Breaking the Nuclear Barrier: How DNA–Peptide Conjugates Are Advancing Non-Viral Gene Delivery
Abstract
Non-viral gene delivery has emerged as a promising alternative to viral vectors, offering advantages in safety, scalability, and payload flexibility. Yet despite major progress in lipid nanoparticles and synthetic carriers, one fundamental biological hurdle continues to limit its effectiveness: DNA must reach the nucleus to be expressed. A recent Nature Communications study introduces a compelling solution to this problem through a precisely engineered DNA–peptide conjugation strategy known as DNA-PepTAG. By enabling efficient nuclear import of DNA in non-dividing cells, this work reframes nuclear targeting as a solvable design challenge rather than an intractable barrier.
The Nuclear Bottleneck: Why DNA Delivery Still Limits Non-Viral Gene Therapy
Unlike mRNA therapeutics—which only need to access the cytoplasm—DNA-based therapies face the additional and more restrictive requirement of nuclear entry. In actively dividing cells, the nuclear envelope temporarily disassembles during mitosis, allowing a fraction of exogenous DNA to enter passively. However, many therapeutically relevant cell types, including hepatocytes, muscle cells, neurons, and differentiated tissues, divide slowly or not at all.
In these contexts, nuclear import becomes a major bottleneck. Estimates suggest that less than 1% of transfected DNA reaches the nucleus in non-dividing cells. To compensate, researchers often increase DNA dosage, which introduces new problems such as cytotoxicity, innate immune activation, and poor reproducibility. These limitations have significantly constrained the clinical translation of non-viral DNA delivery systems.
One long-standing idea has been to attach nuclear localization signals (NLSs) to DNA. NLS peptides are short amino-acid sequences that naturally guide proteins into the nucleus via the importin transport machinery. However, decades of work on DNA–NLS conjugates have produced inconsistent results. The underlying issue is not the biological concept, but rather the lack of precise chemical control. Previous approaches relied on electrostatic interactions, labile linkages, or poorly defined conjugation sites—conditions that undermine reliable nuclear targeting.
DNA-PepTAG: A Precision Strategy for Site-Specific DNA–Peptide Conjugation
DNA-PepTAG directly addresses these shortcomings by introducing site-specific, irreversible, and modular DNA–peptide conjugation. The platform is built on a chemoenzymatic strategy that allows unprecedented control over peptide placement on DNA.
At its core is Escherichia coli tRNA guanine transglycosylase (TGT), an enzyme repurposed here to modify DNA. TGT selectively replaces a single guanine within a short DNA stem-loop with a functionalized preQ1-DBCO moiety. This step is highly efficient and exquisitely site-specific, ensuring that each DNA molecule carries exactly one reactive handle.
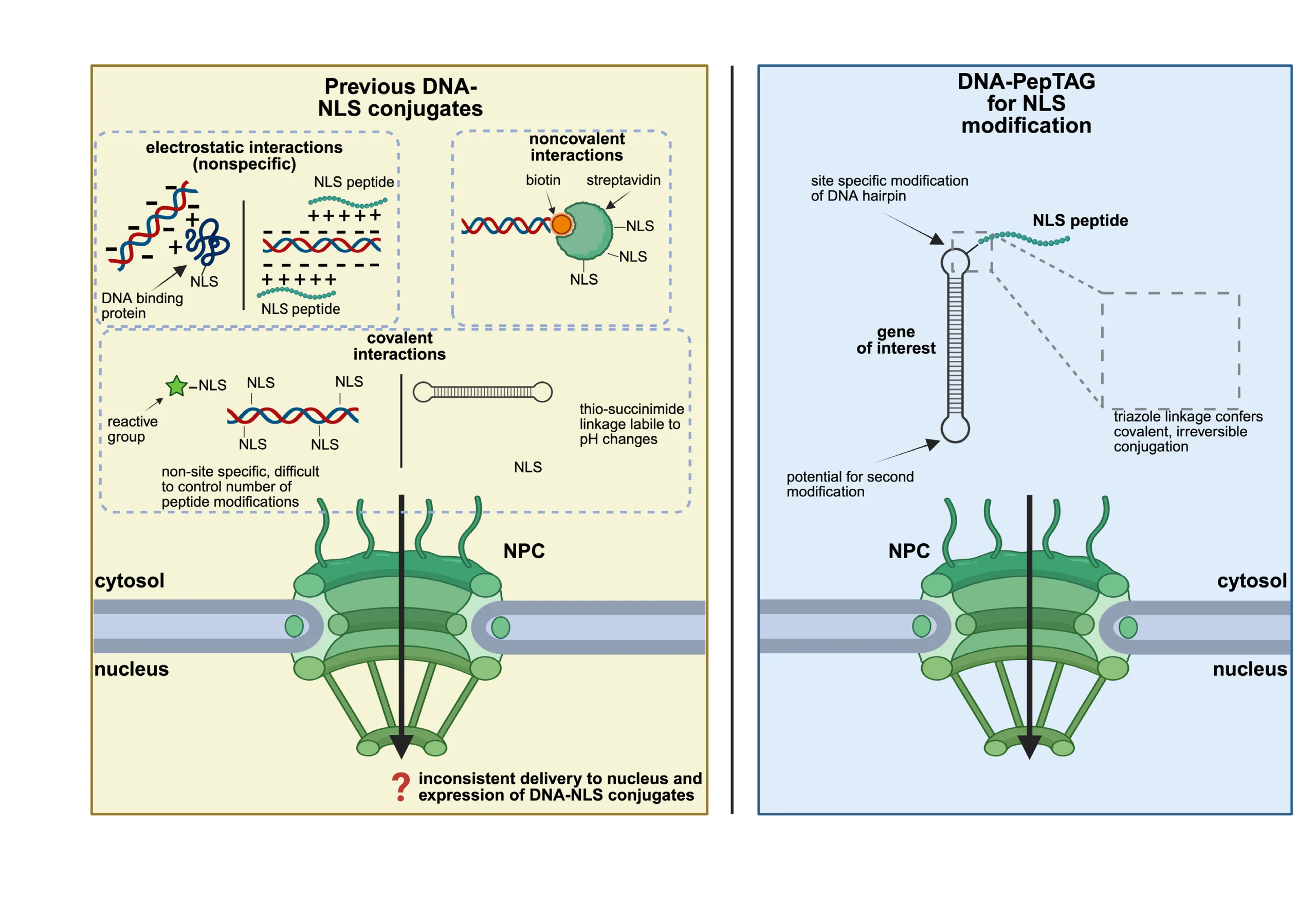
The installed DBCO group then undergoes copper-free strain-promoted azide–alkyne cycloaddition (SPAAC) with azide-modified peptides. This reaction forms a stable triazole linkage that is non-reversible under biological conditions. Importantly, the chemistry avoids copper catalysts, preserving DNA integrity and biocompatibility.
The resulting peptide-modified oligonucleotides are ligated onto linear gene cassettes, yielding capped DNA constructs with precisely positioned peptide modifications. This level of chemical definition—control over peptide identity, number, orientation, and spacing—represents a major advance over earlier DNA nuclear targeting strategies.
Engineering Nuclear Entry: Design Principles That Govern DNA Import
With DNA-PepTAG in hand, the authors systematically examined how molecular design parameters influence nuclear delivery and gene expression. Several clear design rules emerged.
First, one NLS is optimal.
DNA constructs modified with a single NLS peptide exhibited markedly enhanced nuclear import and gene expression. In contrast, attaching NLS peptides to both ends of the DNA cassette drastically reduced expression, nearly to baseline levels. This counterintuitive result likely arises from competing interactions with nuclear pore complexes, effectively trapping large DNA molecules at the nuclear envelope.
Second, linker length matters.
The distance between the DNA and the NLS peptide plays a critical role. A flexible (GGGGS)₃ linker provided the highest expression levels, outperforming shorter linkers. Longer linkers, however, offered no additional benefit, indicating an optimal balance between flexibility and spatial control that allows the NLS to engage nuclear transport factors efficiently.
Third, nuclear import is active and importin-dependent.
Quantitative PCR analysis revealed 10–20-fold enrichment of DNA in the nucleus for NLS-modified constructs, while total intracellular DNA levels remained unchanged. This confirms that enhanced expression arises from improved nuclear translocation rather than increased cellular uptake. Further validation using importazole, an importin-β inhibitor, demonstrated that nuclear entry proceeds through the canonical importin pathway.
Together, these findings transform nuclear DNA delivery from an empirical problem into a rational engineering task governed by clear molecular principles.
Choosing the Right Nuclear Localization Signal: Cell-Type Matters
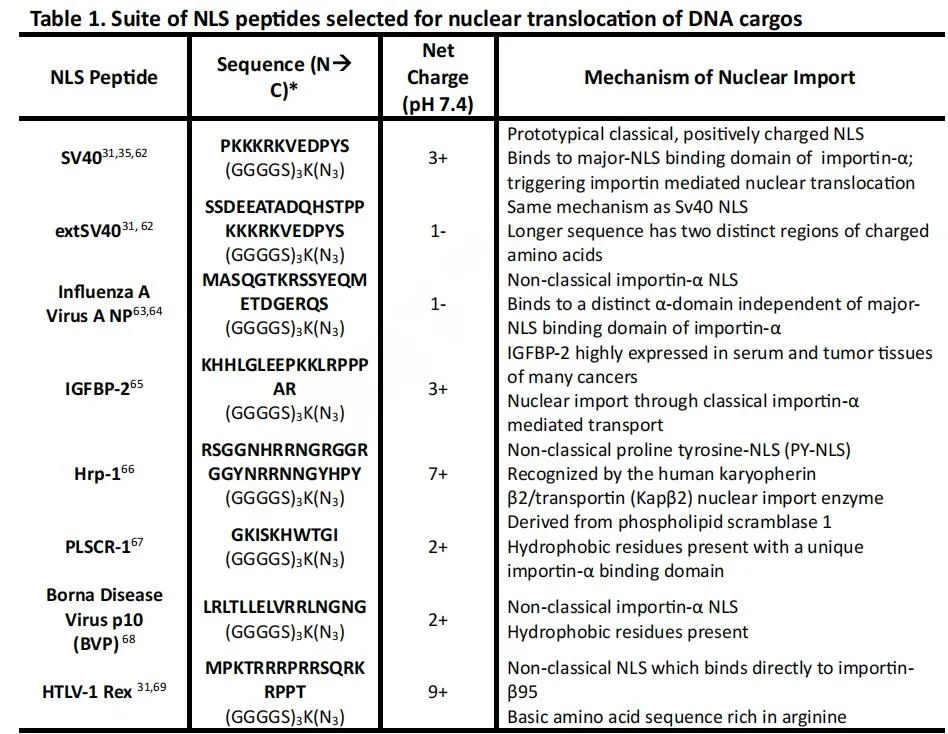
Beyond structural optimization, the study addressed a crucial but often overlooked question: do different NLS peptides perform equally across cell types? The answer is decisively no.
The authors screened a panel of NLS peptides in growth-arrested liver (HepG2), kidney (HEK293T), and cardiac (AC16) cells. While all functional NLSs improved expression relative to controls, performance varied significantly depending on both peptide identity and cell type.
Two peptides consistently outperformed the classical SV40 NLS:
- PLSCR-1 NLS, a short sequence derived from phospholipid scramblase
- extSV40 NLS, an extended, charge-balanced variant of SV40
These peptides share a key feature: reduced net positive charge, which likely minimizes unfavorable electrostatic interactions with the DNA backbone and preserves NLS accessibility to importin receptors. In contrast, highly basic NLSs may become partially sequestered by DNA, limiting their effectiveness.
The observed cell-type specificity is biologically meaningful. Importin-α isoforms are differentially expressed across tissues, influencing which NLS motifs are most efficiently recognized. This finding suggests that NLS selection can be tailored for tissue-specific gene delivery, opening new opportunities for precision therapeutics.
From Design to Therapy: Implications for Next-Generation Gene Delivery
The therapeutic relevance of DNA-PepTAG is substantial. NLS-modified DNA constructs achieved significantly higher expression at lower DNA doses, directly addressing concerns around toxicity and immune activation. The platform remained effective across DNA payloads ranging from ~1.3 kb to nearly 7 kb, accommodating large genes and genome-editing systems.
Importantly, the approach was validated using a clinically relevant target: coagulation Factor IX, a secreted protein used to treat hemophilia B. NLS-modified gene cassettes produced up to 10-fold higher and sustained Factor IX secretion, demonstrating durable expression without repeated DNA delivery.
Looking forward, DNA-PepTAG is well suited for integration with lipid nanoparticle (LNP) delivery systems, which currently dominate the non-viral therapeutic landscape. Because peptide modification is covalent and precisely defined, it should be compatible with established formulation strategies. Moreover, the platform is not limited to nuclear localization signals—any azide-functionalized peptide or ligand could, in principle, be conjugated, extending its utility beyond nuclear targeting.
Conclusion
This study represents a significant advance in non-viral gene delivery. By uniting precise enzymatic chemistry with rigorous biological validation, DNA-PepTAG demonstrates that nuclear targeting of DNA can be engineered predictably and efficiently. The work provides not only a powerful new tool, but also a set of actionable design principles that will inform the next generation of DNA-based therapeutics. As non-viral gene therapy continues to evolve, strategies like DNA-PepTAG may prove essential for unlocking its full clinical potential.
Reference
Mohamedshah, Z. Y., Chi, C. C., Tota, E. M., Komor, A. C., & Devaraj, N. K. (2026). Highly efficient expression of DNA-peptide conjugates in growth-arrested cells. Nature Communications.https://doi.org/10.1038/s41467-025-68167-5
Crooke, S. T., Baker, B. F., Crooke, R. M., & Liang, X. H. (2021). Antisense technology: an overview and prospectus. Nature reviews Drug discovery, 20(6), 427-453.https://doi.org/10.1038/s41573-021-00162-z
Hou, X., Zaks, T., Langer, R., & Dong, Y. (2021). Lipid nanoparticles for mRNA delivery. Nature Reviews Materials, 6(12), 1078-1094.https://doi.org/10.1038/s41578-021-00358-0
Sun, Y., Xian, L., Xing, H., Yu, J., Yang, Z., Yang, T., … & Ding, P. (2016). Factors influencing the nuclear targeting ability of nuclear localization signals. Journal of drug targeting, 24(10), 927-933.https://doi.org/10.1080/1061186X.2016.1184273
 LinkPeptide
LinkPeptide