Taspoglutide: Advancements, Challenges, and Insights in GLP-1 Receptor Agonist Therapy for Type 2 Diabetes
Abstract
Taspoglutide, a once-weekly glucagon-like peptide-1 receptor agonist (GLP-1 RA), was developed to improve glycemic control and reduce weight in patients with type 2 diabetes. With unique amino acid substitutions that enhance stability and resistance to enzymatic degradation, Taspoglutide demonstrated promising efficacy in early clinical trials, significantly lowering HbA1c levels and achieving comparable weight loss to existing GLP-1 RAs. However, its development faced considerable challenges due to high rates of gastrointestinal side effects, injection site reactions, and the formation of anti-drug antibodies, leading to increased dropout rates and adverse immune responses. These hurdles ultimately halted Taspoglutide’s progression to market. Despite this, the insights gained from its development have contributed to refining GLP-1 RA formulations, underscoring the importance of balancing efficacy with safety in long-acting peptide therapies. Taspoglutide’s story offers valuable lessons for future advancements in diabetes management.
Taspoglutide: A Novel GLP-1 Receptor Agonist for Type 2 Diabetes
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) have emerged as a cornerstone in the treatment of type 2 diabetes, offering unique mechanisms that aid in glucose control and weight management. GLP-1 RAs work primarily by enhancing glucose-dependent insulin secretion, inhibiting glucagon release, slowing gastric emptying, and increasing satiety, thereby addressing both hyperglycemia and obesity in diabetic patients [1,2]. Traditionally, GLP-1 RAs required frequent injections, but advances in pharmaceutical development have led to formulations with longer durations of action, enhancing patient adherence and convenience.
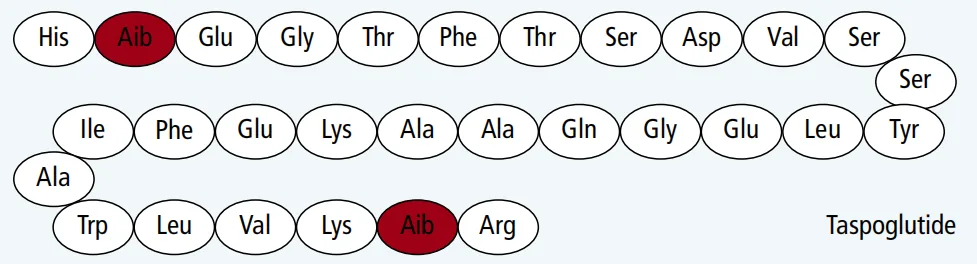
Taspoglutide was developed as a once-weekly GLP-1 RA aimed at reducing injection frequency while maintaining therapeutic efficacy. Structurally, it is a modified version of the native GLP-1 hormone with amino acid substitutions that protect it from degradation by dipeptidyl peptidase-4 (DPP-4) and other plasma proteases. These substitutions, particularly at positions 8 and 35, allow Taspoglutide to maintain a stable pharmacokinetic profile, making it more effective for sustained glucose control [3]. Unlike earlier GLP-1 RAs, Taspoglutide also incorporates a sustained-release formulation achieved by adding zinc chloride, allowing steady plasma levels over two weeks after a single subcutaneous injection. This innovation highlights the evolving design of GLP-1 RAs to improve the lives of patients with type 2 diabetes.
Mechanism of Action and Pharmacology: Structural Innovations in Taspoglutide
Taspoglutide’s unique pharmacology and mechanism of action stem from its structural modifications and formulation designed to resist enzymatic degradation, offering extended stability and activity in the bloodstream. As a 30-amino-acid derivative of native GLP-1, Taspoglutide incorporates specific amino acid substitutions that prevent rapid breakdown. In particular, the substitution of alanine with aminoisobutyric acid at position 8 protects it from dipeptidyl-peptidase-4 (DPP-4), an enzyme that would typically degrade GLP-1 analogues. Another substitution at position 35, where glycine is replaced by aminoisobutyric acid, provides further resistance to other proteases such as plasmin and kallikrein [4].
This structural modification enables Taspoglutide to bind effectively to the GLP-1 receptor, activating it with high potency and maintaining therapeutic levels over time. To enhance its pharmacokinetic profile, Taspoglutide is formulated with zinc chloride, creating a sustained-release system. After subcutaneous injection, zinc chloride prolongs Taspoglutide’s release, allowing a steady plasma concentration to be achieved approximately 24 hours post-injection, with effects lasting over two weeks. This combination of molecular stability and sustained-release technology makes Taspoglutide a potent GLP-1 RA candidate, although challenges with side effects ultimately limited its development.
Clinical Trials and Efficacy: Insights from the T-emerge Program
The clinical efficacy of Taspoglutide was rigorously evaluated in the T-emerge trial program, which focused on its potential benefits for glycemic control and weight reduction in patients with type 2 diabetes. T-emerge 1, a 24-week, randomized, placebo-controlled trial, enrolled 373 patients who had not previously received diabetes medications. In this study, Taspoglutide demonstrated significant glycemic control benefits compared to placebo, with HbA1c reductions of -1.01% for the 10 mg dose and -1.18% for the 20 mg dose, whereas the placebo group only saw a -0.09% change. Weight loss was another positive outcome, with patients on Taspoglutide losing an average of -1.45 kg (10 mg) and -2.25 kg (20 mg), compared to -1.23 kg in the placebo group [3,4].
Further validating its efficacy, the T-emerge 2 trial compared Taspoglutide directly with exenatide, a well-established twice-daily GLP-1 receptor agonist. This study involved 1,189 overweight patients with type 2 diabetes whose HbA1c levels were inadequately controlled with metformin, with or without a thiazolidinedione. Taspoglutide demonstrated statistically superior HbA1c reductions over exenatide, with HbA1c reductions of -1.24% for the 10 mg dose and -1.31% for the 20 mg dose, compared to -0.98% with exenatide. In terms of weight loss, both Taspoglutide doses achieved results comparable to exenatide, with reductions of -1.6 kg (10 mg) and -2.3 kg (20 mg), highlighting Taspoglutide’s potential as an effective, once-weekly GLP-1 RA option.
These trials underscored Taspoglutide’s efficacy in HbA1c control and weight reduction, marking it as a promising therapeutic candidate for patients needing more manageable dosing schedules and effective glycemic control.
Adverse Events and Developmental Challenges: The Safety Profile of Taspoglutide
The T-emerge trials, while showcasing Taspoglutide’s efficacy, also highlighted significant side effects and challenges that ultimately hindered its development. Adverse events were prevalent across multiple studies, with gastrointestinal (GI) issues being among the most common. In the T-emerge 1 trial, for example, 53% of patients on the 10 mg dose and 59% on the 20 mg dose experienced nausea, compared to 35% for exenatide-treated patients. Vomiting was also frequent, affecting 33% of patients on the 10 mg dose and 37% on the 20 mg dose of Taspoglutide, while exenatide showed a lower rate at 16%.
Another notable adverse effect was related to local injection site reactions. These reactions, including nodule formation, induration, and pruritus, were significantly more frequent among patients receiving Taspoglutide than those on placebo. In the T-emerge 1 trial, 36% of patients on the 10 mg dose and 34% on the 20 mg dose reported injection site reactions, compared to just 11% in the placebo group. Such side effects raised concerns about patient comfort and adherence.
An unexpected challenge was the development of anti-taspoglutide antibodies in a large number of patients. In the T-emerge 2 trial, antibody formation was detected in 43% of patients on the 10 mg dose and 55% on the 20 mg dose. The presence of these antibodies was associated with hypersensitivity reactions, which further contributed to the high discontinuation rate. Over time, the dropout rate for patients on Taspoglutide reached 34%, in contrast to 16% for those on exenatide, underscoring the adverse impact of these immune responses on long-term tolerability.
Lessons Learned and Future Implications for GLP-1 RA Development
Taspoglutide’s journey in clinical development reflects both the potential and challenges associated with designing long-acting GLP-1 receptor agonists for diabetes management. Initially viewed as a promising candidate for once-weekly dosing, Taspoglutide offered an appealing alternative to daily injections, with enhanced HbA1c reduction and weight loss benefits. However, the high prevalence of adverse events, including severe GI side effects, injection site reactions, and immune responses, presented substantial barriers to patient adherence and long-term tolerability.
One of the major lessons from Taspoglutide’s trials is the critical need to balance efficacy with safety in developing sustained-release formulations for GLP-1 RAs. The high incidence of anti-drug antibody formation in patients treated with Taspoglutide, for instance, highlighted an immunogenic response that could be a factor in hypersensitivity reactions, reducing the drug’s feasibility as a viable treatment option. This immune response underscores the importance of evaluating immunogenicity when designing long-acting peptide therapies for chronic diseases like diabetes.
Ultimately, Roche, the pharmaceutical company developing Taspoglutide, ceased further development and returned the drug to Ipsen, its originator, after conducting a root cause analysis. While Taspoglutide did not reach the market, its development has informed future GLP-1 RA research by underscoring the potential adverse impacts of prolonged exposure to modified peptide drugs. This knowledge has guided the design of newer GLP-1 RAs with a focus on minimizing immunogenicity and enhancing patient safety, pointing to a future where safer long-acting treatments may still fulfill Taspoglutide’s initial promise.
 LinkPeptide
LinkPeptide