Murepavadin: A Breakthrough in Combatting Pseudomonas aeruginosa and Antibiotic Resistance Through Dual Mechanisms
Abstract
Murepavadin represents an innovative approach in the fight against multi-drug-resistant Pseudomonas aeruginosa by combining direct antibacterial activity with immunomodulatory effects. This small-molecule mimetic of host defense peptides (HDPs) selectively targets the LptD protein in the outer membrane of P. aeruginosa, disrupting its structure and inhibiting growth. Additionally, Murepavadin activates the MRGPRX2 receptor in human mast cells, triggering immune responses that enhance bacterial clearance and promote wound healing. Its dual mechanism makes Murepavadin uniquely suited to address the limitations of traditional antibiotics. However, clinical development has faced challenges, including renal toxicity with intravenous formulations. Research into alternative administration methods, particularly inhaled formulations, is underway to improve safety and expand its applications, especially for pulmonary infections in cystic fibrosis. This paper explores Murepavadin’s mechanisms, therapeutic applications, and potential future directions in infection management.
Revolutionizing Antibacterial Treatment: Murepavadin’s Role in Tackling Antibiotic Resistance
Murepavadin is a novel small-molecule antimicrobial peptide mimetic specifically targeting Pseudomonas aeruginosa, a common cause of hospital-acquired infections with significant antibiotic resistance. Designed to mimic host defense peptides (HDPs), Murepavadin offers a unique approach by binding to the outer membrane protein LptD on P. aeruginosa, disrupting its cellular function without impacting non-target bacterial species[1]. The rise of multi-drug-resistant pathogens such as P. aeruginosa has intensified the demand for alternatives to traditional antibiotics, which are often ineffective against these resilient organisms[2].
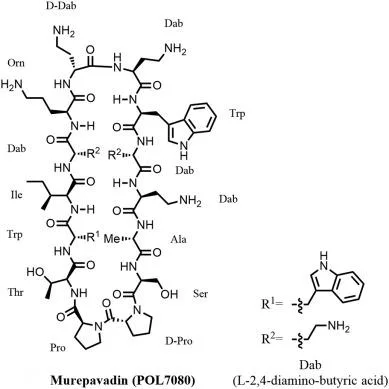
Murepavadin’s selectivity, coupled with its immunomodulatory capabilities, makes it a promising candidate in combating antibiotic resistance. Unlike standard antibiotics, Murepavadin engages immune responses via the MRGPRX2 receptor in human mast cells, leading to enhanced immune cell recruitment and activation at infection sites[3]. This dual mechanism not only supports direct bacterial elimination but also enhances wound healing, positioning Murepavadin as an innovative option for managing resistant infections. These combined features highlight the significance of Murepavadin in advancing next-generation antimicrobial therapies against persistent hospital-based infections.
How Murepavadin Targets P. aeruginosa: A Dual Mechanism of Action
Murepavadin operates as a host defense peptide (HDP) mimetic, targeting Pseudomonas aeruginosa by binding to the essential outer membrane protein LptD, a component involved in lipopolysaccharide transport, which disrupts the bacterial membrane integrity and leads to cell death[4]. This targeted approach makes Murepavadin distinct from conventional antibiotics, as it minimizes off-target effects on non-pathogenic bacteria. In addition to its direct antimicrobial activity, Murepavadin activates the Mas-related G protein-coupled receptor X2 (MRGPRX2) in human mast cells, initiating a cascade that includes mast cell degranulation and the release of pro-inflammatory cytokines[5].
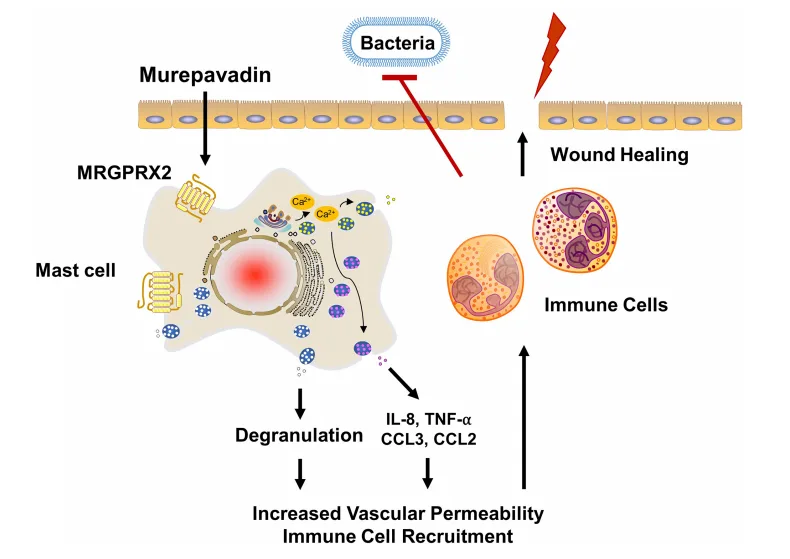
This receptor-mediated activation not only enhances the recruitment of immune cells, such as neutrophils and monocytes, but also amplifies local immune responses, creating a more effective barrier against bacterial proliferation. This dual mechanism—direct bacterial targeting and immune modulation—supports Murepavadin’s potential as a powerful treatment for multi-drug-resistant infections. Studies have shown that its immunomodulatory effects may also promote wound healing by increasing vascular permeability and cell migration to infection sites, a critical factor in treating severe bacterial infections.
Beyond Antibacterial Action: Murepavadin’s Immunomodulatory Benefits in Host Defense
Murepavadin’s immunomodulatory effects are pivotal in enhancing host defenses against Pseudomonas aeruginosa. By activating the MRGPRX2 receptor on human mast cells, Murepavadin induces a series of immune responses that include degranulation and the release of pro-inflammatory cytokines like IL-8 and CCL3[6]. These mediators contribute to immune cell recruitment, drawing neutrophils and other phagocytic cells to the infection site for more robust bacterial clearance. Unlike traditional antibiotics, which often rely solely on direct bacterial inhibition, Murepavadin’s ability to amplify immune activity represents a novel approach in infectious disease therapy[7]. This activation not only supports bacterial elimination but also increases local vascular permeability, promoting wound healing in infected tissues. The absence of receptor desensitization or internalization, typically induced by other agonists, allows Murepavadin to sustain its immune-modulating effects, highlighting its potential as a therapeutic agent that leverages both direct antimicrobial and host defense mechanisms. Furthermore, studies suggest that these properties could extend to broader applications in treating infections caused by other pathogens that exploit immune evasion.
Clinical Applications and Potential of Murepavadin in Combating P. aeruginosa Infections
Murepavadin holds significant promise for treating infections caused by multi-drug-resistant Pseudomonas aeruginosa, a pathogen notorious for biofilm formation and high resistance levels. Its unique mechanism, targeting the LptD protein in the outer membrane, makes it highly selective for P. aeruginosa, which is advantageous for reducing collateral damage to non-pathogenic bacteria[8]. Current applications of Murepavadin primarily focus on severe infections, particularly those acquired in healthcare settings, where P. aeruginosa can cause skin, lung, and systemic infections. The potential use of Murepavadin as an inhaled formulation for cystic fibrosis patients is especially compelling, as it could offer targeted delivery to the lungs, reducing systemic exposure and possibly minimizing adverse effects[9].
Additionally, Murepavadin’s immunomodulatory activation of mast cells amplifies local immune defenses, which can be critical in wound healing and tissue repair. Studies show that Murepavadin promotes immune cell recruitment and cytokine release at infection sites, thereby facilitating bacterial clearance and supporting tissue regeneration. This dual action not only counters the pathogen’s resistance mechanisms but also offers a comprehensive approach to infection management by integrating direct antimicrobial effects with immune system support.
Future Horizons and Overcoming Challenges in Murepavadin’s Clinical Development
Despite its therapeutic potential, Murepavadin faces challenges in clinical development. Intravenous formulations have shown promise against severe P. aeruginosa infections, but adverse effects such as kidney toxicity have led to temporary halts in clinical trials. To overcome this, researchers are exploring alternative administration routes, including an inhaled formulation, which could offer targeted delivery to the lungs, making it particularly beneficial for conditions like cystic fibrosis where pulmonary infections are prevalent. This approach may reduce systemic exposure and mitigate renal side effects, enhancing patient safety.
Another promising direction is the combination of Murepavadin with other therapies to combat biofilm-associated infections more effectively, particularly in multi-drug-resistant cases. Continued optimization of its structure and dosing could also improve its pharmacokinetic profile, allowing for more sustained and targeted action against P. aeruginosa. Additionally, research into Murepavadin’s immunomodulatory effects opens the door to broader applications in treating infections that require a strong host defense response. As new studies emerge, the potential to refine Murepavadin for safer, more effective treatments against resistant bacterial infections grows, positioning it as a critical tool in next-generation antibiotic strategies.
Reference
- Amponnawarat, A., Chompunud Na Ayudhya, C., & Ali, H. (2021). Murepavadin, a small molecule host defense peptide mimetic, activates Mast cells via MRGPRX2 and MrgprB2. Frontiers in Immunology, 12, 689410.
- Martin-Loeches, I., Dale, G. E., & Torres, A. (2018). Murepavadin: a new antibiotic class in the pipeline. Expert review of anti-infective therapy, 16(4), 259-268.
- Lister, P. D., Wolter, D. J., & Hanson, N. D. (2009). Antibacterial-resistant Pseudomonas aeruginosa: clinical impact and complex regulation of chromosomally encoded resistance mechanisms. Clinical microbiology reviews, 22(4), 582-610.
- Srinivas, N., Jetter, P., Ueberbacher, B. J., Werneburg, M., Zerbe, K., Steinmann, J., … & Robinson, J. A. (2010). Peptidomimetic antibiotics target outer-membrane biogenesis in Pseudomonas aeruginosa. Science, 327(5968), 1010-1013.
- Subramanian, H., Gupta, K., Guo, Q., Price, R., & Ali, H. (2011). Mas-related gene X2 (MrgX2) is a novel G protein-coupled receptor for the antimicrobial peptide LL-37 in human mast cells: resistance to receptor phosphorylation, desensitization, and internalization. Journal of Biological Chemistry, 286(52), 44739-44749.
- Subramanian, H., Gupta, K., Lee, D., Bayir, A. K., Ahn, H., & Ali, H. (2013). β-Defensins activate human mast cells via Mas-related gene X2. The Journal of Immunology, 191(1), 345-352.
- Scott, R., & N. Tew, G. (2017). Mimics of host defense proteins; strategies for translation to therapeutic applications. Current Topics in Medicinal Chemistry, 17(5), 576-589.
- Díez-Aguilar, M., Hernández-García, M., Morosini, M. I., Fluit, A., Tunney, M. M., Huertas, N., … & Cantón, R. (2021). Murepavadin antimicrobial activity against and resistance development in cystic fibrosis Pseudomonas aeruginosa isolates. Journal of Antimicrobial Chemotherapy, 76(4), 984-992.
- Wach, A., Dembowsky, K., & Dale, G. E. (2018). Pharmacokinetics and safety of intravenous murepavadin infusion in healthy adult subjects administered single and multiple ascending doses. Antimicrobial Agents and Chemotherapy, 62(4), 10-1128.
 LinkPeptide
LinkPeptide