When Binding Is Not Enough: Solving the Delivery Problem in Antisense PNA Research
Antisense Technologies at a Crossroads: When Delivery Limits Discovery
Antisense technologies have become indispensable tools in modern molecular biology and therapeutic research. Modalities such as antisense oligonucleotides (ASOs), small interfering RNA (siRNA), and peptide nucleic acids (PNAs) allow researchers to modulate gene expression with high sequence specificity. Over the past two decades, advances in target discovery, sequence optimization, and chemical stabilization have significantly improved the intrinsic potency of these molecules. Yet despite this progress, one fundamental obstacle continues to limit their broader application: efficient intracellular delivery. 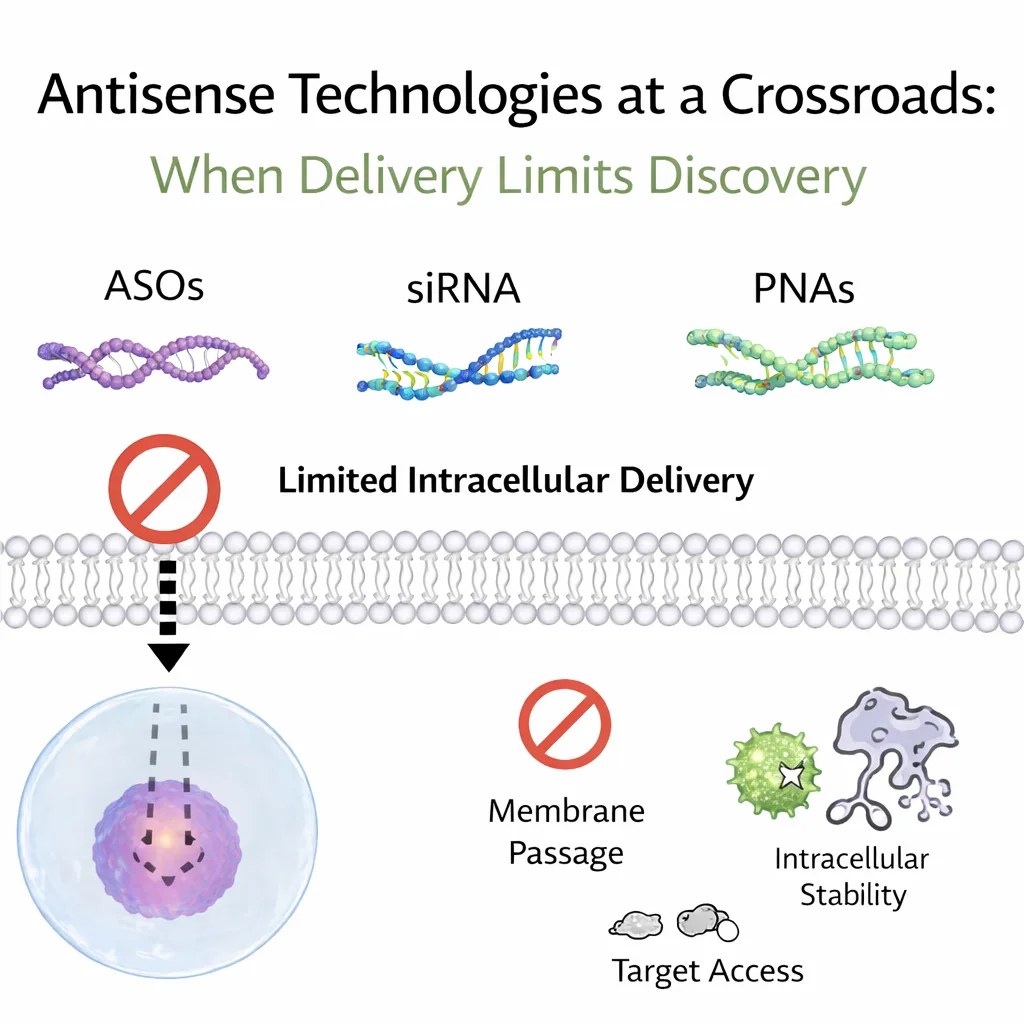
For many antisense platforms, biological performance is constrained less by binding affinity and more by the ability of the molecule to cross the cell membrane, survive the intracellular environment, and reach its intended site of action. PNAs represent a striking example of this paradox. They exhibit exceptional hybridization strength and enzymatic stability, but their neutral backbone severely restricts spontaneous cellular uptake. As a result, PNAs often perform exceptionally well in cell-free systems while showing limited activity in cellular models.
This disconnect has slowed the adoption of PNAs despite their conceptual advantages. Consequently, delivery strategies that can reliably transport PNAs into cells—without excessive complexity or toxicity—are of significant interest to researchers. Recent work exploring cell-penetrating peptides (CPPs) as non-covalent delivery vehicles suggests a practical way forward, reframing PNAs not as delivery-limited curiosities but as viable antisense tools for cellular and translational research.
PNAs Meet CPPs: Matching a Powerful Modality with the Right Delivery Engine
Peptide nucleic acids are synthetic analogues of DNA and RNA in which the sugar–phosphate backbone is replaced by a neutral N-(2-aminoethyl)glycine scaffold. This structural modification eliminates electrostatic repulsion during hybridization, resulting in stronger and more specific binding to complementary DNA or RNA sequences. PNAs are also highly resistant to nucleases and proteases, giving them exceptional stability in biological environments. These properties allow PNAs to form diverse binding modes, including duplexes, triplexes, and strand-invasion complexes, expanding their utility beyond conventional antisense mechanisms.
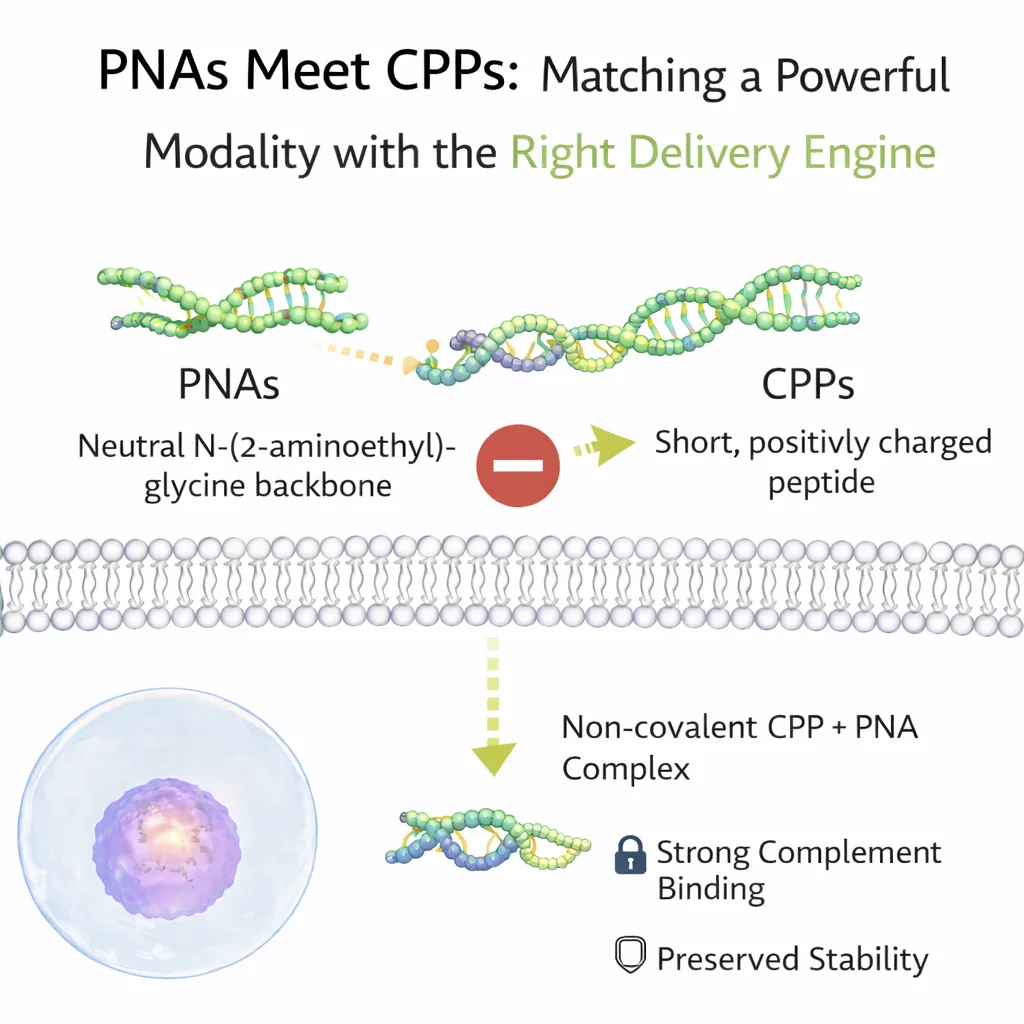
However, the same neutral backbone that confers these advantages also undermines cellular uptake. Without charge-based interactions with the negatively charged cell membrane, PNAs exhibit minimal passive permeability. This limitation has historically necessitated alternative delivery strategies such as electroporation, lipid-based carriers, or covalent conjugation to delivery vectors, each of which introduces its own technical or biological constraints.
Cell-penetrating peptides offer a compelling alternative. CPPs are short peptides, often enriched in basic or amphipathic residues, that can cross cellular membranes while carrying molecular cargo. Depending on their sequence and structure, CPPs may enter cells via direct membrane translocation, endocytosis, or mixed mechanisms. Their modularity, ease of synthesis, and adaptability to diverse cargoes have made them attractive non-viral delivery systems.
Importantly, CPP-mediated delivery does not necessarily require covalent attachment. Non-covalent CPP–cargo complexes can form through electrostatic and hydrophobic interactions, creating transient nanoparticles capable of cellular entry. For PNAs, this approach holds particular promise: it preserves molecular integrity while avoiding the synthetic burden associated with covalent conjugation.
From Concept to Cell: Demonstrating Non-Covalent CPP–PNA Delivery
Recent experimental work has provided a detailed evaluation of non-covalent CPP–PNA delivery in cellular systems. In this study, fluorescently labeled antisense PNAs were designed to target PCSK9 and ACLY—genes central to lipid metabolism and cardiovascular disease research. To promote nuclear accumulation following cellular entry, each PNA was engineered with a C-terminal nuclear localization signal (NLS), ensuring that delivery efficiency could be evaluated at the relevant site of action.
Three CPPs were compared as delivery vehicles: Transportan 10 (TP10), Tat(47–57), and TD2.2. Although Tat and TD2.2 are widely regarded as versatile CPPs, Transportan 10 consistently demonstrated superior performance. TP10 is an amphipathic peptide designed to balance hydrophobic membrane interaction with positive charge, enabling strong yet controlled engagement with the lipid bilayer. Unlike CPPs that rely primarily on endocytosis, TP10 is believed to cross membranes via direct translocation, reducing the risk of endosomal trapping.
Delivery efficiency was confirmed using multiple complementary methods. Fluorescence microscopy revealed robust intracellular accumulation of PNAs only when co-incubated with CPPs, with TP10 producing the strongest signal. Quantitative plate-reader fluorescence measurements corroborated these observations, demonstrating statistically significant increases in intracellular PNA levels across both HeLa and HEK293T cell lines.
To further validate these findings, Western blot analysis and high-performance liquid chromatography (HPLC) with fluorescence detection were employed. Both techniques independently confirmed higher intracellular PNA concentrations in the presence of TP10, providing strong orthogonal support for the delivery mechanism. Notably, simple co-incubation of PNA and CPP was sufficient to achieve these results, underscoring the practicality of the non-covalent approach.
Balancing Efficiency and Usability: Safety, Flexibility, and Experimental Value
Effective delivery must be accompanied by an acceptable safety profile, particularly for applications in translational research. Cell viability was assessed using MTT assays under optimized conditions, revealing no cytotoxic effects associated with PNAs, CPPs, or their combinations. In some cases, mild increases in metabolic activity were observed when PNAs or CPPs were applied individually. Interestingly, these effects were neutralized when both components were combined, suggesting that complex formation may modulate individual bioactivities.
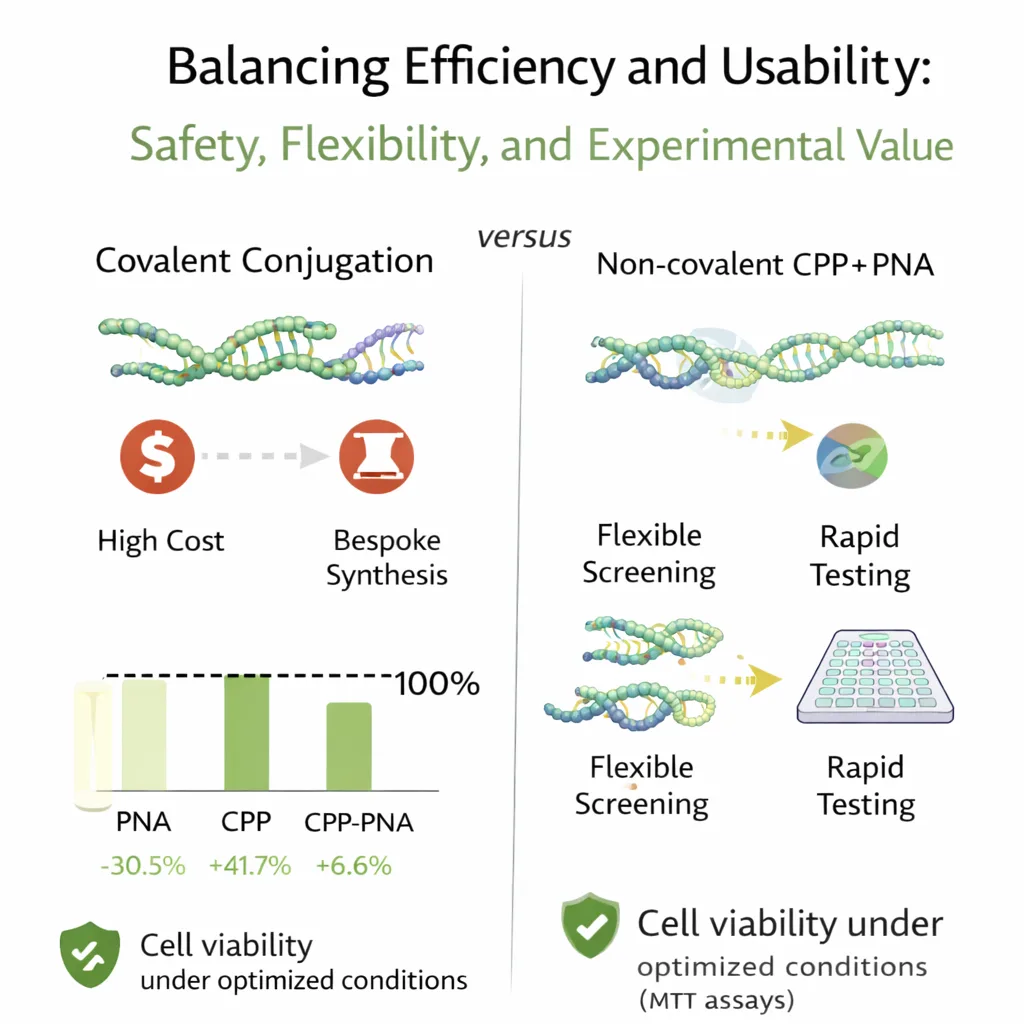
From a practical standpoint, non-covalent CPP–PNA delivery offers several advantages over traditional covalent conjugation strategies. Covalent approaches often provide high efficiency but require bespoke synthesis for each PNA sequence, increasing cost and limiting experimental throughput. In contrast, non-covalent systems enable rapid screening of multiple PNA designs using a single CPP formulation, making them particularly attractive for early-stage research and target validation.
That said, non-covalent delivery is not without limitations. Complex stability and uptake efficiency depend on PNA sequence length and composition, as well as environmental conditions that influence nanoparticle formation. Thorough physicochemical characterization remains essential to ensure reproducibility and scalability. Nonetheless, for many research applications, these trade-offs are outweighed by the flexibility and speed offered by the approach.
Looking Ahead: What CPP-Enabled PNAs Mean for Future Research
The successful delivery of PNAs targeting PCSK9 and ACLY highlights the relevance of CPP-enabled systems to metabolic and cardiovascular research. PCSK9 is already a clinically validated target, with monoclonal antibodies and siRNA-based therapies in use, while ACLY represents an emerging node in lipid metabolism. PNAs delivered via CPPs could complement existing modalities by providing highly stable, sequence-specific tools for mechanistic studies and preclinical screening.
Looking ahead, key challenges include improving cell-type specificity, extending delivery strategies to therapeutically relevant primary cells, and translating in vitro success into in vivo models. Optimization of CPP structure, formulation parameters, and targeting motifs will be critical steps in this process.
Overall, non-covalent CPP–PNA delivery represents a meaningful advance in antisense research. By lowering technical barriers and preserving the intrinsic strengths of PNAs, this strategy repositions them as practical, versatile tools for gene regulation studies and lays the groundwork for future therapeutic innovation.
Reference
Polak, A., Machnik, G., Bułdak, Ł., Wójtowicz, W., Ruczyński, J., Prochera, K., … & Okopień, B. (2025). Application of Cell-Penetrating Peptides (CPP) to Enhance the Efficacy of Antisense Peptide Nucleic Acids (PNA) Molecules Delivery. International Journal of Peptide Research and Therapeutics, 31(6), 114.https://doi.org/10.1007/s10989-025-10776-1
Abifadel, M., Varret, M., Rabès, J. P., Allard, D., Ouguerram, K., Devillers, M., … & Boileau, C. (2003). Mutations in PCSK9 cause autosomal dominant hypercholesterolemia. Nature genetics, 34(2), 154-156.https://doi.org/10.1038/ng1161
Berillo, D., Yeskendir, A., Zharkinbekov, Z., Raziyeva, K., & Saparov, A. (2021). Peptide-based drug delivery systems. Medicina, 57(11), 1209.https://doi.org/10.3390/medicina57111209
Lehto, T., Ezzat, K., Wood, M. J., & Andaloussi, S. E. (2016). Peptides for nucleic acid delivery. Advanced drug delivery reviews, 106, 172-182.https://doi.org/10.1016/j.addr.2016.06.008
 LinkPeptide
LinkPeptide