Peceleganan: A New Era in Antimicrobial Peptide Therapy for Resistant Wound Infections
Abstract
Peceleganan (PL-5) is a novel antimicrobial peptide (AMP) developed specifically to address wound infections, particularly those complicated by antibiotic-resistant bacteria. This blog explores Peceleganan’s unique mechanism of action, which disrupts bacterial membranes through electrostatic interactions, leading to rapid bacterial death with minimal risk of resistance development. Clinical trials have demonstrated Peceleganan’s superior efficacy over traditional treatments, such as silver sulfadiazine, in reducing infection symptoms and clearing bacteria from wound sites. The 2‰ concentration has shown optimal efficacy, balancing safety with high bacterial clearance. Peceleganan’s localized activity minimizes systemic exposure, reducing toxicity risks, and presents a promising alternative for infection management. Future research is expected to expand its applications to more complex and resistant infections, offering a safe, potent solution in wound care.
Introduction to Peceleganan: Addressing the Challenge of Resistant Wound Infections
Wound infections are a major challenge in healthcare, often resulting in delayed healing and increased morbidity. The global rise in antimicrobial resistance has further complicated treatment options, driving the need for novel solutions to combat infection without contributing to resistance[1]. Antimicrobial peptides (AMPs) have emerged as promising alternatives, offering broad-spectrum activity against bacteria, fungi, and even viruses. Unlike traditional antibiotics, AMPs disrupt bacterial cell membranes in ways that reduce the likelihood of resistance development[2].
Peceleganan (PL-5) is an innovative AMP developed specifically for the topical treatment of skin wound infections. This α-helical AMP contains 26 amino acids, offering a robust and selective mechanism that targets bacterial membranes through electrostatic interactions[3]. As the first topical AMP spray designed for wound applications, Peceleganan offers advantages in treating infections caused by antibiotic-resistant bacteria, providing an essential tool in modern wound care. By focusing on bacterial membrane disruption, Peceleganan addresses a crucial gap in managing wound infections and shows promise in reducing the global burden of antimicrobial resistance.
How Peceleganan Targets Bacterial Membranes
Peceleganan’s unique mechanism of action distinguishes it from traditional antibiotics. This antimicrobial peptide (AMP) functions primarily through a “carpet-like” mechanism, binding to bacterial membranes via electrostatic interactions and disrupting membrane integrity, which leads to rapid bacterial death[4]. Its α-helical structure is particularly well-suited for this role, allowing it to insert itself into the lipid bilayer of bacterial cells. This action causes bacterial lysis without affecting mammalian cells, providing high selectivity and low toxicity.
This mode of action is significant in the battle against antibiotic resistance. Unlike many antibiotics, which target specific bacterial processes and can eventually lead to resistant strains, Peceleganan operates on a structural level, which reduces the likelihood of resistance development. Additionally, studies have shown that Peceleganan is effective against both Gram-positive and Gram-negative bacteria, including drug-resistant strains, making it a versatile option for infection control[5]. As a topical agent, Peceleganan offers a localized approach to wound infections, preserving systemic antibiotic effectiveness and minimizing potential adverse reactions associated with other antimicrobial treatments.
Evaluating Peceleganan’s Efficacy in Wound Care
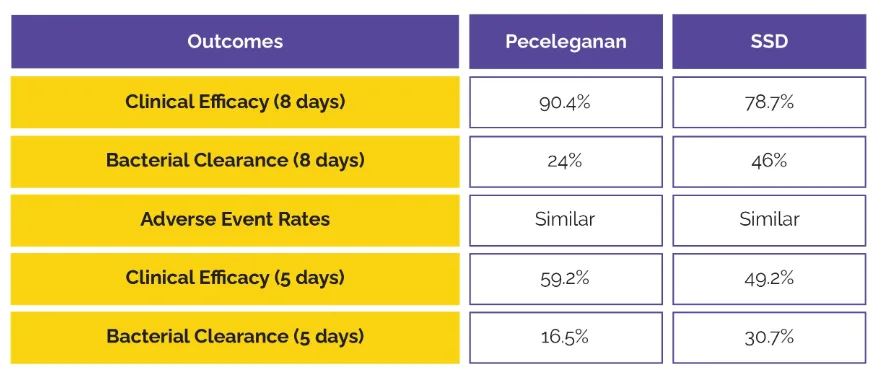
Peceleganan’s efficacy in treating wound infections has been demonstrated through a Phase IIb clinical trial, highlighting its potential as a powerful topical antimicrobial treatment. The trial, conducted across multiple centers, compared Peceleganan to silver sulfadiazine (SSD), a commonly used topical agent, in patients with secondary open wound infections. Peceleganan was tested in three concentrations (1‰, 2‰, and 4‰), and efficacy was evaluated on multiple clinical and microbiological parameters, including infection reduction and bacterial clearance.
Results indicated that Peceleganan was significantly more effective than SSD in reducing symptoms such as exudate, erythema, and inflammation by the eighth day of treatment. Each concentration of Peceleganan demonstrated high efficacy rates, with the 2‰ concentration showing the optimal balance between clinical improvement and bacterial clearance. Furthermore, Peceleganan was effective against both Gram-positive and Gram-negative bacteria, including drug-resistant strains, positioning it as a versatile option for wound infection treatment[6]. These findings suggest that Peceleganan’s broad-spectrum antimicrobial activity, combined with its safety profile, could make it a crucial tool in managing challenging wound infections in clinical settings.
Optimizing Peceleganan for Maximum Benefit
The safety of Peceleganan was a key focus in its clinical trials, especially given its application directly on wound sites. Across all test groups, Peceleganan showed an excellent safety profile, with no severe adverse events attributed to its use. Importantly, pharmacokinetic analyses revealed that Peceleganan did not enter the bloodstream at detectable levels, even at its highest concentration (4‰), suggesting minimal systemic absorption and reduced risk of systemic toxicity. This localized activity makes Peceleganan particularly suitable for treating wound infections without the risks associated with systemic antibiotics, such as adverse drug reactions and impacts on the microbiome[7].
The clinical trials also explored the optimal concentration for maximizing efficacy while maintaining safety. The 2‰ concentration emerged as the most effective, combining high bacterial clearance rates with robust clinical outcomes, such as reduced infection-related symptoms and improved wound healing. Higher concentrations (4‰) did not show a significant increase in efficacy, likely due to structural changes in Peceleganan at higher doses that may reduce its membrane-disrupting activity[8]. These findings support the use of Peceleganan at 2‰ concentration as the recommended dosage for clinical applications, offering a safe, efficient, and well-tolerated option for wound care.
Expanding Peceleganan’s Role in Advanced Wound Care
Peceleganan’s demonstrated efficacy and safety profile position it as a promising addition to wound care, particularly in cases involving antibiotic-resistant infections. With antibiotic resistance becoming a critical public health challenge, Peceleganan provides a much-needed alternative for local treatment that minimizes systemic antibiotic exposure. Moving forward, upcoming Phase III clinical trials are expected to assess its efficacy in more diverse and severe wound types, such as surgical site infections and large-scale trauma wounds. These studies will help establish Peceleganan’s broader clinical utility and potentially expand its indications to more complex wound management scenarios.
Beyond traditional wound infections, Peceleganan’s versatility could extend to specialized applications, such as treating chronic wounds in immunocompromised patients or in healthcare settings where the risk of multidrug-resistant bacterial colonization is high. Future research may also explore combination therapies with other antimicrobial agents to enhance efficacy. As Peceleganan progresses through clinical trials and regulatory approvals, it could set a new standard for antimicrobial peptide-based treatments in wound care, offering a safer, resistance-proof option for infection control and wound management.
 LinkPeptide
LinkPeptide