From Innate Immunity to Therapeutic Potential: Evaluating Recombinant LL-37 Against Resistant Pathogens
Introduction: The Persistent Gap in Antimicrobial Development
The rapid escalation of antimicrobial resistance (AMR) continues to undermine the effectiveness of conventional antibiotics, particularly against pathogens belonging to the ESKAPE group. Despite decades of antibiotic discovery, the clinical pipeline for new antibacterial agents remains limited, while resistance mechanisms evolve at an accelerating pace. This imbalance has renewed interest in alternative antimicrobial strategies that operate beyond classical small-molecule paradigms.
Among these strategies, host defense peptides (HDPs) have re-emerged as promising candidates. These molecules are integral components of innate immunity and exhibit rapid, broad-spectrum antimicrobial activity that is often less susceptible to resistance development. Human cathelicidin LL-37 is one of the most extensively studied HDPs, yet its therapeutic translation has faced persistent challenges related to production, stability, and safety.

Recent work evaluating recombinantly produced LL-37 against a wide panel of clinical isolates provides new evidence that this peptide may be closer to translational relevance than previously assumed. By revisiting LL-37 through the lens of recombinant production and rigorous antimicrobial testing, this study contributes valuable data to the ongoing reassessment of peptide-based antimicrobials.
LL-37 in Innate Immunity: More Than a Membrane-Disrupting Peptide
LL-37 is the only member of the cathelicidin family expressed in humans. It is generated through proteolytic processing of the precursor protein hCAP18, yielding a 37-amino-acid cationic peptide with a pronounced amphipathic α-helical structure. This structural organization underpins its ability to interact with and disrupt microbial membranes, a mechanism fundamentally distinct from those employed by most antibiotics.
However, LL-37 cannot be reduced to a simple membrane-lytic agent. Beyond direct antimicrobial activity, it plays a multifaceted role in immune regulation. LL-37 has been shown to modulate cytokine production, influence chemotaxis, promote wound healing, and interact with pattern recognition receptors. Its expression is inducible and spatially regulated, appearing in high concentrations in tissues such as the skin, respiratory epithelium, gastrointestinal tract, and urogenital system.
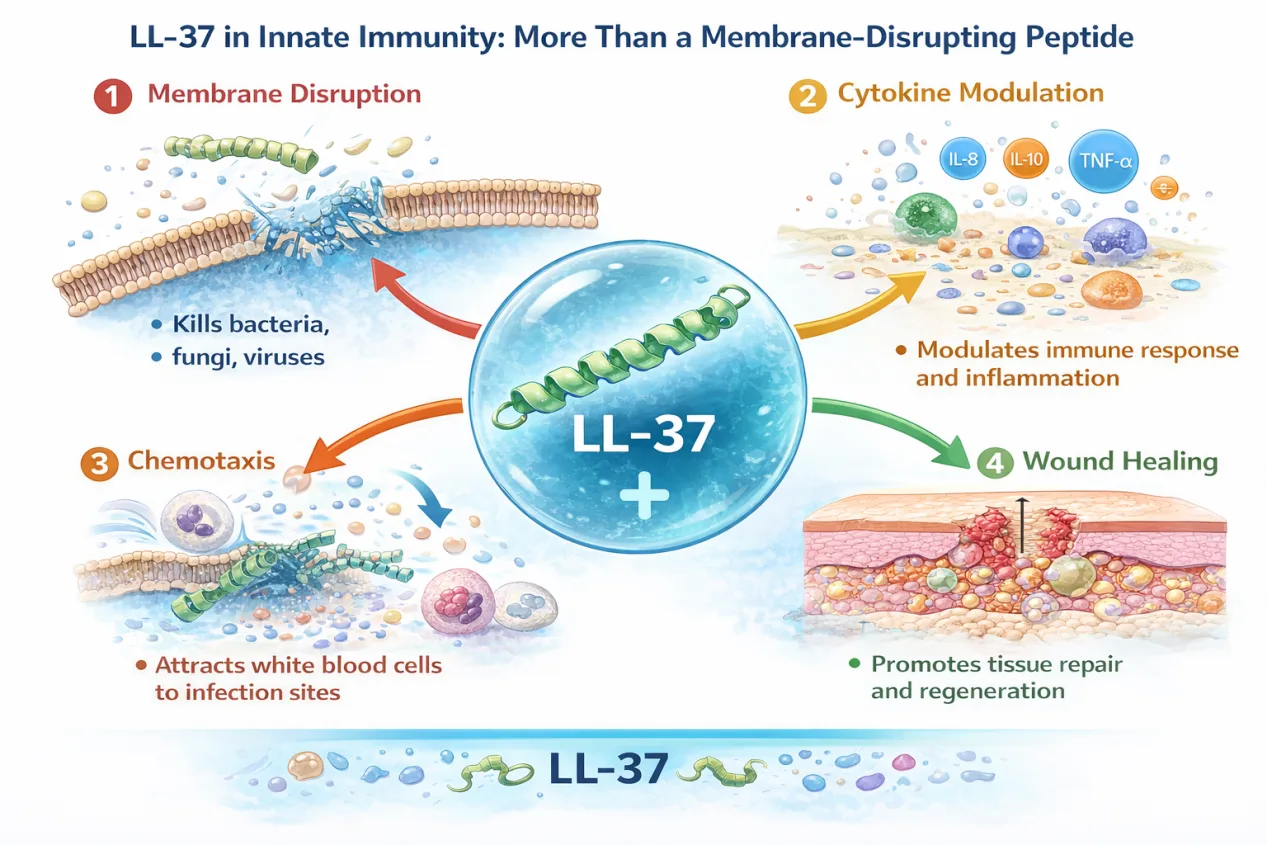
This dual antimicrobial–immunomodulatory profile makes LL-37 particularly attractive from a therapeutic perspective. Rather than acting solely as a bactericidal compound, LL-37 may help reshape local immune responses at infection sites, potentially enhancing pathogen clearance while supporting tissue repair.
Why Recombinant LL-37? Production Challenges and Strategic Advantages
Despite its biological appeal, LL-37 has faced notable barriers to clinical translation. Chemical synthesis of short peptides is technically feasible but often cost-intensive, particularly when scaling beyond laboratory quantities. Additionally, subtle differences in synthesis protocols can influence peptide purity, folding, and activity, leading to wide variability in reported antimicrobial potency.
Recombinant expression offers an attractive alternative, enabling scalable production at reduced cost. However, antimicrobial peptides pose unique challenges in recombinant systems. Their bactericidal nature can harm host cells, and their susceptibility to proteolytic degradation complicates expression and purification.
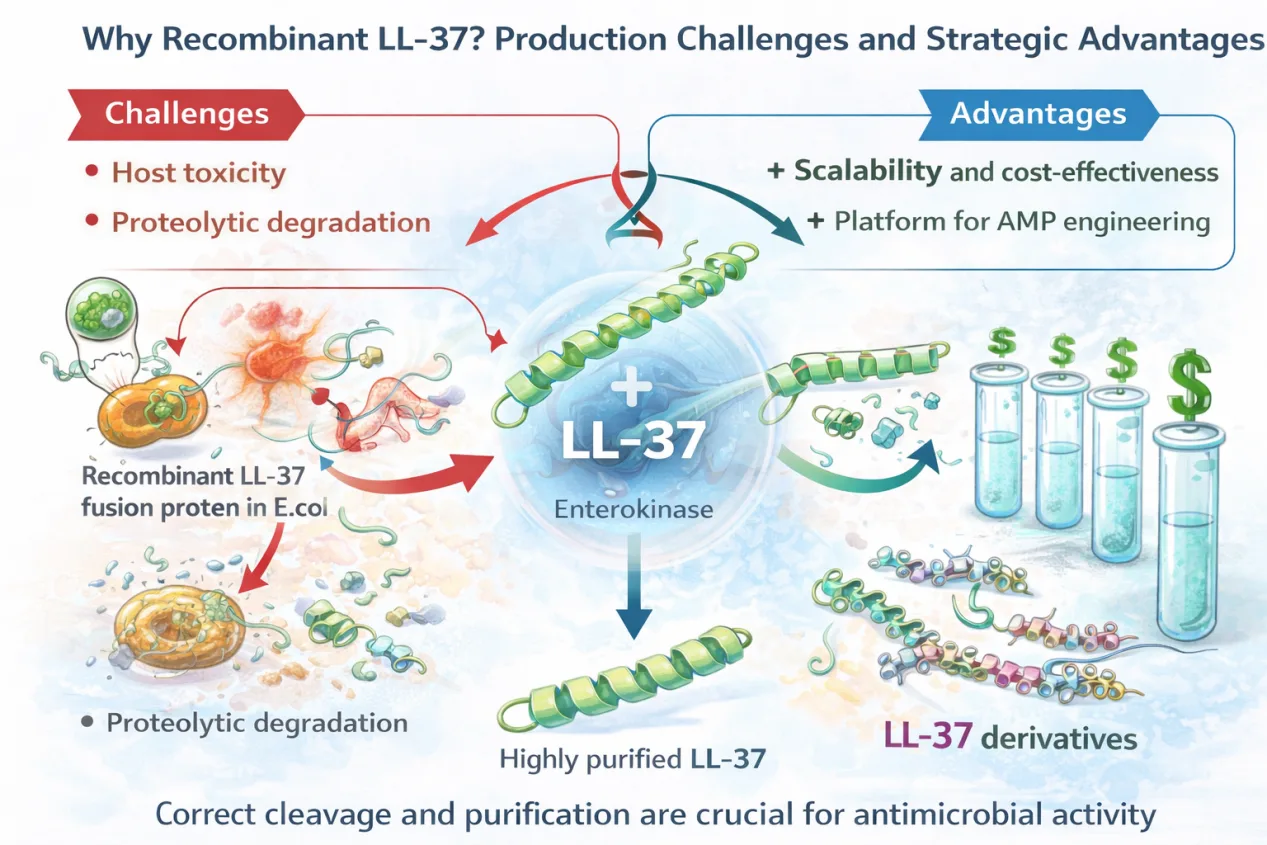
To address these challenges, fusion-protein strategies have become central to recombinant AMP production. In the study under discussion, LL-37 was expressed in Escherichia coli as a thioredoxin (Trx) fusion protein, which mitigates host toxicity and enhances solubility. Subsequent cleavage with human enterokinase allowed precise release of the native LL-37 sequence, followed by rigorous purification steps to ensure high peptide quality.
This production strategy is critical, as accumulating evidence suggests that even minor deviations in peptide structure or processing can significantly alter antimicrobial activity.
Study Design: Testing LL-37 Against Clinical Reality
A key strength of the study lies in its experimental design. Rather than relying solely on laboratory reference strains, the authors evaluated LL-37 against 43 clinical bacterial isolates obtained from patients with diverse infections. This panel encompassed Gram-positive and Gram-negative bacteria, aerobic and obligate anaerobic species, and pathogens associated with urogenital, ocular, auricular, and opportunistic infections.
Antimicrobial activity was assessed using a modified antimicrobial radial diffusion assay. Unlike qualitative diffusion tests, this approach enabled quantitative determination of minimum inhibitory concentrations (MICs) through image-based analysis and calibration. Each bacterial strain was tested in multiple biological replicates, strengthening the robustness of the data.
By prioritizing clinical isolates and quantitative analysis, the study moves beyond proof-of-concept and begins to address how LL-37 might perform under conditions more reflective of real-world infections.
Broad-Spectrum Antimicrobial Activity with Low MIC Values
Across all tested isolates, recombinant LL-37 demonstrated measurable antimicrobial activity. Notably, no strain exhibited detectable resistance under the experimental conditions, reinforcing the idea that natural resistance to LL-37 remains uncommon among bacterial pathogens.
MIC values varied by species and strain, as expected, but several clinically relevant observations emerged. Gram-negative bacteria, including Escherichia coli, Citrobacter koseri, Klebsiella pneumoniae, and Pseudomonas aeruginosa, were inhibited at relatively low LL-37 concentrations. In some cases, MICs fell in the low single-digit microgram per milliliter range, highlighting potent activity against organisms that frequently display multidrug resistance.
Gram-positive bacteria generally required higher LL-37 concentrations, a difference that reached strong statistical significance. This finding is particularly interesting, as Gram-negative pathogens are often more difficult to treat with conventional antibiotics due to their complex outer membrane structures. The observed susceptibility of Gram-negative isolates to LL-37 suggests that membrane-targeting peptides may circumvent some classical resistance mechanisms.
The study also extended LL-37 testing to obligate anaerobes and bacteria associated with bacterial vaginosis, such as Gardnerella vaginalis and Prevotella bivia. These organisms are frequently underrepresented in antimicrobial research, yet they play critical roles in urogenital health and disease. LL-37 exhibited consistent activity against many of these strains, supporting its potential relevance in difficult-to-treat polymicrobial infections.
Contextualizing the Results: Comparison with Existing LL-37 Literature
One of the longstanding challenges in LL-37 research has been the wide variability in reported MIC values across studies. Differences in peptide source, production method, purification, and assay conditions have all contributed to inconsistent findings.
When compared with earlier reports using chemically synthesized or commercially sourced LL-37, the recombinant LL-37 evaluated in this study demonstrated comparable or lower MIC values against many bacterial species. In some cases, the recombinant peptide outperformed previously reported preparations, particularly against Gram-negative pathogens.
The authors reasonably attribute these differences to production-related factors, including the choice of fusion partner, efficiency of protease cleavage, and post-cleavage purification. Importantly, prior studies have shown that residual amino acids or incomplete processing can significantly reduce LL-37 activity. The data presented here reinforce the notion that production strategy is not a technical detail but a determinant of biological performance.
Translational Implications and Remaining Challenges
The findings of this study strengthen the case for LL-37 as a viable antimicrobial candidate, particularly in topical or localized applications. Urogenital infections, wound infections, and infections involving biofilm-forming bacteria may represent especially promising targets.
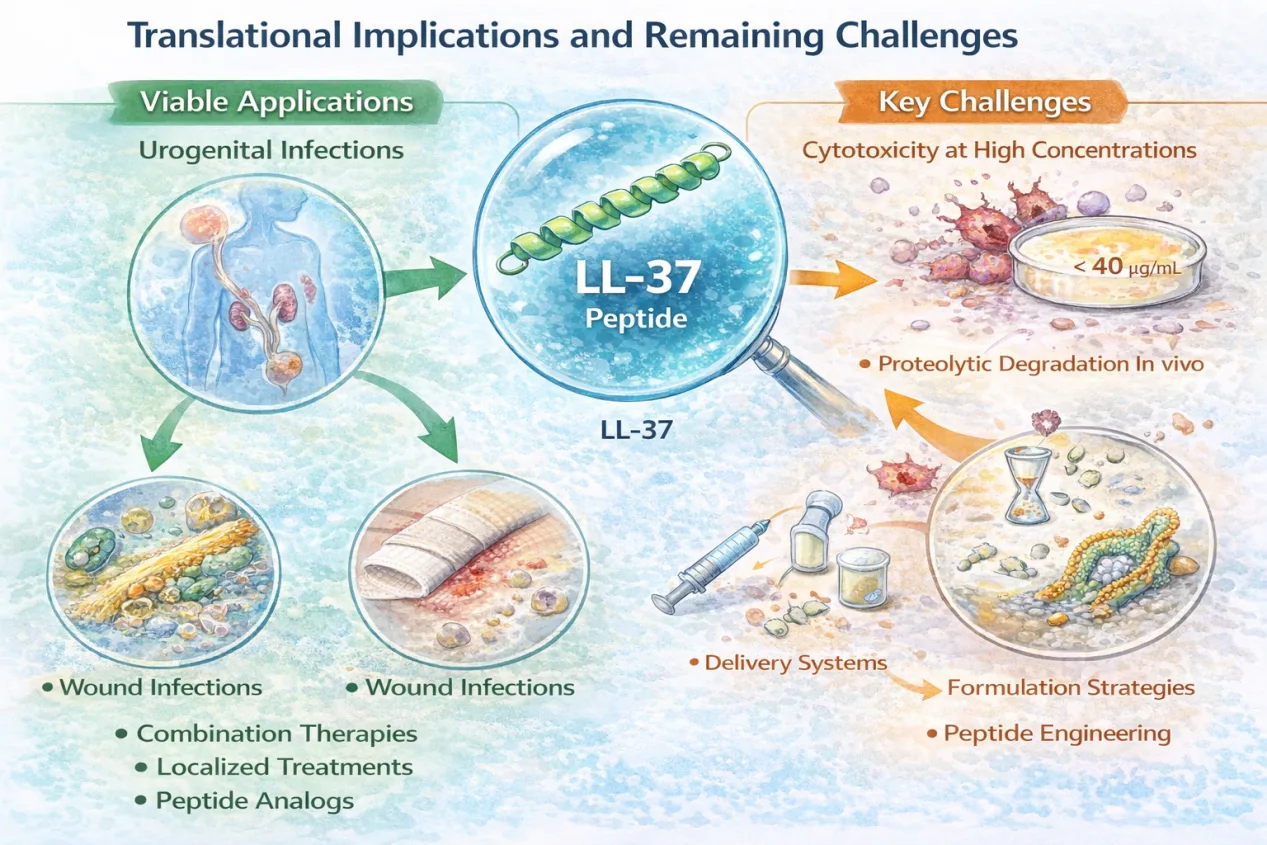
Nonetheless, key challenges remain. LL-37 is known to exhibit cytotoxic effects at higher concentrations, and its susceptibility to proteolytic degradation in vivo must be addressed. These limitations highlight the importance of delivery systems, formulation strategies, and peptide engineering approaches aimed at enhancing stability while preserving antimicrobial activity.
Rather than positioning LL-37 as a direct replacement for antibiotics, its future may lie in combination therapies, localized treatments, or as a scaffold for next-generation peptide analogs.
Conclusion: Re-Establishing LL-37 as a Serious Antimicrobial Candidate
By combining recombinant production with comprehensive testing against clinical isolates, this study provides compelling evidence that LL-37 retains robust, broad-spectrum antimicrobial activity when produced and purified with appropriate rigor. The absence of detectable resistance, coupled with low MIC values against key pathogens, underscores its therapeutic promise.
More broadly, the work illustrates how revisiting well-known innate immune peptides through improved production strategies can yield renewed translational insights. LL-37 may not be a panacea for antimicrobial resistance, but it represents a valuable platform molecule—one that bridges innate immunity and therapeutic innovation and warrants continued investigation in the fight against resistant infections.
Reference
Lukáčová, L., Dusíková, A., Krahulcová, L., Dolník, M., & Krahulec, J. (2025). The Potential of Human Recombinant Cathelicidin LL-37 in the Treatment of Infections. International Journal of Peptide Research and Therapeutics, 32(1), 1.https://doi.org/10.1007/s10989-025-10778-z
Al-Farsi, H. M., Al-Adwani, S., Ahmed, S., Vogt, C., Ambikan, A. T., Leber, A., … & Bergman, P. (2019). Effects of the antimicrobial peptide LL-37 and innate effector mechanisms in colistin-resistant Klebsiella pneumoniae with mgrB insertions. Frontiers in Microbiology, 10, 2632.https://doi.org/10.3389/fmicb.2019.02632
Cabrera‐Aguas, M., Chidi‐Egboka, N., Kandel, H., & Watson, S. L. (2024). Antimicrobial resistance in ocular infection: A review. Clinical & Experimental Ophthalmology, 52(3), 258-275.https://doi.org/10.1111/ceo.14377
Cai, X., Peng, Y., Li, M., Qiu, Y., Wang, Y., Xu, L., & Hou, Q. (2023). Comparative genomic analyses of the clinically-derived Winkia strain NY0527: the reassignment of W. neuii subsp. neuii and W. neuii subsp. antitratus into two separate species and insights into their virulence characteristics. Frontiers in Microbiology, 14, 1147469.https://doi.org/10.3389/fmicb.2023.1147469
 LinkPeptide
LinkPeptide