Beyond Serelaxin: Engineering Durable RXFP1 Agonists for Next-Generation Cardiovascular and Renal Therapy
Abstract
Relaxin-2, a hormone with well-documented roles in vascular and renal physiology, has demonstrated therapeutic potential in acute heart failure and renal dysfunction. However, the clinical utility of serelaxin, its recombinant form, is hindered by a short half-life and intravenous delivery requirements. R2R01, a novel single-chain peptide agonist of RXFP1, was developed to address these limitations through strategic structural modifications, including fatty acid acylation and albumin-binding enhancements. Preclinical studies confirm R2R01’s potent and selective activation of RXFP1, extended pharmacokinetics, and robust hemodynamic efficacy across acute and chronic rodent models. Importantly, comprehensive safety evaluations revealed minimal pseudo-allergic or immunogenic responses, supporting its advancement to clinical trials. With its favorable pharmacological and safety profile, R2R01 represents a promising therapeutic innovation for chronic cardiovascular and renal diseases, offering a more durable and convenient alternative to traditional relaxin-based interventions.
Targeting RXFP1: The Therapeutic Promise of Relaxin Beyond Reproduction
Relaxin-2, a member of the insulin-like peptide hormone family, plays a critical role in reproductive physiology, notably during pregnancy, where it mediates uterine quiescence, cervical remodeling, and mammary gland development. Beyond its reproductive effects, relaxin-2 has garnered significant interest for its vasodilatory, anti-fibrotic, and renoprotective properties. These pleiotropic actions are largely mediated through its primary receptor, the relaxin family peptide receptor 1 (RXFP1), a G protein-coupled receptor (GPCR) expressed in key organs such as the heart, kidneys, lungs, and vasculature.
In both preclinical and early clinical studies, recombinant human relaxin-2 (serelaxin) demonstrated promise in improving hemodynamic parameters in acute heart failure and in alleviating renal dysfunction. However, its clinical utility has been limited by pharmacokinetic constraints, notably a short systemic half-life and the need for intravenous administration, restricting its application to acute inpatient settings. These limitations underscore a critical unmet need for a long-acting RXFP1 agonist suitable for chronic outpatient use.
To address these challenges, novel molecular strategies have been pursued to enhance receptor engagement while extending peptide stability and bioavailability. Among these, R2R01, a fatty acid-acylated, single-chain peptide agonist of RXFP1, has emerged as a promising candidate. Engineered for subcutaneous delivery and sustained pharmacodynamic action, R2R01 offers a potential paradigm shift in the treatment of renal and cardiovascular diseases driven by RXFP1 dysfunction.
Engineering R2R01: A Next-Generation Single-Chain RXFP1 Agonist
R2R01 represents a next-generation engineered peptide agonist of RXFP1, rationally designed to overcome the pharmacokinetic and pharmacodynamic limitations associated with native relaxin-2. Unlike the endogenous heterodimeric relaxin hormone, R2R01 is a single-chain peptide derived from the B-chain of relaxin, optimized to retain receptor specificity while enhancing in vivo stability and duration of action.
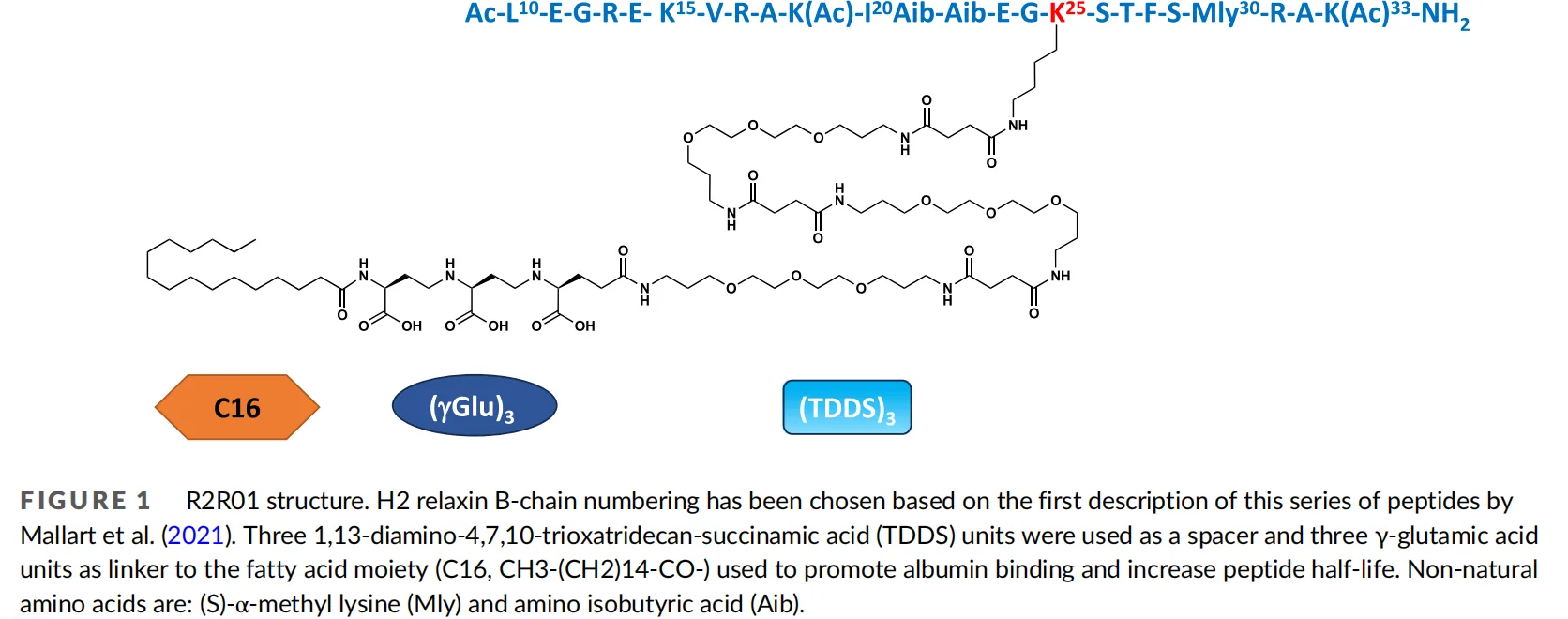
The molecular design of R2R01 incorporates several critical innovations. A key feature is the site-specific conjugation of a palmitoyl (C16) fatty acid moiety, which enhances plasma protein binding—particularly to human serum albumin (HSA)—and reduces renal clearance, thereby prolonging systemic exposure. To mitigate the oxidative metabolism previously observed with earlier analogs (e.g., SA10), the fatty acid was repositioned from the C-terminus to the ε-amino group of lysine at position 25, using a TTDS-based hydrophilic spacer to preserve RXFP1 activation capacity. Additionally, incorporation of non-natural amino acids, including (S)-α-methyl-lysine (Mly) and amino isobutyric acid (Aib), further improved the molecule’s proteolytic resistance and bioactivity.
Functionally, R2R01 exhibits high-affinity binding to RXFP1 in human cell lines, with a low nanomolar IC50, and elicits robust intracellular cAMP production—comparable in potency and efficacy to native relaxin. Importantly, R2R01 demonstrates full agonism without evidence of signaling bias, supporting its capacity to mimic the full spectrum of relaxin-mediated RXFP1 activation.
Selectivity profiling confirmed that R2R01 does not activate other GPCRs, including members of the relaxin receptor family (RXFP2–4) and other leucine-rich repeat GPCRs (e.g., LGR4–6), highlighting its high receptor specificity. These features collectively position R2R01 as a pharmacologically precise and durable RXFP1 agonist suitable for therapeutic use in chronic cardiovascular and renal conditions.
Extended Exposure, Sustained Action: R2R01’s Pharmacokinetic and Hemodynamic Profile
The therapeutic viability of peptide-based agonists is largely constrained by their metabolic instability and rapid clearance. R2R01 was specifically engineered to address these limitations through fatty acid acylation and albumin binding, which are well-established strategies to extend peptide half-life. In vitro binding assays confirmed that R2R01 binds extensively to plasma proteins across multiple species, including humans, with >99.9% binding observed. This high degree of albumin association significantly slows renal elimination and prolongs systemic exposure.
Pharmacokinetic studies conducted in Sprague–Dawley rats and Göttingen minipigs demonstrated favorable profiles following both intravenous and subcutaneous administration. In rats, the terminal half-life was approximately 10 hours, while in minipigs—a more translatable model for human PK—the half-life extended to over 26 hours. The subcutaneous bioavailability of R2R01 in minipigs was calculated to be nearly 90%, supporting the feasibility of once-daily or extended-interval dosing in future clinical applications.
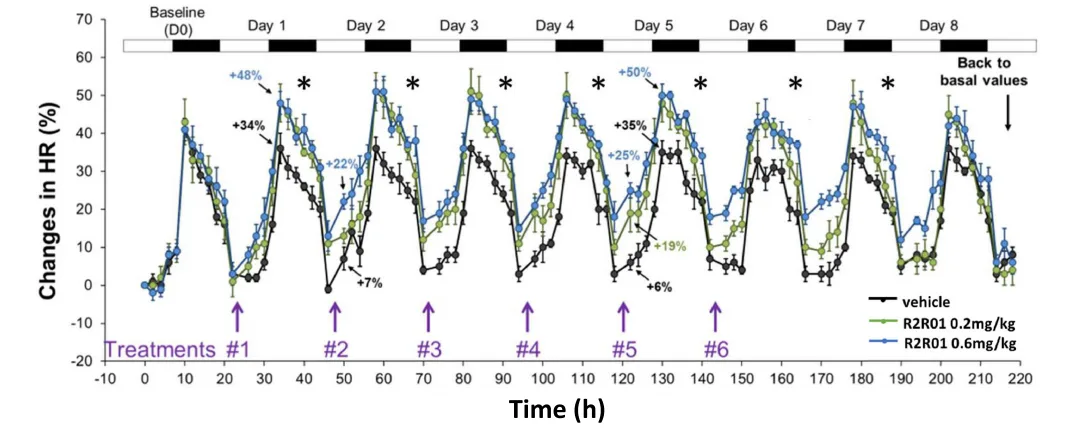
Pharmacodynamic effects of R2R01 in conscious healthy telemetered CD® rats.
Pharmacodynamic studies further validated the translational relevance of R2R01’s in vivo activity. In conscious, telemetered rats, subcutaneous R2R01 administration induced dose-dependent and sustained increases in heart rate, a known surrogate marker of RXFP1 engagement. More significantly, R2R01 produced robust and reproducible increases in renal blood flow (RBF) in both normotensive and hypertensive rodent models, accompanied by reductions in renal vascular resistance.
Given that relaxin-induced renal vasodilation is a hallmark of RXFP1 activation, RBF serves as a quantitative, translational biomarker of R2R01 efficacy. The preservation of this response across acute and chronic models affirms R2R01’s potential for long-term modulation of renal hemodynamics in clinical settings.
De-risking R2R01: Immunogenicity and Pseudo-Allergy Evaluation in Preclinical Models
A critical component in the development of therapeutic peptides is the early identification and mitigation of potential safety risks, particularly pseudo-allergic reactions and immunogenicity. R2R01 underwent a comprehensive preclinical safety evaluation, incorporating both in vitro and in vivo approaches to assess its tolerability and immune activation potential.
To investigate the risk of non-IgE-mediated mast cell degranulation, R2R01 was tested in LAD2 human mast cells—a standard model for evaluating pseudo-allergic reactions via MRGPRX2 activation. Degranulation was measured through β-hexosaminidase release. While the positive control (substance P) elicited robust responses at sub-micromolar concentrations, R2R01 induced negligible mast cell activation, even at ≥10 μM, suggesting a low potential for pseudo-allergic events in humans.
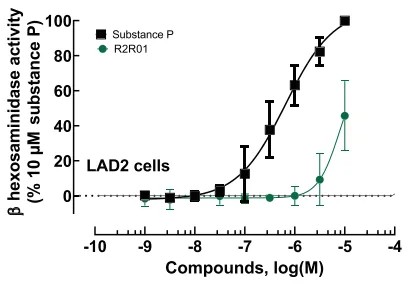
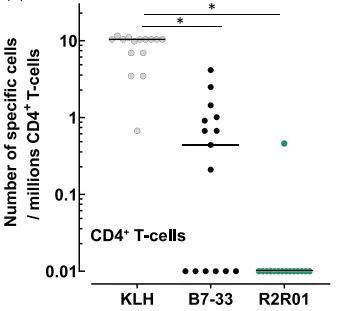
De-risking potential pseudo-allergic and immune reactions with R2R01
Additionally, immunogenicity was assessed through a sensitive CD4+ T-cell assay using peripheral blood mononuclear cells (PBMCs) from 15 healthy human donors with diverse HLA-DR genotypes. The frequency of R2R01-specific T-cells was extremely low, with only one donor exhibiting a weak response. In contrast, B7-33, a comparator RXFP1 peptide agonist, elicited broader T-cell recognition. These findings were contextualized with historical clinical data, where continuous serelaxin infusion was associated with high immunogenicity rates and anti-relaxin antibody formation.
The minimal immune activation observed with R2R01 underscores the importance of rational peptide design and early de-risking strategies. By incorporating non-natural amino acids and avoiding known immunodominant epitopes, R2R01 achieves an improved safety profile, supporting its further clinical development as a chronic therapy for renal and cardiovascular diseases.
From Bench to Bedside: R2R01 as a Transformative Therapy for Cardiorenal Disease
The development of R2R01 marks a significant advancement in peptide-based pharmacotherapy for chronic cardiorenal conditions. Building upon robust preclinical data, R2R01 has now advanced through Phase 1 clinical trials, where its pharmacokinetics, tolerability, and safety were confirmed in healthy volunteers. The compound is currently undergoing evaluation in Phase 2 studies, targeting patients with renal and cardiovascular dysfunction, where the sustained activation of RXFP1 may offer both symptomatic relief and disease modification.
One of the most promising attributes of R2R01 is its once-daily subcutaneous administration, a considerable improvement over the short-lived and intravenously administered serelaxin. This extended dosing window enhances patient convenience and adherence, particularly in outpatient and chronic care settings. Furthermore, the strong albumin binding and long half-life of R2R01 open the door to less frequent dosing schedules, pending further clinical validation.
Given RXFP1’s established roles in vascular tone regulation, anti-fibrotic activity, and renal hemodynamics, R2R01 holds therapeutic potential beyond heart failure. Ongoing investigations may explore its application in chronic kidney disease, systemic fibrosis, and resistant hypertension, where traditional treatments have shown limited efficacy.
In conclusion, R2R01 exemplifies the power of structure-based peptide engineering to overcome historical limitations of biologic therapies. Its emergence reflects a broader paradigm shift toward long-acting, receptor-specific peptide drugs that combine precision, durability, and safety. If current clinical findings are upheld, R2R01 may inaugurate a new class of RXFP1 agonists capable of transforming the management of complex cardiovascular and renal diseases.
 LinkPeptide
LinkPeptide