Beyond the Blue: How Methylene Blue Shapes Medicine and the Environment
Abstract
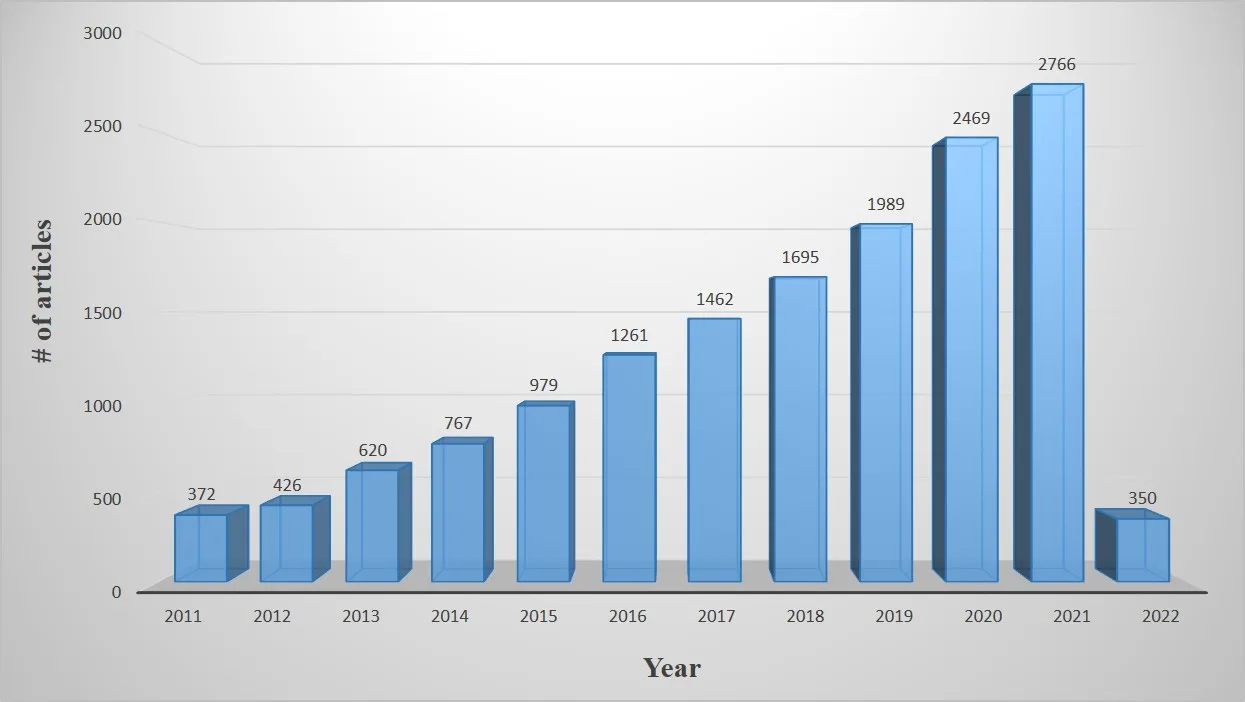
Methylene blue (MB) is a compound that has journeyed far beyond its origins as a textile dye. Today, it plays a pivotal role in medicine, industry, and environmental science, thanks to its unique chemical properties. In healthcare, methylene blue serves as a treatment for methemoglobinemia, a diagnostic tool in surgery, and a subject of ongoing research in neurodegenerative diseases. In laboratories and factories, it functions as a reliable dye and redox indicator, while also acting as a model pollutant in wastewater studies. However, MB is not without risks—high doses can be toxic, and its environmental persistence poses challenges. Promisingly, research into photodegradation technologies is transforming methylene blue into a model system for developing sustainable water treatment solutions. This blog explores methylene blue’s properties, applications, risks, and remediation, highlighting how a century-old dye continues to inspire modern scientific innovation.
Introduction – More Than Just a Blue Dye
When we hear the word “dye,” we often think of colorful fabrics, inks, or everyday stains. Yet one of the world’s most remarkable dyes, methylene blue (MB), tells a story that stretches far beyond textiles. First synthesized in the late 19th century, methylene blue was initially valued for its deep, stable blue color in fabric production. But scientists soon realized that this compound was more than just a pigment—it carried a set of unique chemical and biological properties that opened doors in medicine, research, and environmental science.
Over the years, methylene blue evolved from a simple textile dye into a lifesaving medical agent, used to treat blood disorders, combat infections, and assist in surgical diagnostics. Its extraordinary ability to shuttle electrons and respond to light has made it indispensable in laboratories, where it functions as a redox indicator, and in industry, where it serves as a model pollutant for water treatment research. More recently, it has even attracted attention in neurological research, offering intriguing possibilities in the study of Alzheimer’s and Parkinson’s disease.
Yet methylene blue is also a compound of contradictions. At controlled doses, it can heal; in excess, it becomes toxic. In the environment, it lingers as a pollutant, while simultaneously providing a pathway for scientists to test new cleanup technologies. This blog explores methylene blue’s **multifaceted identity—beneficial, hazardous, and environmentally significant—**to understand why a dye discovered over a century ago continues to shape modern science.
The Chemistry of Methylene Blue: What Makes It Unique
The remarkable versatility of methylene blue lies in its chemical identity. Classified as a thiazine dye, it is a positively charged, or cationic, compound. This positive charge gives MB a strong affinity for negatively charged molecules, such as proteins, nucleic acids, and cell membranes—an interaction that underpins many of its medical and laboratory uses.
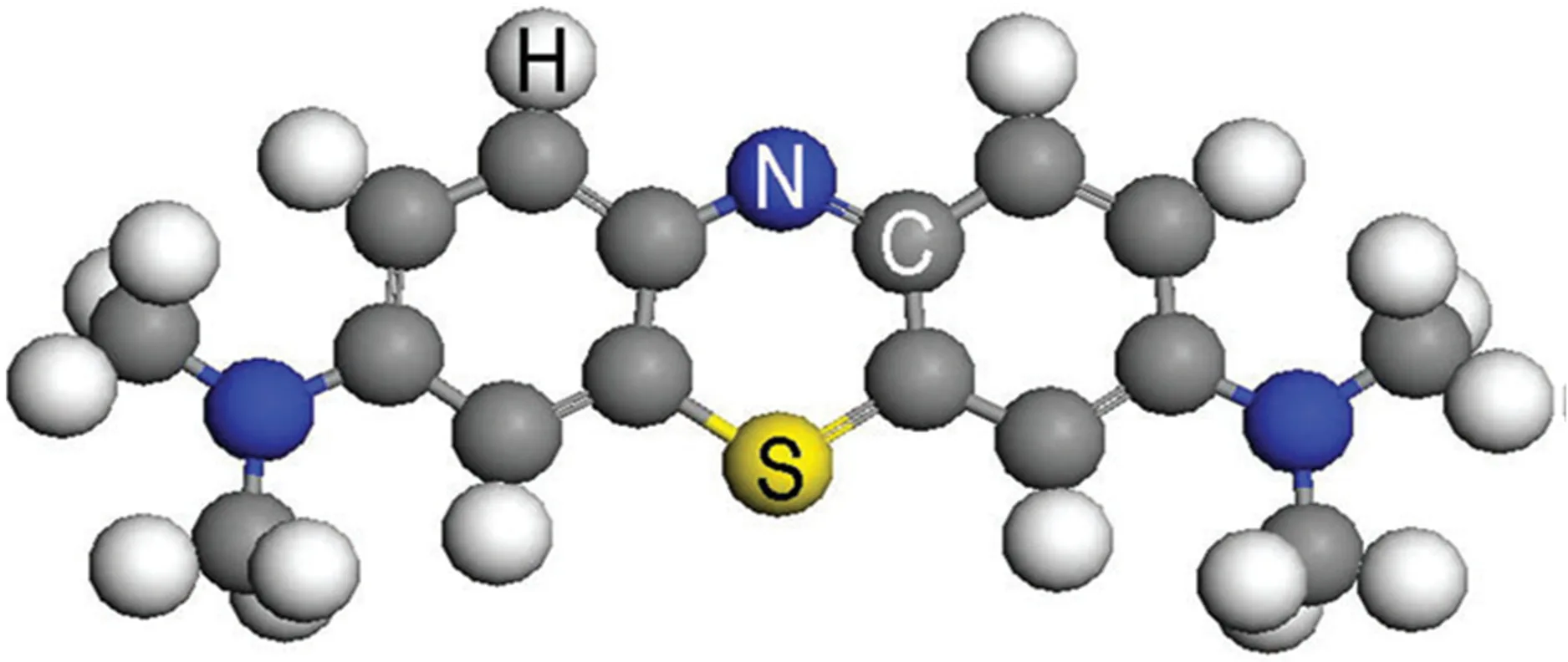
In its pure form, methylene blue appears as dark green crystals or powder. When dissolved in water or ethanol, however, it transforms into the vibrant deep-blue solution that gave it its name. One of MB’s most striking features is its ability to reversibly change color depending on its oxidation state. In the oxidized form, it is an intense blue, while in the reduced form—known as leucomethylene blue—it becomes almost colorless. This reversible redox cycling makes it a powerful redox indicator in analytical chemistry and a therapeutic tool in restoring the oxygen-carrying capacity of blood in certain medical emergencies.
Equally important is MB’s photosensitivity. It absorbs strongly in the visible spectrum, meaning it can be activated by light. When exposed to light energy, methylene blue can transfer that energy to oxygen molecules, generating reactive oxygen species (ROS). These highly reactive molecules can attack biological structures or break down pollutants, a property that makes MB valuable in both photodynamic therapy and wastewater treatment research.
In short, methylene blue is far more than a dye. Its chemical reactivity, redox flexibility, and light sensitivity make it a hybrid tool that crosses disciplines—from medicine to chemistry to environmental engineering.
From Hospitals to Factories: A Compound with Many Lives
Few compounds can claim such a diverse portfolio of uses as methylene blue. What began as a textile dye has expanded into a chemical tool and even a life-saving medicine. Its applications today span across healthcare, laboratories, and industrial processes.
Medical Applications
One of the best-known uses of methylene blue is as an antidote for methemoglobinemia, a condition where hemoglobin loses its ability to carry oxygen. By reducing oxidized hemoglobin (Fe3+) back to its functional state (Fe2+), MB can rapidly restore oxygen delivery to tissues, making it invaluable in emergency medicine.
Beyond this, MB exhibits antimicrobial activity, effective against bacteria, fungi, and certain parasites. Historically, it was even used as one of the first synthetic antimalarials before more targeted drugs became available. Researchers are also investigating its neuroprotective potential in diseases like Alzheimer’s and Parkinson’s, where MB’s ability to support mitochondrial function and act as an antioxidant could slow disease progression. In clinical practice, MB is additionally employed as a diagnostic stain and surgical tracer, helping surgeons identify tissues and pathways with precision.
Industrial and Environmental Uses
In industry, methylene blue remains a staple in textile and paper dyeing thanks to its intense color and binding properties. In the laboratory, it serves as a redox and pH indicator, making it a familiar tool for chemists and biologists alike.
Ironically, while methylene blue itself can be a pollutant, it is widely used as a model contaminant in environmental research. Scientists rely on MB to test and refine new methods for wastewater treatment, particularly photocatalytic degradation. Its stability and detectability make it an ideal “test dye” for developing eco-friendly cleanup technologies.
Taken together, these roles illustrate methylene blue’s extraordinary versatility. It is both a medicine and a pollutant, a problem and a solution, embodying the paradoxical nature of many powerful scientific tools.
The Other Side of the Story: Toxicity and Risks
As with many powerful compounds, methylene blue is not without risks. At therapeutic doses, it is generally safe and effective, but crossing that threshold can tip its effects from beneficial to harmful.
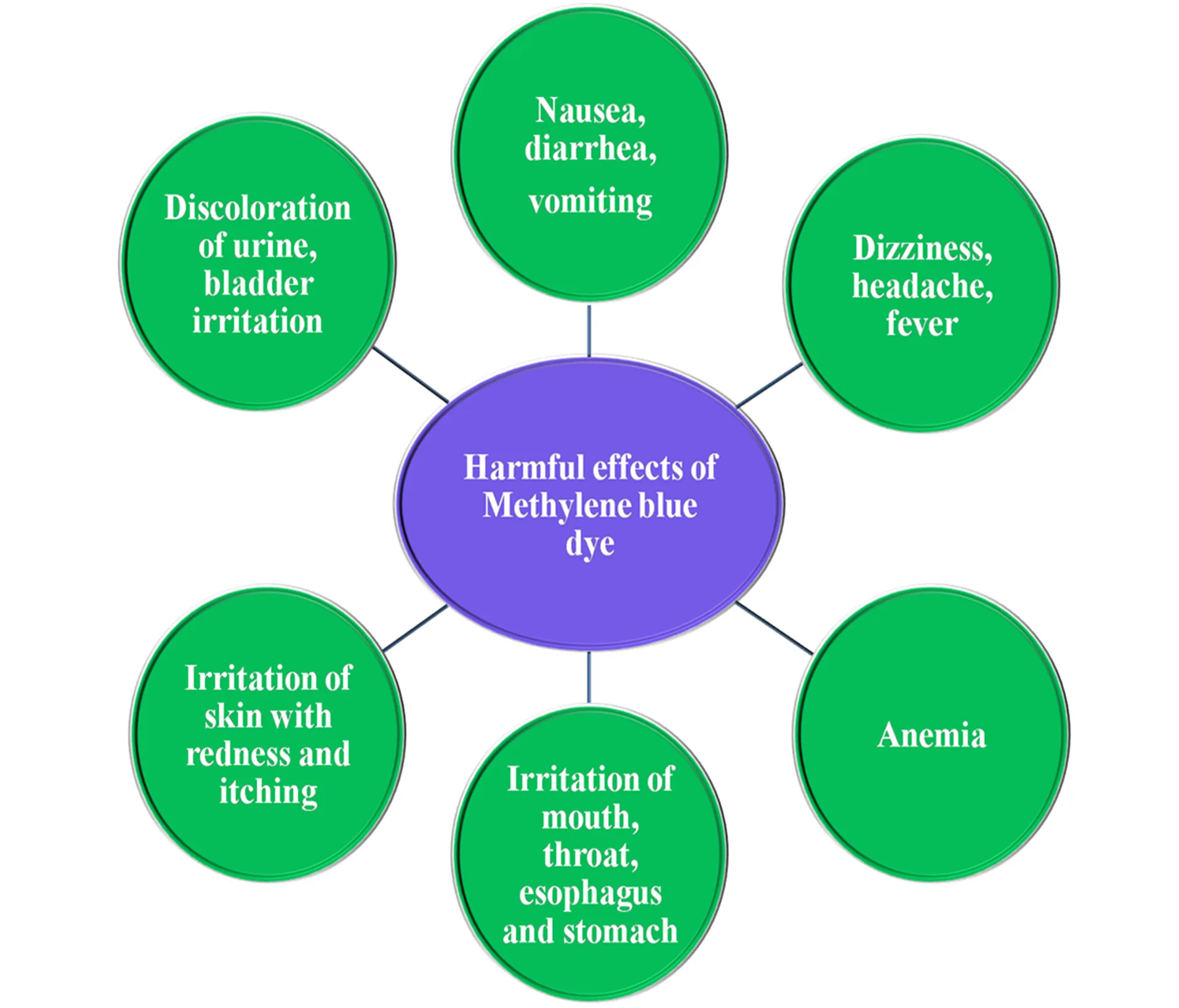
In clinical use, one of the most concerning risks arises in individuals with glucose-6-phosphate dehydrogenase (G6PD) deficiency. For these patients, methylene blue can trigger hemolytic anemia, a dangerous breakdown of red blood cells. Even in the general population, high doses may cause nausea, abdominal pain, dizziness, or headaches. Another serious concern is the potential for serotonin syndrome when MB is combined with antidepressants such as SSRIs, due to its interaction with serotonin metabolism.
In industrial or laboratory settings, methylene blue can also cause problems with prolonged exposure. Contact with skin or mucous membranes may result in irritation, while inhalation of dust or mist can affect the respiratory tract. Chronic exposure in animal studies has been linked to oxidative stress and tissue damage, highlighting the importance of protective measures when handling MB outside controlled medical contexts.
This dual nature—therapeutic at one dose, toxic at another—underscores why methylene blue must always be used under strict medical supervision and handled with occupational safety protocols in industrial environments. Understanding its risks ensures that its many benefits can be harnessed responsibly, without tipping the balance toward harm.
Light as a Remedy: Photodegradation and Environmental Solutions
While methylene blue has proven invaluable in medicine and industry, its persistence in the environment presents a serious challenge. When released into wastewater from textile factories, laboratories, or hospitals, MB does not break down easily. Even at low concentrations, it can reduce light penetration in aquatic ecosystems, disrupt photosynthesis, and harm aquatic organisms. This stability, once a benefit in industry, becomes a liability in nature.
To address this, scientists have turned to photodegradation—a process that uses light to break MB down into harmless byproducts. The principle is straightforward: when methylene blue absorbs UV or visible light, it becomes excited and transfers energy to surrounding oxygen molecules, creating reactive oxygen species (ROS). These ROS, including hydroxyl radicals and singlet oxygen, attack the dye’s molecular structure, leading to its breakdown and eventual mineralization.
The process is greatly enhanced by photocatalysts, such as titanium dioxide (TiO₂), zinc oxide (ZnO), doped semiconductors, and engineered nanomaterials. These catalysts accelerate the generation of ROS under light, drastically improving the efficiency of dye degradation. Recent studies have shown that combining visible light with advanced catalysts can achieve near-complete removal of MB from water samples, offering a sustainable approach to wastewater treatment.
Beyond treating dye pollution, methylene blue serves as a benchmark pollutant in environmental science. By studying its degradation, researchers refine photocatalytic technologies that can later be applied to more complex and hazardous contaminants. In this way, MB transforms from an environmental problem into a model system driving innovation in green chemistry and pollution control.
Conclusion – A Century-Old Dye Shaping Modern Science
Methylene blue is a compound that defies simple categorization. Born as a textile dye, it has since proven its worth as a therapeutic agent, laboratory tool, industrial dye, and environmental model pollutant. Its redox flexibility and light sensitivity have allowed it to cross boundaries between disciplines, shaping advances in medicine, chemistry, and environmental engineering alike.
Yet, with its many benefits come challenges. In medicine, it must be carefully dosed to avoid toxicity; in the environment, it demands effective strategies to prevent pollution. Encouragingly, research into photodegradation is turning this once-persistent dye into a catalyst for innovation in sustainable wastewater treatment.
In the end, methylene blue exemplifies how a seemingly ordinary chemical can have an extraordinary impact, reminding us that even the most familiar substances may hold the key to solving tomorrow’s problems.
Reference
Khan, I., Saeed, K., Zekker, I., Zhang, B., Hendi, A. H., Ahmad, A., … & Khan, I. (2022). Review on methylene blue: its properties, uses, toxicity and photodegradation. Water, 14(2), 242.
https://doi.org/10.3390/w14020242
Benkhaya, S., M’rabet, S., & El Harfi, A. (2020). A review on classifications, recent synthesis and applications of textile dyes. Inorganic Chemistry Communications, 115, 107891.
https://doi.org/10.1016/j.inoche.2020.107891
Derakhshan, Z., Baghapour, M. A., Ranjbar, M., & Faramarzian, M. (2013). Adsorption of methylene blue dye from aqueous solutions by modified pumice stone: kinetics and equilibrium studies.
https://www.sid.ir/paper/330349/en
Ali, S., Khan, S. A., Khan, I., Yamani, Z. H., Sohail, M., & Morsy, M. A. (2017). Surfactant-free synthesis of ellipsoidal and spherical shaped TiO2 nanoparticles and their comparative photocatalytic studies. Journal of environmental chemical engineering, 5(4), 3956-3962.
 LinkPeptide
LinkPeptide