From Thymus to Immunomodulation: Linking Physicochemical Profiles to the Biological Activity of Bovine Thymic Peptides
Abstract
Thymic peptides are recognized for their central role in immune regulation, yet many thymus-derived preparations remain insufficiently characterized despite long-standing therapeutic and nutraceutical use. This blog examines a recent comparative study evaluating two experimental bovine thymic peptide preparations alongside the clinically authorized reference Thymus Factor X® (TFX). By integrating physicochemical profiling with functional cell-based assays, the study elucidates how extraction and purification strategies influence peptide composition, structural stability, and immunomodulatory activity. Marked differences were observed in molecular weight distribution, solubility, amino acid composition, and thermal behavior among the preparations. Functional evaluation using an NF-κB reporter model revealed that one experimental preparation exhibited superior inhibition of inflammatory signaling while maintaining a favorable safety profile. These findings underscore the importance of comprehensive structural and functional characterization in predicting biological performance and support the rational development of thymic peptide preparations as immunomodulatory pharmaceutical or nutraceutical ingredients.
Thymic Peptides as Endogenous Immunomodulators: Biological Significance and Translational Challenges
The thymus is a cornerstone of immune system development, responsible for the maturation, selection, and functional programming of T lymphocytes. Beyond its role in immune education, the thymus secretes a diverse array of bioactive peptides—most notably thymosin α1, thymosin β4, thymulin, and thymopoietin—that regulate inflammation, immune responsiveness, tissue repair, and cellular signaling. These peptides have been studied extensively for their therapeutic potential, particularly in conditions associated with immune dysregulation and chronic inflammation.
While individual thymic peptides have been synthesized and clinically evaluated, many thymus-derived products used in pharmaceutical and nutraceutical contexts remain complex peptide mixtures. Their biological effects are typically attributed to the combined action of multiple components rather than to a single defined molecule. However, this complexity presents a major challenge: without detailed physicochemical characterization, it is difficult to predict biological efficacy, safety, or batch-to-batch consistency.
A 2025 study published in the International Journal of Peptide Research and Therapeutics directly addresses this issue by systematically examining how extraction and purification strategies shape the physicochemical properties and immunomodulatory activity of bovine thymic peptide preparations. By comparing two experimental formulations with a clinically authorized reference product, the study provides valuable insight into the structure–function relationships that underpin thymic peptide bioactivity.
Comparative Study Design and Analytical Methodology for Thymic Peptide Evaluation
The study investigated three thymic peptide preparations derived from calf thymus. Two experimental formulations—designated Pa and Pb—were produced using identical acid extraction conditions but diverged during downstream processing. Pa underwent a protein precipitation step, resulting in a preparation enriched in higher-molecular-weight components, whereas Pb was subjected to a cascade of filtration processes designed to enrich low-molecular-weight peptides. As a reference standard, the active pharmaceutical ingredient of Thymus Factor X® (TFX) was included, allowing direct comparison with a thymic peptide fraction that has demonstrated clinical utility.

To comprehensively assess these preparations, the authors employed a suite of complementary analytical techniques. Molecular weight distribution and peptide complexity were evaluated using SDS-PAGE, reversed-phase high-performance liquid chromatography (RP-HPLC), and size-exclusion HPLC (SEC-HPLC). Amino acid composition was determined following acid hydrolysis, while differential scanning calorimetry (DSC) was used to probe thermal stability and structural organization. Biological activity was assessed using a THP-1-based NF-κB reporter assay, with parallel cell viability measurements to distinguish genuine immunomodulation from cytotoxic effects.
This integrated strategy allowed the authors not only to describe compositional differences but also to directly relate physicochemical properties to functional immune outcomes.
Processing-Driven Physicochemical Diversity in Bovine Thymic Peptide Preparations
The physicochemical analyses revealed clear and systematic differences among the three preparations. Pa exhibited limited solubility in aqueous media and the broadest molecular weight distribution, spanning approximately 3 to 25 kDa. SDS-PAGE analysis showed distinct protein bands around 15 and 25 kDa, along with numerous lower-molecular-weight fractions. This heterogeneity indicates that Pa contains a mixture of peptides and larger protein components, potentially acting as reservoirs of bioactive fragments.
In contrast, Pb and API TFX were readily soluble and dominated by peptides below 10 kDa. Pb displayed an intense low-molecular-weight smear on SDS-PAGE, suggesting a high abundance of short peptides or degraded fragments, while the electrophoretic profile of TFX aligned well with previously reported thymic peptide fractions, including a prominent component around 4.2 kDa.
Chromatographic profiling further underscored these distinctions. RP-HPLC analysis showed that Pa possessed a more complex elution pattern, with both early-eluting hydrophilic peptides and later-eluting, more hydrophobic components. Pb and TFX were characterized by early-eluting peaks, consistent with short, hydrophilic peptides commonly associated with immunomodulatory signaling.
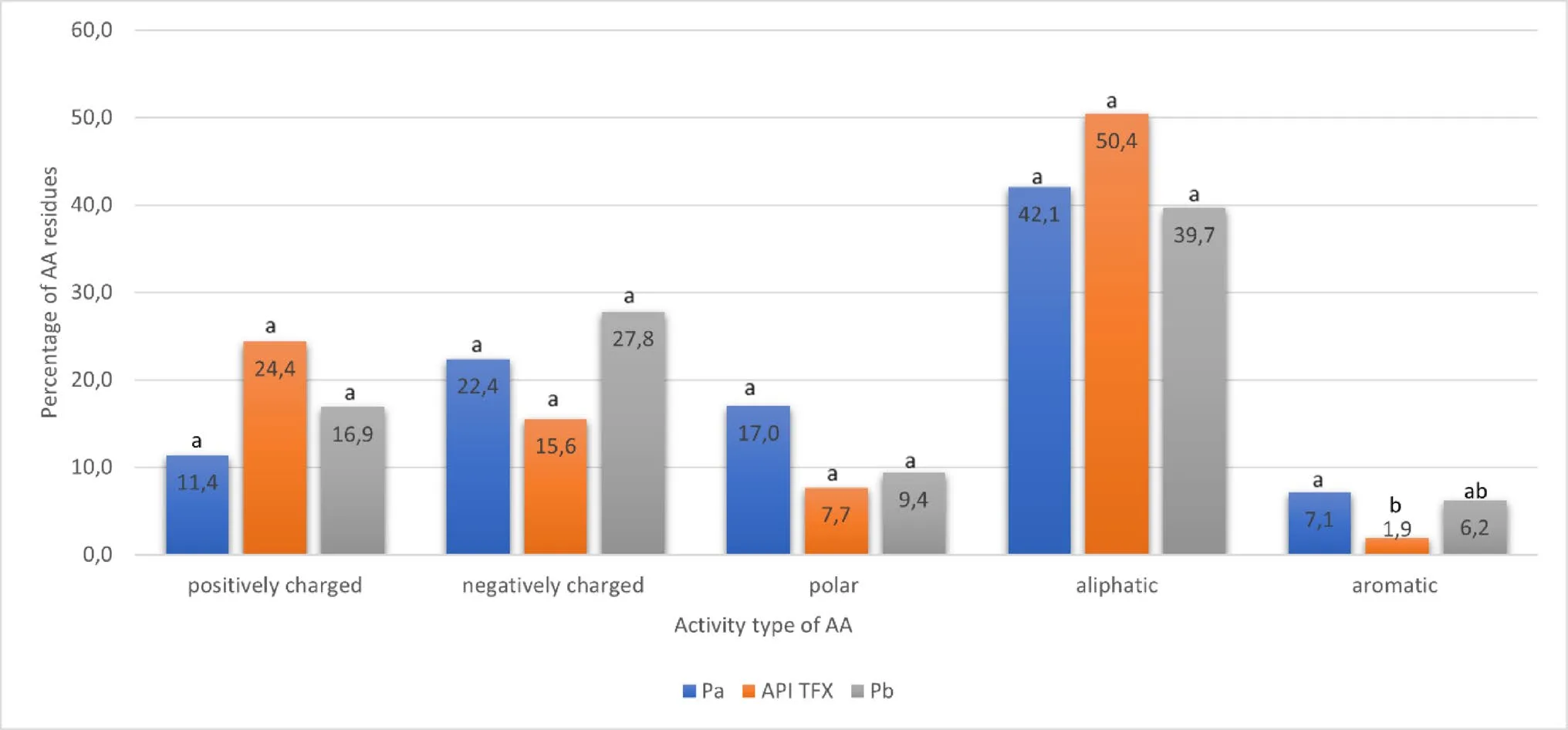
Amino acid analysis revealed broadly similar residue classes across preparations, with enrichment in glutamic acid, proline, lysine, and glycine—amino acids frequently found in flexible, intrinsically disordered peptides. Notably, Pa contained higher proportions of acidic residues, whereas TFX was particularly rich in proline and lysine, residues known to promote conformational flexibility and receptor interaction. Pb, while enriched in glutamate and lysine, showed substantially lower proline content, a feature that may influence peptide structure and biological behavior.
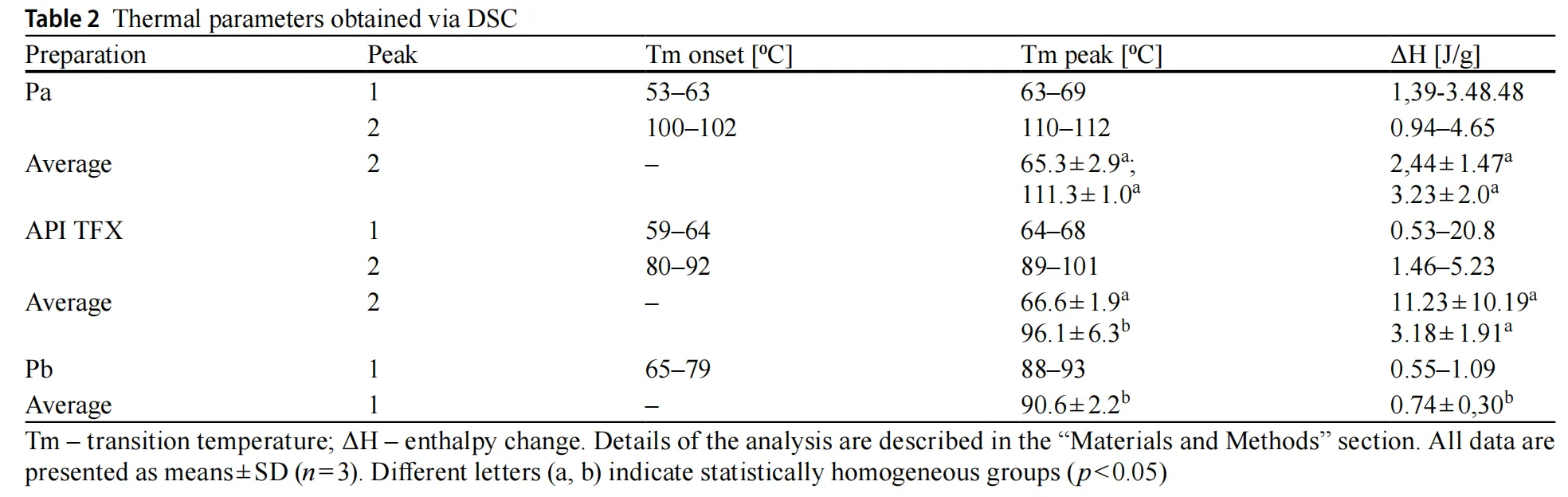
Thermal analysis by DSC provided additional insight. API TFX exhibited the highest thermal stability, consistent with its pharmaceutical-grade purity and defined composition. Pa showed reproducible but moderate thermal transitions, suggesting partial structural organization, while Pb displayed broad, low-intensity transitions indicative of a heterogeneous and less stable mixture.
Modulation of NF-κB Signaling by Thymic Peptides: Efficacy and Safety Considerations
To determine whether physicochemical differences translated into functional consequences, the authors evaluated immunomodulatory activity using a THP-1-Blue™ NF-κB reporter cell line. NF-κB is a central regulator of inflammatory signaling, controlling the transcription of numerous cytokines and immune mediators. Its activation by lipopolysaccharide (LPS) provides a robust and widely accepted model for assessing anti-inflammatory and immunomodulatory effects.
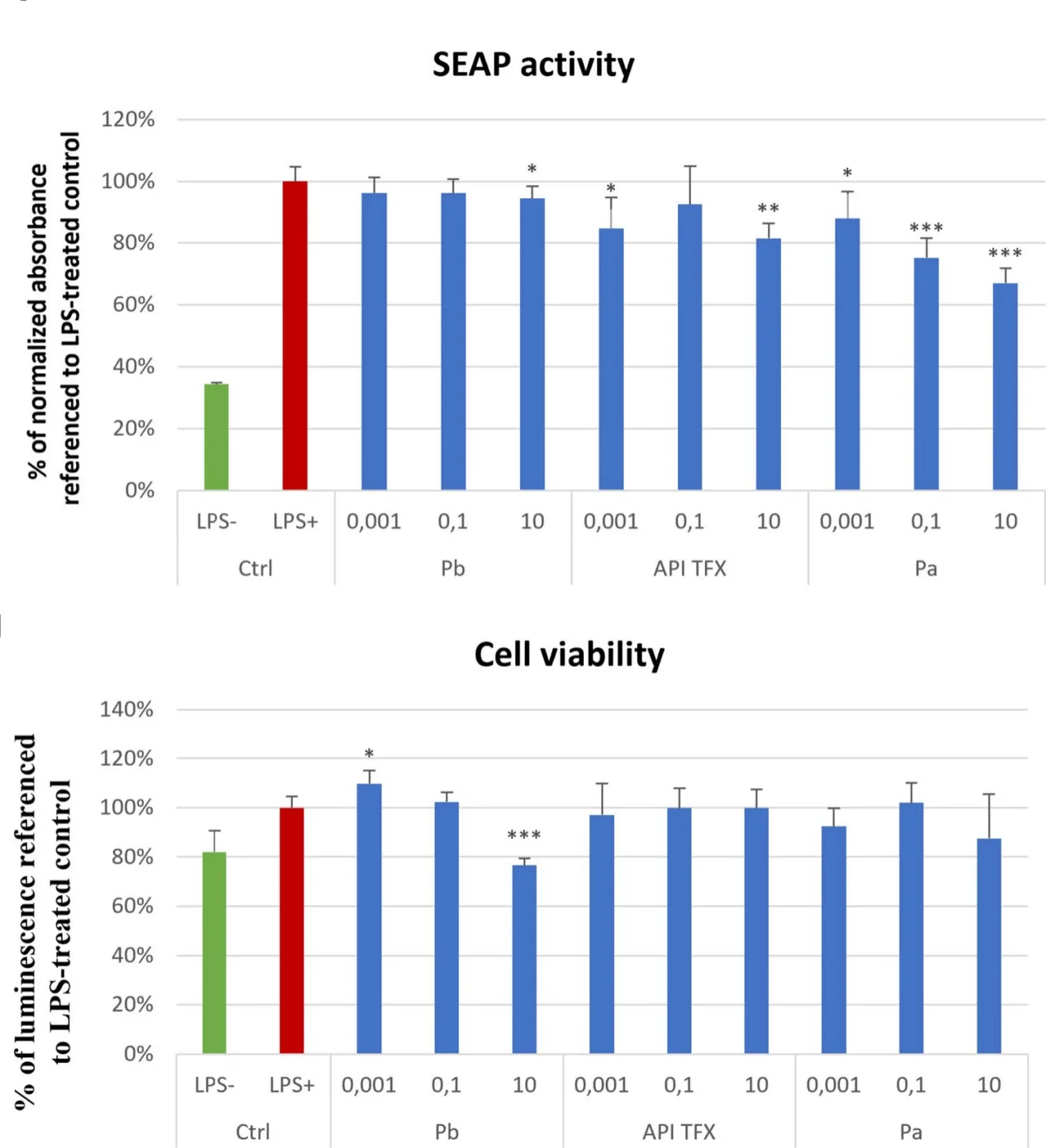
All three thymic peptide preparations attenuated NF-κB activation to some degree, but their efficacy and safety profiles differed markedly. Among them, Pa demonstrated the strongest and most consistent inhibitory effect. NF-κB activity decreased in a clear dose-dependent manner, reaching approximately 67% of the LPS-stimulated control at the highest tested concentration. Importantly, this suppression was not accompanied by reduced cell viability, confirming that the observed effect reflected genuine immunomodulation rather than nonspecific cytotoxicity.
The reference API TFX produced a moderate but statistically significant inhibition of NF-κB activation, consistent with its established immunoregulatory profile. Interestingly, its activity followed a non-linear, U-shaped dose–response curve, a phenomenon commonly observed with bioactive peptides and anti-inflammatory agents.
In contrast, Pb exhibited the weakest NF-κB inhibition and induced significant cytotoxicity at higher concentrations. This finding suggests that although Pb is enriched in low-molecular-weight peptides, its overall composition—or potentially residual inorganic components introduced during filtration—may compromise cellular tolerance.
Implications for Pharmaceutical and Nutraceutical Development of Thymic Peptide Products
This study highlights the critical importance of comprehensive physicochemical profiling in predicting the biological performance of thymic peptide preparations. Pa emerged as the most promising experimental formulation, combining robust NF-κB inhibition with a favorable safety profile. Despite its lower solubility and greater compositional heterogeneity, Pa’s broad molecular weight distribution may enable diverse biological interactions or serve as a source of bioactive fragments upon further processing in vivo.
API TFX remains the benchmark for pharmaceutical thymic peptides, offering high thermal stability and clinically validated immunomodulatory effects. Pb, while rich in small peptides, underscores the risks associated with insufficient purification and highlights the necessity of thorough safety evaluation, particularly for preparations intended for nutraceutical use.
Several limitations warrant consideration. The biological evaluation was confined to a single cell model and a single inflammatory pathway, and peptide sequences were not identified at the molecular level. Future studies incorporating cytokine profiling, additional immune cell systems, and peptidomic analysis using mass spectrometry would further elucidate structure–function relationships and support translational development.
Overall, this work provides a compelling demonstration that the biological activity of thymic peptide preparations cannot be divorced from their physicochemical identity. Rational development of thymic peptide products—whether pharmaceutical or nutraceutical—must be grounded in rigorous analytical characterization coupled with functional validation. As interest in immune-modulating bioactive peptides continues to grow, such integrated approaches will be essential for translating biological complexity into safe and effective applications.
Reference
Besman, M., Matwiejczyk, M., Cywoniuk, P., Mieloch, A. A., Porzucek, F., Rybka, J. D., … & Zambrowicz, A. (2025). Evaluation of the Physicochemical Properties and Potential Immunomodulatory Effects of Bovine Thymic Peptide-Based Preparations. International Journal of Peptide Research and Therapeutics, 31(6), 1-16.https://doi.org/10.1007/s10989-025-10769-0
Abdul Hakim, B. N., Xuan, N. J., & Oslan, S. N. H. (2023). A comprehensive review of bioactive compounds from lactic acid bacteria: Potential functions as functional food in dietetics and the food industry. Foods, 12(15), 2850.https://doi.org/10.3390/foods12152850
Besman, M., Zambrowicz, A., & Matwiejczyk, M. (2024). Review of Thymic Peptides and Hormones: From Their Properties to Clinical Application. International Journal of Peptide Research and Therapeutics, 31(1), 10.https://doi.org/10.1007/s10989-024-10666-y
Oliveira, C. R., de Lima Kaminski, V., de Paula Vieira, R., Ferreira, A., da Silva Olímpio, F. R., Loures, F. V., … & Polonini, H. (2024). Peptides with immunomodulatory properties (Imuno TF®) increase the frequency of the CD8+ T cell population in vitro. Research, Society and Development, 13(8), e10813846597-e10813846597.https://doi.org/10.33448/rsd-v13i8.46597
 LinkPeptide
LinkPeptide