the retention of hematopoietic stem cells (HSCs) within the bone marrow. The drug works by disrupting the interaction between CXCR4 and its ligand, CXCL12 (also known as stromal cell-derived factor 1, or SDF-1), which is essential for HSC retention. By inhibiting this interaction, Motixafortide promotes the release of HSCs from the bone marrow into peripheral blood, facilitating their collection for transplantation purposes.
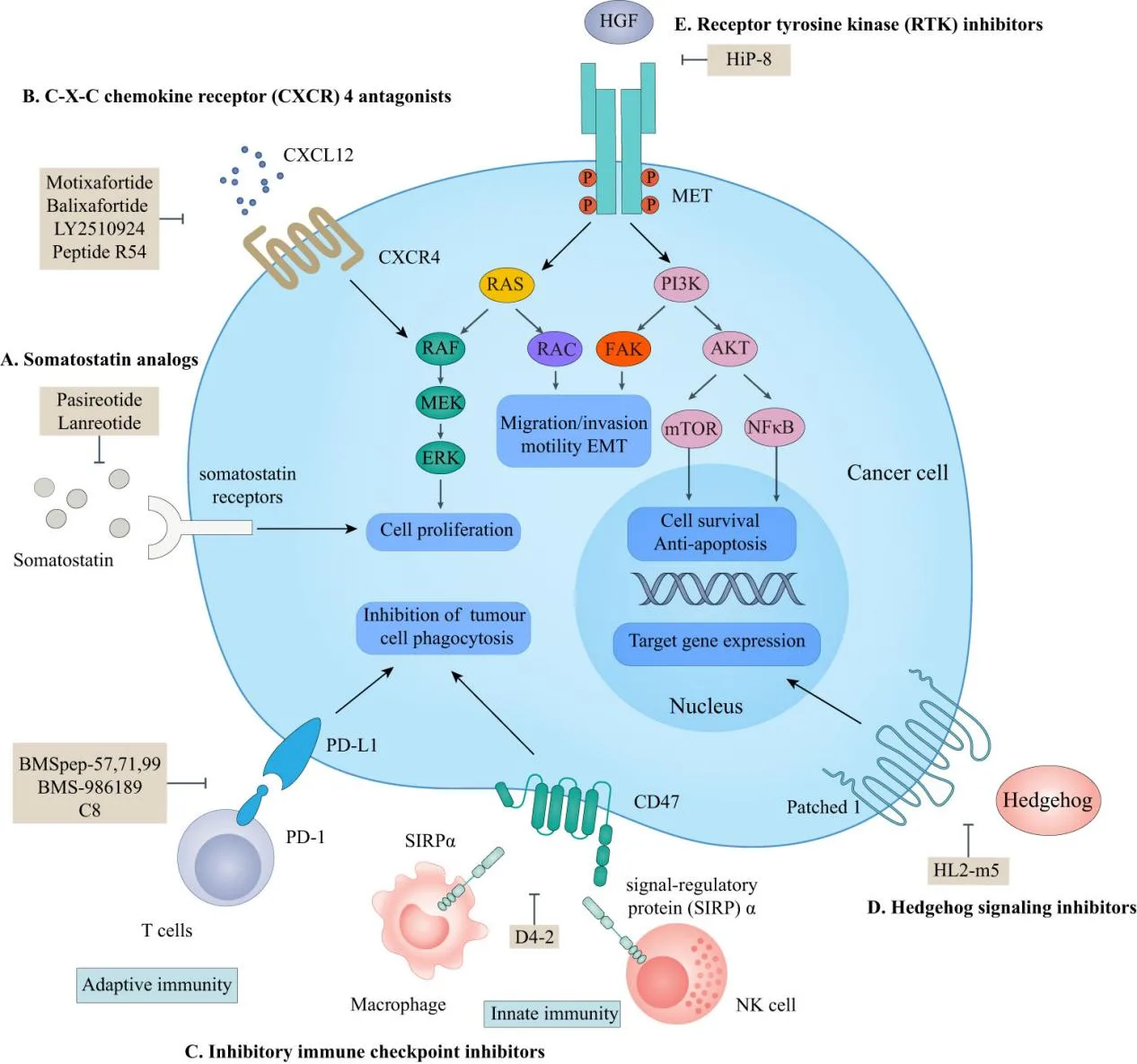
In cancer treatment, this same mechanism proves beneficial in halting cancer cell migration, proliferation, and survival. The CXCL12/CXCR4 axis is overexpressed in various malignancies, contributing to tumor growth and metastasis. Motixafortide blocks this pathway, preventing cancer cells from utilizing the chemokine signaling to evade immune responses and metastasize to distant organs[4]. Pharmacodynamic studies have shown that Motixafortide has a high binding affinity to CXCR4 (IC50 of 0.42–4.5 nmol/L) and an extended receptor occupancy time, with effects lasting over 72 hours after administration[5]. This prolonged activity makes it an effective agent for both short-term stem cell mobilization and long-term cancer treatment strategies.
Motixafortide’s Role in Stem Cell Transplantation and Oncology
Motixafortide has demonstrated significant clinical efficacy across several therapeutic areas, particularly in the mobilization of hematopoietic stem cells (HSCs) for autologous stem cell transplantation and cancer treatment. One of the most prominent uses of Motixafortide is in combination with granulocyte-colony stimulating factor (G-CSF) to mobilize HSCs in patients with multiple myeloma. In a phase III clinical trial (GENESIS), 92.5% of patients treated with Motixafortide plus G-CSF achieved the optimal target of ≥ 6 × 106 CD34+ cells/kg within two apheresis sessions, compared to only 26.2% of patients treated with G-CSF alone. This success reduces the need for additional apheresis procedures, thereby lowering treatment costs and minimizing patient discomfort.
Beyond its role in stem cell mobilization, Motixafortide has also shown promise in cancer therapy, particularly in the treatment of pancreatic cancer and acute myeloid leukemia (AML). In the COMBAT trial, Motixafortide combined with pembrolizumab (an immune checkpoint inhibitor) and chemotherapy demonstrated clinical efficacy in patients with metastatic pancreatic ductal adenocarcinoma (PDAC), with an objective response rate of 21.1% in patients receiving second-line therapy[6]. Additionally, in AML, Motixafortide has been evaluated for its ability to enhance the effects of chemotherapy, contributing to the mobilization of leukemia cells from the bone marrow and improving clinical outcomes in patients with relapsed or refractory AML.
A Comprehensive Overview of Motixafortide’s Efficacy and Tolerability
Motixafortide exhibits pharmacokinetic properties that support its effectiveness in both healthy individuals and patients with multiple myeloma, acute myeloid leukemia (AML), and other cancers. After subcutaneous administration, Motixafortide reaches peak plasma concentration within 0.25 to 1.17 hours, with an effective plasma half-life of approximately two hours. This rapid absorption allows for timely mobilization of hematopoietic stem cells (HSCs), which is crucial in the autologous stem cell transplantation process. Motixafortide is highly bound to plasma proteins (greater than 99%), and it is primarily metabolized through nonspecific degradation pathways into smaller peptides and amino acids[7]. Minimal accumulation is observed following repeated doses, and its pharmacokinetics are not significantly altered by mild to moderate renal or hepatic impairment.
The safety profile of Motixafortide has been well-documented in clinical trials. Common adverse events include local injection-site reactions, such as pain, erythema, and pruritus, which are generally mild and transient. Systemic reactions like flushing and urticaria have also been observed but are typically self-limiting. In studies involving multiple myeloma patients, no grade 4 adverse events were reported, and grade 3 events occurred in only 27.5% of patients, suggesting a favorable safety profile. Furthermore, Motixafortide demonstrated a low potential for drug-drug interactions, making it suitable for combination therapies in cancer treatment.
Expanding the Clinical Horizons of Motixafortide in Medicine
Motixafortide’s clinical success in hematopoietic stem cell (HSC) mobilization and cancer treatment has opened the door to further research and broader therapeutic applications. Ongoing clinical trials are investigating its potential in various cancers and gene therapy approaches. One key area of research is Motixafortide’s role in gene therapy for sickle cell disease, where it is being studied for its ability to mobilize CD34+ HSCs, enabling successful genetic modifications to treat this genetic disorder. This marks an exciting advancement in regenerative medicine, where HSC mobilization plays a pivotal role in curative therapies.
In cancer treatment, clinical studies are exploring the potential of Motixafortide in combination with immunotherapies. Specifically, trials are evaluating its efficacy alongside immune checkpoint inhibitors, such as pembrolizumab, in solid tumors like pancreatic cancer. The goal is to enhance the anti-tumor response by targeting the CXCL12/CXCR4 axis, which often enables tumors to evade immune detection and therapy. Additionally, Motixafortide is being studied in combination with chemotherapy, aiming to improve outcomes in difficult-to-treat cancers like metastatic pancreatic ductal adenocarcinoma.
As these trials progress, Motixafortide may emerge as a cornerstone of both cancer immunotherapy and regenerative medicine, providing hope for patients with limited treatment options. The continued investigation of this CXCR4 antagonist will likely reveal new insights into its versatility and potential for broader clinical application.
 LinkPeptide
LinkPeptide