Bismuth Bicycles: A Single-Atom Strategy for Next-Generation Bicyclic Peptides
Abstract
Bicyclic peptides have emerged as a powerful therapeutic modality that bridges the gap between small molecules and antibodies by combining high binding affinity with synthetic accessibility. Recent advances introduce bismuth Bicycle molecules, a novel class of constrained peptides in which three cysteine residues are coordinated to a single bismuth(III) ion, replacing conventional C3-symmetric organic scaffolds. This single-atom scaffold enforces exceptional conformational rigidity while enabling rapid, quantitative cyclisation under biocompatible conditions. As a result, bismuth Bicycles demonstrate markedly improved target affinity, enhanced proteolytic stability, and superior cellular uptake compared with linear peptides. Importantly, the embedded bismuth core enables label-free quantification and imaging using elemental analysis techniques, and is fully compatible with phage display for large-scale ligand discovery. Together, these properties position bismuth Bicycles as a versatile platform for next-generation peptide therapeutics, with promising applications in oncology, infectious diseases, intracellular targeting, and targeted radiopharmaceutical development.
Why Bicyclic Peptides Matter to the Peptide Industry
Over the past decade, bicyclic peptides have transitioned from academic curiosities to clinically validated therapeutic modalities. Positioned between small molecules and antibodies, bicyclic peptides offer a unique combination of high binding affinity, structural precision, and synthetic accessibility. For peptide suppliers and CROs, this class of molecules represents not only a growing market but also an opportunity to support next-generation drug discovery programs.
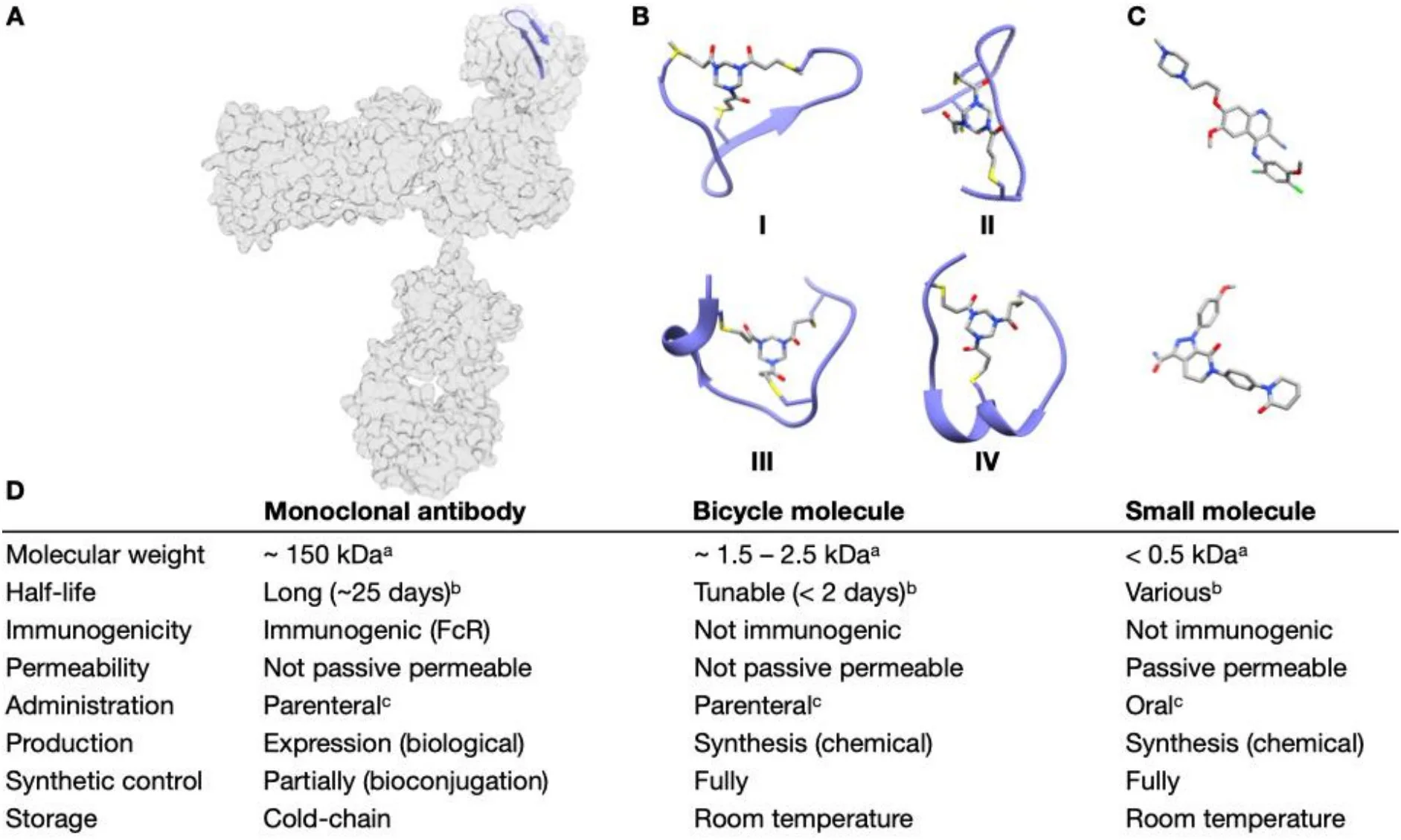
Unlike linear peptides, bicyclic peptides are conformationally constrained, allowing them to engage challenging targets such as protein–protein interactions with antibody-like specificity. At the same time, their small size enables better tissue penetration, rapid clearance, and flexible chemical modification—attributes highly valued in modern therapeutic design.
Historically, bicyclic peptides have been generated using C3-symmetric organic scaffolds that link three cysteine residues into a rigid framework. This approach has enabled the discovery of high-affinity ligands and supported the development of clinically advanced Bicycle Drug Conjugates. However, organic scaffolds introduce limitations related to molecular size, scaffold rigidity, and chemical diversity.
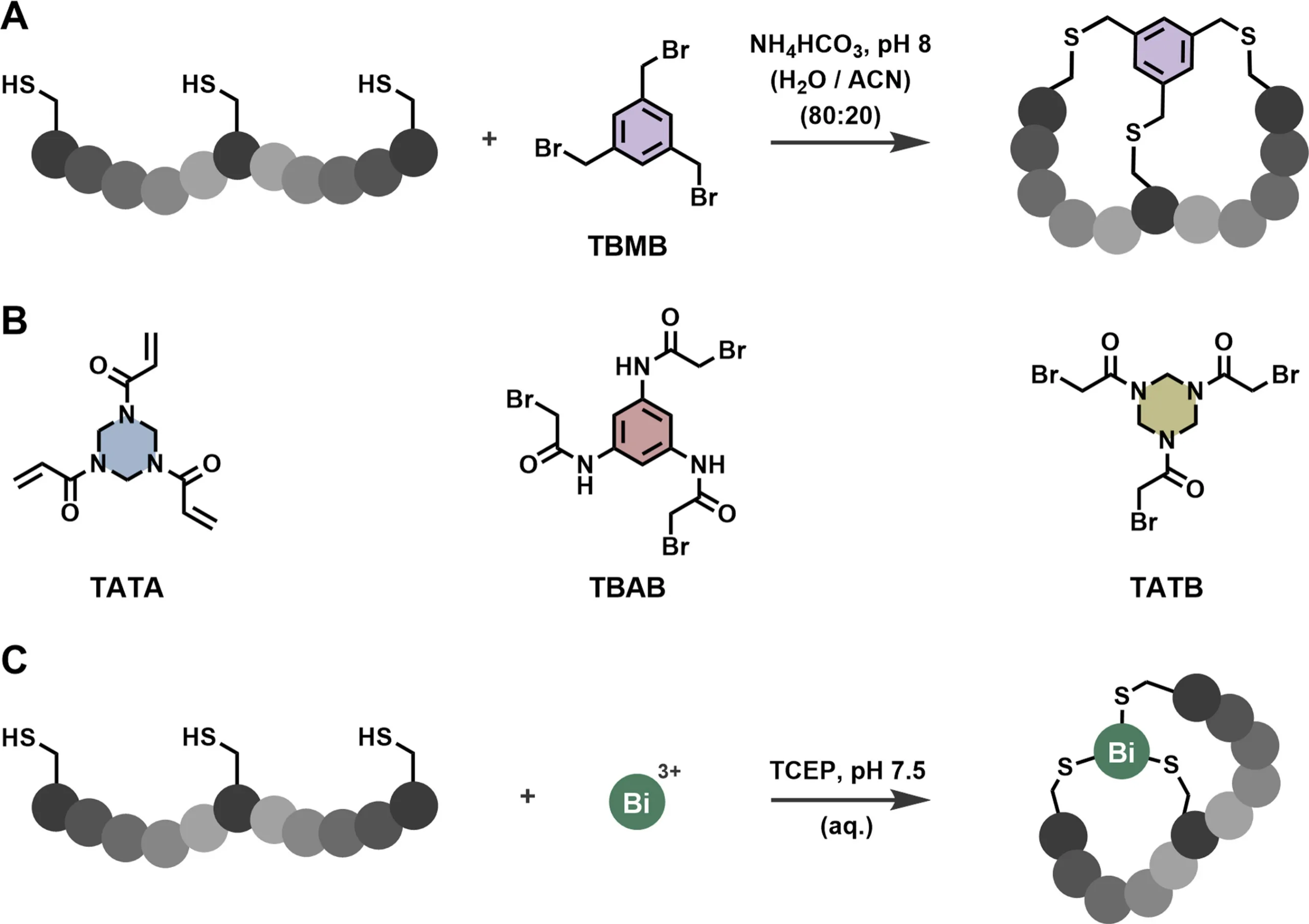
As peptide drug discovery matures, clients are increasingly seeking more compact, more tunable, and more functionally versatile constraint strategies. This demand has opened the door to an unexpected but powerful solution: metal-based peptide cyclisation using bismuth.
Bismuth Bicycles: A Minimalist Scaffold with Maximum Impact
Bismuth Bicycle molecules represent a fundamental shift in how bicyclic peptides can be constructed. Rather than relying on multi-atom organic linkers, three cysteine thiolates within a peptide coordinate directly to a single Bi(III) ion, forming a tridentate metal complex that enforces bicyclic topology.
From a chemistry standpoint, this strategy is remarkably practical. Bismuth exhibits strong and selective affinity for thiolates, enabling clean, quantitative cyclisation even in the presence of other functional groups commonly found in peptides. Importantly for CRO workflows, bismuth bicyclisation occurs under physiological conditions, tolerates reducing agents such as TCEP, and proceeds rapidly without harsh reagents.
For peptide suppliers, the most striking advantage is structural efficiency. Computational and experimental studies show that bismuth is the smallest and most constraining scaffold tested for bicyclic peptides. Compared with traditional organic scaffolds, Bi(III)-linked peptides adopt tighter, more uniform conformations with reduced flexibility.
This minimalist design offers clear benefits:
- Reduced molecular bulk
- Improved structural definition
- Greater design freedom within the peptide sequence itself
In practical terms, bismuth bicyclisation allows CROs and peptide manufacturers to offer highly constrained peptide architectures without increasing synthetic complexity—a compelling value proposition for medicinal chemistry teams.
Functional Advantages That Matter to Drug Discovery Programs
For clients, structural elegance only matters if it translates into functional gains. Bismuth Bicycles deliver on this front.
Enhanced Binding Affinity
Conformational preorganization reduces the entropic cost of target binding. In experimental systems, bismuth-cyclised peptides demonstrate substantially higher potency than their linear counterparts, including improvements of over 100-fold in enzymatic inhibition assays. For CROs supporting hit-to-lead optimization, this translates into faster affinity maturation with fewer design iterations.
Improved Proteolytic Stability
Proteolytic degradation remains a key limitation of peptide therapeutics. Bismuth Bicycle molecules exhibit 6–19× increased resistance to proteolysis compared with linear peptides. This improvement is particularly attractive for clients developing peptides for systemic administration or intracellular targets.
Tunable Stability Profiles
Stability can be further enhanced through:
- Backbone macrocyclisation prior to bismuth coordination
- Substitution of cysteine with selenocysteine to strengthen metal–ligand interactions
This modularity allows peptide service providers to customize stability profiles based on client requirements rather than relying on a one-size-fits-all scaffold.
Platform-Level Advantages: Uptake, Analytics, and Discovery
Beyond classical binding metrics, bismuth Bicycles introduce platform-level advantages that are highly relevant to CRO and supplier service offerings.
Improved Cellular Uptake
Bismuth-cyclised polycationic peptides demonstrate exceptionally efficient cellular internalisation, outperforming well-known cell-penetrating peptides at nanomolar concentrations. This positions bismuth Bicycles as attractive scaffolds for intracellular targets—an area of growing interest among biotech clients.
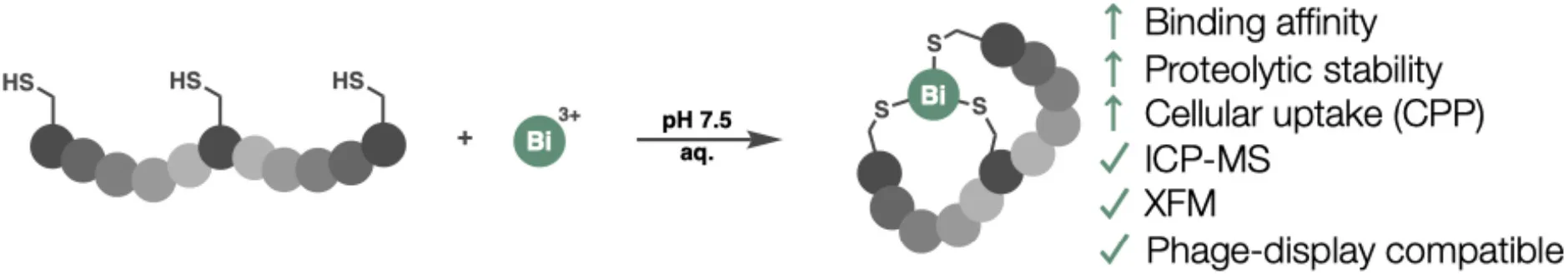
Built-In Analytical Traceability
The presence of a heavy metal core fundamentally changes how peptides can be quantified and imaged:
ICP-MS enables label-free quantification in biological samples
X-ray fluorescence microscopy (XFM) allows subcellular localisation
For CROs, this reduces reliance on fluorescent labeling, which can alter peptide behavior and complicate data interpretation. Offering bismuth-based analytical workflows can differentiate service portfolios.
Compatibility with Phage Display
Perhaps most importantly, bismuth bicyclisation is compatible with genetically encoded peptide libraries, including phage display. This enables direct selection of bismuth Bicycles from large combinatorial libraries, providing a scalable route to high-affinity binders.
For discovery-focused CROs, this opens new opportunities to offer metal-enabled peptide library screening as a premium service.
Commercial and Therapeutic Outlook
From a commercial perspective, bismuth Bicycles arrive at a moment when peptide therapeutics are expanding beyond traditional hormones and GPCR ligands into oncology, infectious disease, and intracellular biology.
Potential application areas include:
- Antiviral and antimicrobial peptides
- Intracellular enzyme inhibitors
- Targeted delivery vehicles
- Peptide-based radiopharmaceuticals
Notably, radioactive bismuth isotopes such as bismuth-213 are already used in targeted alpha therapy. The ability to rapidly and selectively generate bismuth-cyclised peptides suggests a future role for Bismuth Bicycles in precision radiotherapeutics—an area of increasing investment by biotech and pharma companies.
For peptide suppliers and CROs, bismuth Bicycles should be viewed not as a niche chemistry, but as a next-generation peptide constraint platform. They combine synthetic practicality, functional superiority, and discovery compatibility in a way that aligns closely with industry needs.
Final Takeaway for Peptide Suppliers and CROs
Bismuth Bicycle technology demonstrates how a single-atom scaffold can unlock new value across peptide design, discovery, and analysis. For service providers, it offers:
- Differentiation from conventional peptide offerings
- Access to highly constrained architectures without added synthetic burden
- New analytical and discovery workflows
- Alignment with emerging therapeutic modalities
As peptide therapeutics continue to evolve, platforms that integrate chemistry, biology, and scalability will define the next competitive edge. Bismuth Bicycles are well positioned to be one of them.
Reference
Voss, S., Sagar, A., Tiberghien, A., Hughes, R. J. L., Chen, L., Rioja, I., … & Spring, D. R. (2026). Bismuth Bicycles. Journal of Peptide Science, 32(3), e70071.https://doi.org/10.1002/psc.70071
Colas, K., Bindl, D., & Suga, H. (2024). Selection of nucleotide-encoded mass libraries of macrocyclic peptides for inaccessible drug targets. Chemical Reviews, 124(21), 12213-12241.https://doi.org/10.1021/acs.chemrev.4c00422
Ullrich, S., & Nitsche, C. (2024). Bicyclic peptides: paving the road for therapeutics of the future. Peptide Science, 116(2), e24326.https://doi.org/10.1002/pep2.24326
 LinkPeptide
LinkPeptide