How Permeation Enhancers Enable Oral Peptide Drugs: A Molecular View from SNAC and Semaglutide
For decades, oral delivery of peptide drugs has been one of the most persistent challenges in pharmaceutical science. Peptides are intrinsically ill-suited for oral administration: they are rapidly degraded by proteases and exhibit extremely low passive permeability across epithelial membranes. While injectable formulations have enabled the clinical success of many peptide therapeutics, oral delivery remains the most desirable route from both patient-compliance and dosing-control perspectives.
Against this backdrop, the success of oral semaglutide, co-formulated with the permeation enhancer salcaprozate sodium (SNAC), represents a landmark achievement. Yet despite its clinical impact, the molecular mechanism by which a large, linear, and polar peptide such as semaglutide crosses a lipid membrane without destroying epithelial integrity has remained poorly understood. A recent Nature Communications study now provides a detailed and experimentally supported answer—one that reframes how permeation enhancers should be understood and designed.
What We Thought We Knew About SNAC-Enabled Absorption
Prior to this work, explanations for SNAC-enabled oral absorption were largely phenomenological. Experimental and clinical studies suggested that SNAC acts locally in the stomach to raise pH, thereby inhibiting pepsin and protecting semaglutide from proteolysis. Additional evidence pointed toward a transcellular absorption pathway, as opposed to paracellular transport, with SNAC proposed to “fluidize” epithelial membranes.
However, these explanations left critical questions unanswered. Membrane fluidization alone does not explain how a long, polar peptide can cross the phospholipid headgroup barrier without catastrophic membrane disruption. Moreover, existing models did not account for the unusually high local concentrations of SNAC near the dissolving tablet or the dynamic physicochemical behavior of SNAC itself. As a result, formulation design remained largely empirical, with limited mechanistic guidance.
A Methodological Leap: Large-Scale Constant-pH Molecular Dynamics
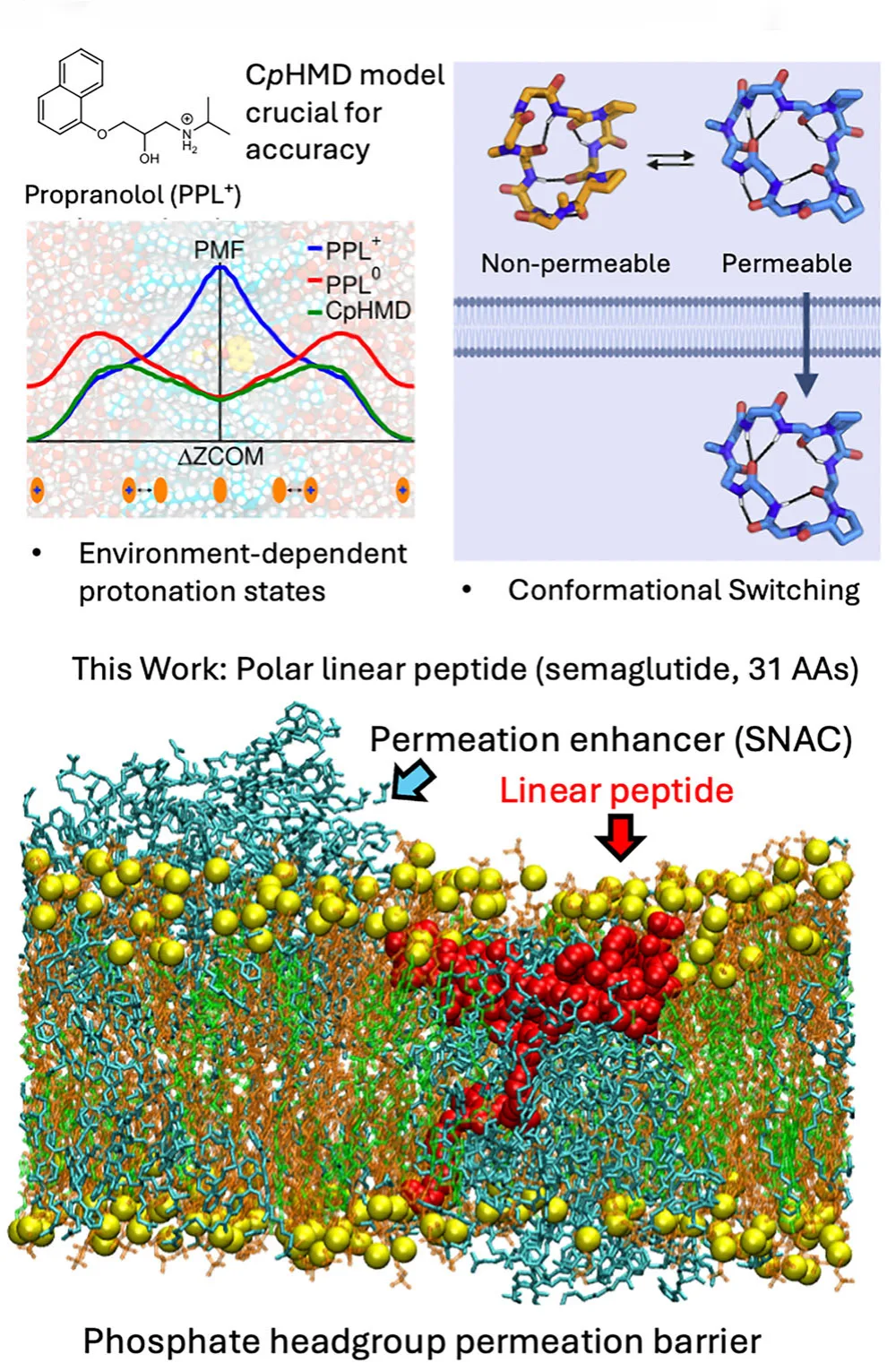
The new study addresses this gap using continuous constant-pH molecular dynamics (CpHMD) simulations, combined with extensive experimental validation. Unlike conventional molecular dynamics, which assigns fixed protonation states to ionizable groups, CpHMD allows protonation states to fluctuate dynamically in response to the local environment.
This distinction is crucial for systems like SNAC–semaglutide–membrane assemblies. Both SNAC and semaglutide contain multiple weakly ionizable functional groups whose pKa values can shift dramatically near membranes, within aggregates, or during peptide–excipient complexation. Fixed-charge models cannot capture these effects.
A major technical advance of the study is the successful simulation of more than 400 ionizable groups simultaneously—including all SNAC molecules and the peptide—at close to the computational efficiency of standard MD. This scale enables, for the first time, a realistic molecular description of permeation-enhancer-assisted peptide transport.
SNAC Is Not an Inert Excipient: Aggregation and Co-Assembly in Solution
One of the earliest insights from the CpHMD simulations is that SNAC is intrinsically self-associating in aqueous environments. At concentrations relevant to oral semaglutide formulations, SNAC rapidly forms dynamic aggregates. More importantly, it co-aggregates with semaglutide, enveloping the peptide in mixed SNAC–peptide clusters.
These computational observations are directly supported by diffusion-ordered NMR spectroscopy (DOSY), which shows increased hydrodynamic radii for both SNAC and semaglutide when mixed together. The data indicate the presence of multiple aggregate populations, suggesting rapid exchange between free and clustered SNAC on the NMR timescale.
Functionally, this co-aggregation helps explain previously reported peptide monomerization in the presence of SNAC. Rather than stabilizing semaglutide as isolated monomers through simple solubilization, SNAC acts as a surfactant-like supramolecular partner, reorganizing peptide–peptide interactions and preparing the peptide for membrane engagement.
Aggregation in Nonpolar Environments and the Origin of Membrane Defects
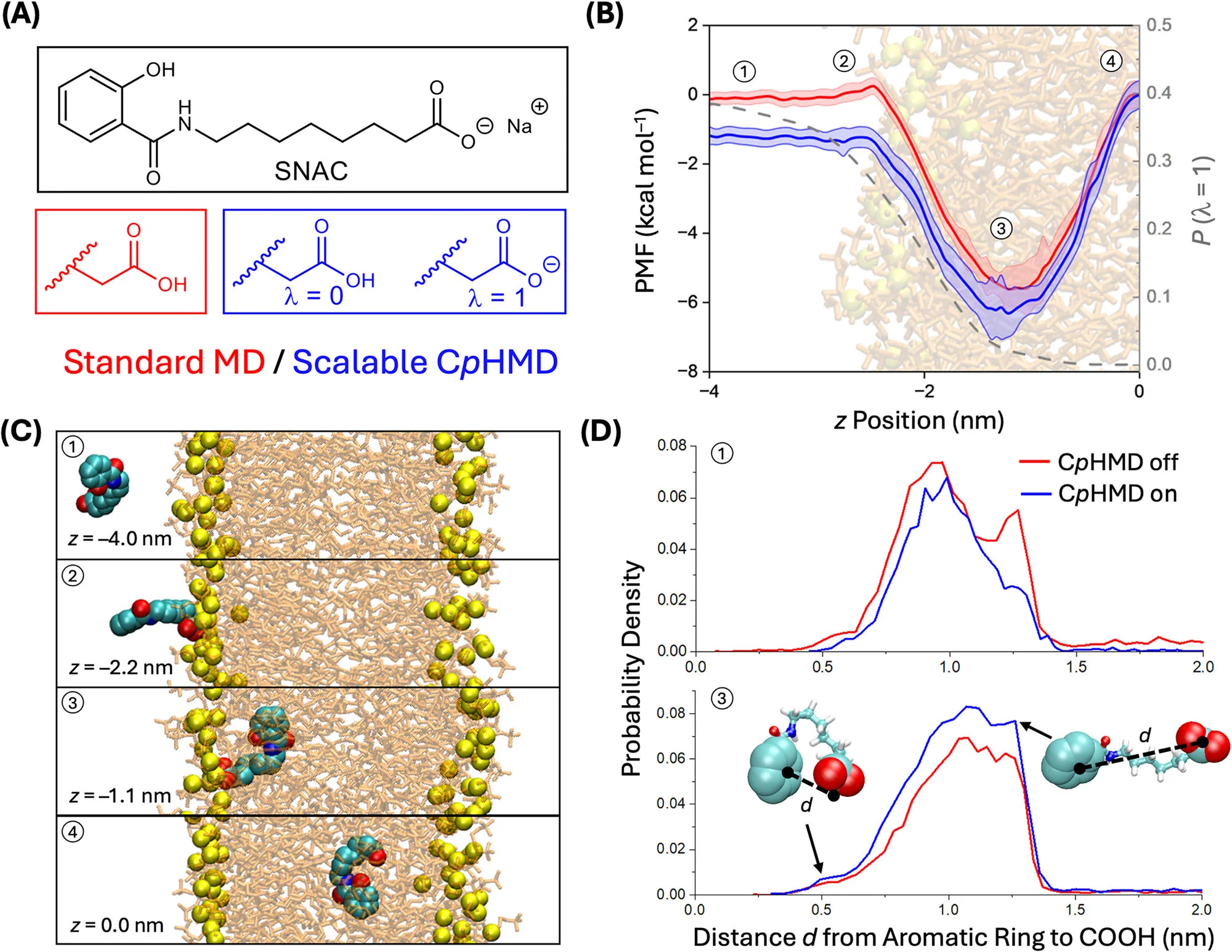
The most unexpected behavior of SNAC emerges in nonpolar environments that mimic the membrane interior. Using a combination of CDCl₃ as a hydrophobic solvent model, CTAB micelles, and explicit lipid bilayers, the study demonstrates that SNAC continues to aggregate even after membrane insertion.
NMR titrations reveal concentration-dependent chemical shift changes consistent with intermolecular hydrogen bonding, while NOESY experiments show π–π stacking interactions between aromatic SNAC moieties. Dynamic light scattering further confirms the formation of large, heterogeneous aggregates.
Crucially, CpHMD simulations show that these aggregates do not form rigid, pore-like structures. Instead, SNAC creates dynamic, fluid-filled clusters within the membrane, displacing lipid headgroups locally but preserving overall bilayer integrity. These transient defects are neither classical pores nor detergent-induced ruptures; they are mobile, self-healing regions enriched in SNAC.
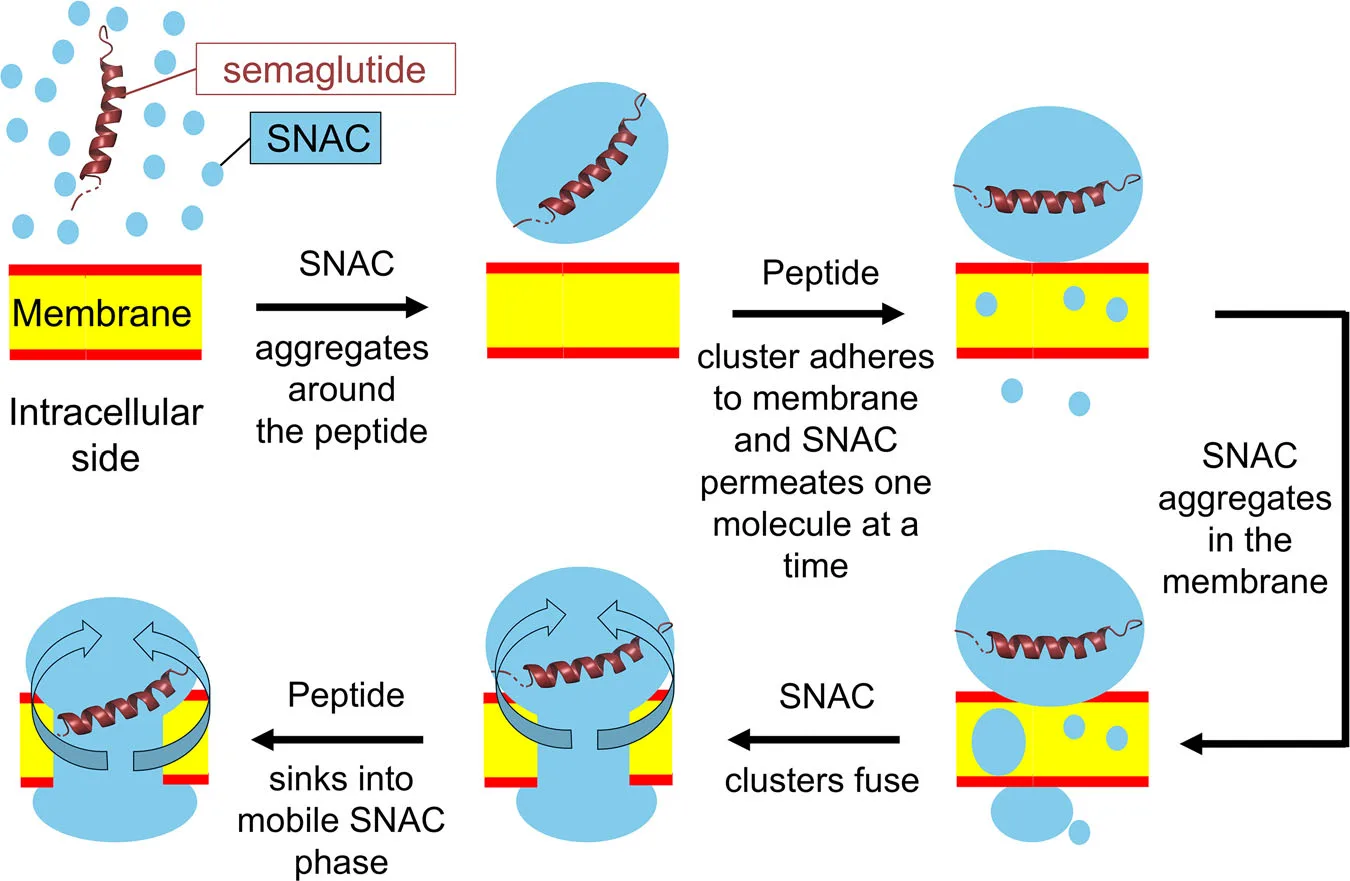
A “Quicksand” Model for Peptide Membrane Permeation
Building on these observations, the authors propose a stepwise permeation mechanism best described as quicksand-like sinking:
- High local concentrations of SNAC near the tablet surface promote extensive SNAC–SNAC and SNAC–peptide aggregation.
- Individual SNAC molecules dynamically insert into the membrane, nucleating SNAC-rich defects that bypass the phospholipid headgroup barrier.
- Solution-phase semaglutide–SNAC aggregates fuse with membrane-bound SNAC clusters, adsorbing the peptide onto the membrane surface.
- The lipidated side chain of semaglutide anchors the peptide into the membrane.
- As SNAC molecules continuously rearrange within the defect, the peptide slowly sinks deeper into the membrane, eventually exiting on the opposite side together with excess SNAC.
Throughout this process, simulations show minimal water leakage and no irreversible membrane damage. The membrane behaves elastically, reorganizing around the moving SNAC–peptide assembly.
This mechanism resolves a long-standing paradox: how transcellular permeation can occur efficiently without compromising epithelial barrier function.
Implications for Oral Peptide and Beyond
The implications of this work extend far beyond semaglutide. First, it reframes permeation enhancers not as simple membrane disruptors, but as dynamic, environment-responsive supramolecular systems. Aggregation, protonation state switching, and mobility are not side effects—they are essential design features.
Second, the study suggests that lipidation of peptides may synergize with permeation enhancers by providing initial membrane anchoring points. This insight may be directly relevant to other lipidated or amphiphilic peptide therapeutics.
Finally, the integration of scalable CpHMD with experimental biophysics establishes a powerful framework for rational formulation design. Rather than screening enhancers empirically, researchers can now interrogate how candidate excipients behave across aqueous, interfacial, and membrane environments at atomic resolution.
Concluding Perspective
The oral delivery of peptide drugs has long been constrained by incomplete mechanistic understanding. By combining state-of-the-art constant-pH simulations with rigorous experimental validation, this study provides the most detailed molecular picture to date of permeation-enhancer-assisted peptide transport.
The emerging “quicksand” mechanism highlights a fundamental shift: successful oral peptide delivery may depend less on overcoming membranes through force, and more on cooperatively reshaping them through dynamic, reversible interactions. As the field moves toward increasingly complex peptide and biologic therapeutics, such mechanism-driven insights will be essential for translating molecular design into clinical reality.
Reference
Colston, K. J., Faivre, K. T., & Schneebeli, S. T. (2025). Permeation enhancer-induced membrane defects assist the oral absorption of peptide drugs. Nature Communications, 16(1), 9512.https://doi.org/10.1038/s41467-025-64891-0
Chen, G., Kang, W., Li, W., Chen, S., & Gao, Y. (2022). Oral delivery of protein and peptide drugs: From non-specific formulation approaches to intestinal cell targeting strategies. Theranostics, 12(3), 1419.https://doi.org/10.7150/thno.61747
Kommineni, N., Sainaga Jyothi, V. G., Butreddy, A., Raju, S., Shapira, T., Khan, W., … & Domb, A. J. (2023). SNAC for enhanced oral bioavailability: an updated review. Pharmaceutical Research, 40(3), 633-650.https://doi.org/10.1007/s11095-022-03459-9
Solis-Herrera, C., Kane, M. P., & Triplitt, C. (2024). Current understanding of sodium n-(8-[2-hydroxylbenzoyl] amino) caprylate (SNAC) as an absorption enhancer: The oral semaglutide experience. Clinical Diabetes, 42(1), 74-86.https://doi.org/10.2337/cd22-0118
 LinkPeptide
LinkPeptide