Cell-Penetrating Peptides: Unlocking the Next Generation of Intracellular Drug Delivery
Abstract
Cell-penetrating peptides (CPPs) have emerged as one of the most versatile tools for transporting therapeutic and diagnostic molecules into cells. These short peptides can cross biological membranes and deliver cargos ranging from nucleic acids and proteins to imaging probes and nanoparticles. Their diversity—spanning natural sequences, engineered hybrids, and fully synthetic designs—allows researchers to fine-tune structure, stability, and targeting capabilities. At the same time, advancing analytical techniques are revealing how CPPs navigate complex uptake pathways, from direct membrane translocation to endocytic routes. Today, CPPs are gaining momentum in applications such as gene therapy, precision oncology, and molecular imaging, with several candidates progressing through clinical trials. Despite remaining hurdles like endosomal entrapment, off-target uptake, and in vivo instability, CPPs continue to evolve, offering tremendous promise for next-generation drug delivery systems. This blog explores their classification, mechanisms, analytical tools, and emerging therapeutic potential.
Why Cell-Penetrating Peptides Are Transforming Drug Delivery
Modern biomedical research faces a familiar but stubborn problem: how do we get therapeutic molecules to the right place inside the cell? Small molecules often slip through membranes on their own, but today’s most promising treatments—peptides, proteins, nucleic acids, and advanced imaging probes—are typically large, charged, and poorly permeable. In many cases, delivery has become just as critical as the drug itself.
Cell-penetrating peptides (CPPs) offer a powerful solution. These are short sequences, usually fewer than 40 amino acids, that can cross cell membranes without causing permanent damage. Just as importantly, they can act as molecular shuttles, carrying a wide variety of cargos, including biologics, fluorescent labels, nanoparticles, and even gene-editing tools. What began with iconic examples like the HIV-1 Tat peptide has grown into a diverse and rapidly evolving family of delivery vehicles.
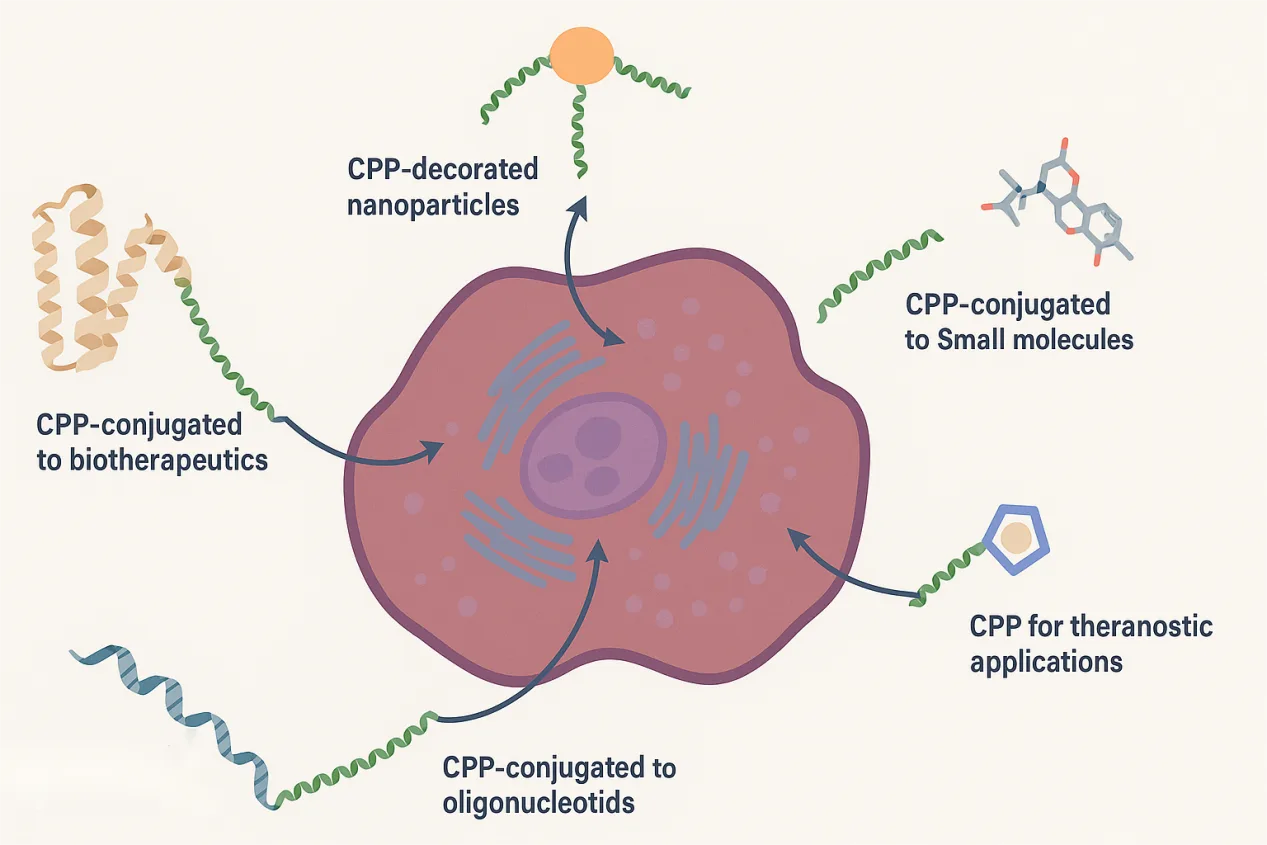
CPPs are attracting intense interest because they combine simple sequences with remarkable functional versatility. By tweaking charge, hydrophobicity, structure, and backbone chemistry, researchers can fine-tune cellular uptake, intracellular routing, and biological activity. At the same time, ongoing work is revealing how these peptides actually enter cells and how we can overcome barriers such as endosomal entrapment and in vivo instability.
In this blog, we’ll explore the current landscape of cell-penetrating peptides: how they are classified, how they cross biological membranes, the methods used to study them, and where they are already making an impact in drug delivery and molecular imaging.
Inside the Diversity of CPPs: Origins, Structures, and Modern Engineering
Although cell-penetrating peptides share a common ability to cross biological membranes, they are remarkably diverse. This diversity makes classification challenging, but it also highlights why CPPs have become such adaptable tools in drug delivery and molecular imaging.
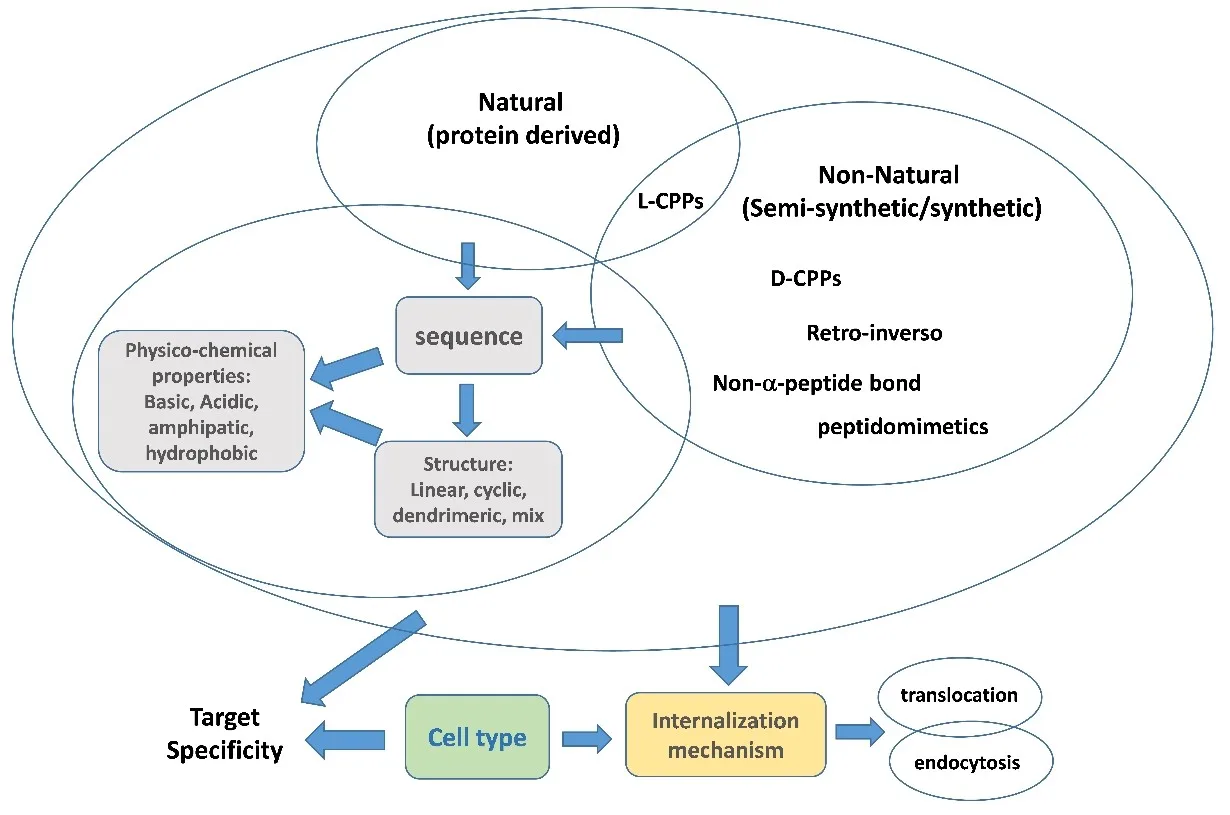
One way to categorize CPPs is by origin. Some come from natural proteins, such as the Antennapedia-derived penetratin or the well-known HIV-1 Tat peptide. Others arise from antimicrobial peptides, venoms, or bacterial toxins—sources that offer built-in membrane activity. A growing number are chimeric or fully synthetic, designed by combining functional segments or by building entirely new sequences optimized for uptake, stability, or specificity.
Another useful layer of classification looks at physicochemical properties.
- Cationic CPPs, rich in arginine and lysine, are the most common because their positive charge helps them interact strongly with negatively charged cell membranes.
- Amphipathic CPPs contain both hydrophobic and hydrophilic regions, often forming helices that insert into lipid bilayers.
- Hydrophobic and even anionic CPPs are emerging as interesting alternatives, expanding the design space and enabling unique modes of entry.
Modern peptide engineering has further broadened what CPPs can do. Techniques like cyclization, stapling, D-amino acid substitution, peptidomimetics, and dendrimeric scaffolds are frequently used to enhance proteolytic stability, prolong circulation time, or achieve more selective intracellular targeting. Researchers are also increasingly developing bioportides—CPPs that not only deliver cargos but also exert their own biological activity inside the cell.
In short, CPPs are no longer defined by one archetypal sequence. Instead, they form a flexible design platform that can be tailored to the needs of cutting-edge therapeutics.
How Do CPPs Enter Cells? Unpacking the Pathways Behind Membrane Crossing
Despite decades of research, the question “How do CPPs enter cells?” still has no single, universal answer. Instead, CPPs use a spectrum of pathways, and the dominant route often depends on the peptide’s sequence, concentration, cargo, and even the cell type being studied. Understanding these mechanisms is essential, because the efficiency of cargo delivery—and ultimately therapeutic success—depends on how CPPs navigate the complex landscape of the cell membrane.
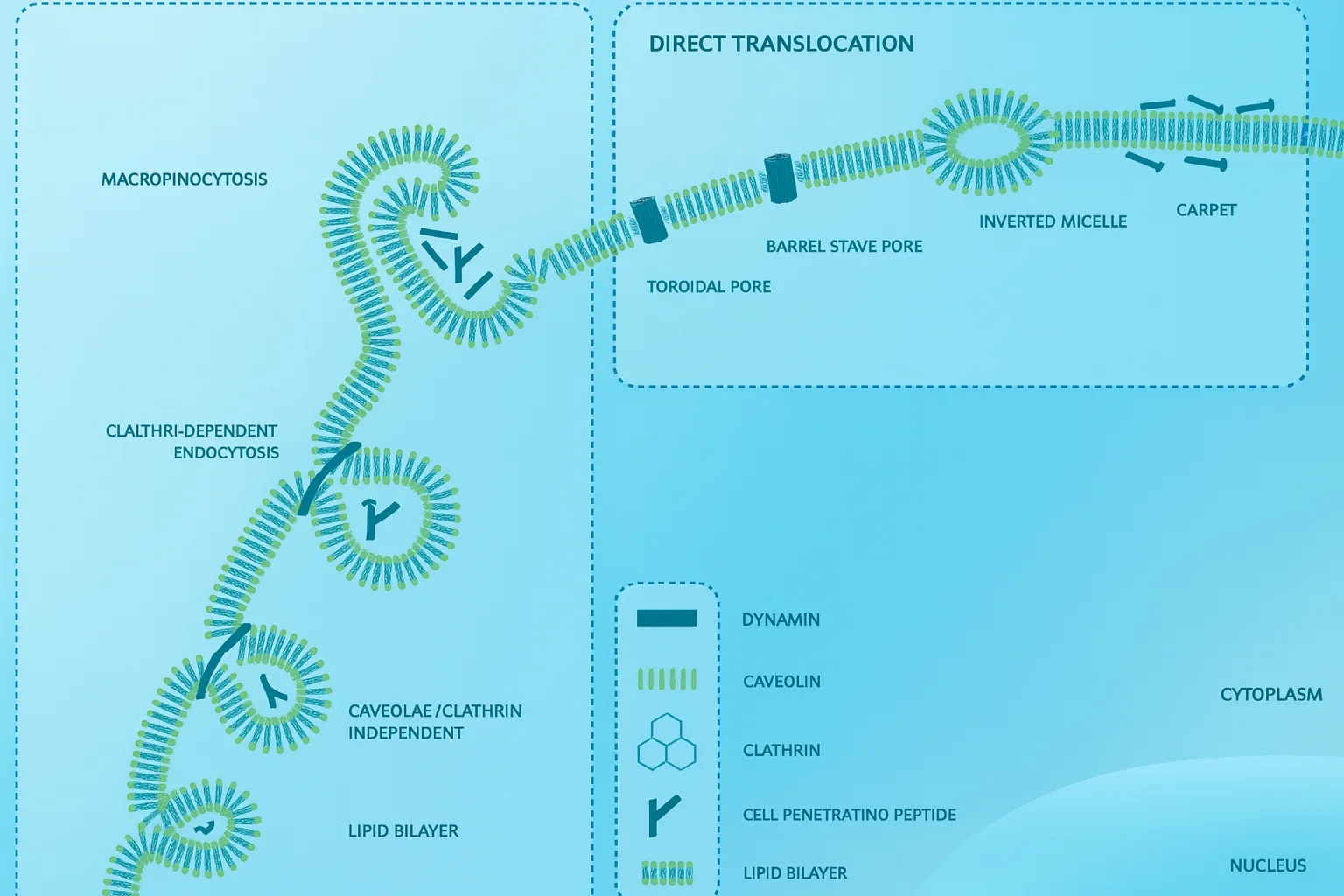
Broadly, CPP uptake falls into two categories: direct translocation and endocytosis.
Direct translocation refers to energy-independent entry, where CPPs cross the plasma membrane without relying on cellular machinery. Several models have been proposed to explain this behavior. Some peptides appear to form inverted micelles that ferry them through the lipid bilayer. Others induce transient pores—either peptide-lined or formed by bending lipids—allowing rapid penetration. A “carpet model” has also been described, in which peptides spread across the membrane surface until reaching a threshold that triggers local disruption. In many cases, the guanidinium groups in arginine-rich CPPs play a central role by forming strong electrostatic interactions with phosphates and sulfates on the membrane.
On the other hand, at lower concentrations or when carrying bulky cargos, CPPs often rely on endocytic pathways. These include macropinocytosis, clathrin-mediated endocytosis, caveolar uptake, and other clathrin-independent processes. Cell-surface molecules such as heparan sulfates and scavenger receptors can help initiate these routes by binding and clustering the peptides.
A major challenge is endosomal entrapment: many CPP–cargo complexes are internalized but remain sequestered inside vesicles. Improving endosomal escape is therefore a major focus in CPP engineering, using pH-responsive motifs, hydrophobic domains, or membrane-lytic segments.
Together, these mechanisms illustrate why CPPs are both powerful and complex—mastering their behavior is key to unlocking their full therapeutic potential.
How Scientists Study CPPs: Technologies Revealing Their Secrets
Because cell-penetrating peptides interact with membranes in complex and often subtle ways, understanding their behavior requires a diverse toolkit. No single analytical method can capture the full story. Instead, researchers combine structural, biochemical, and imaging techniques to quantify uptake, pinpoint intracellular localization, and dissect peptide–membrane interactions at atomic or near-atomic resolution.
Fluorescence-based methods are among the most widely used. Techniques such as confocal microscopy, flow cytometry, and live-cell imaging provide fast, visual confirmation of CPP uptake and distribution. Fluorescence correlation spectroscopy (FCS) and related approaches offer more quantitative insights into diffusion and binding events. However, these methods come with an important caveat: fluorescent labels can sometimes alter the peptide’s properties or detach during processing, leading to misleading signals if not carefully controlled.
To obtain label-free evidence, researchers often turn to mass spectrometry, which can accurately quantify the amount of peptide that truly enters cells versus what remains surface-bound. This distinction is especially important for CPPs, which tend to interact strongly with membranes and extracellular components.
Circular Dichroism (CD) is another essential tool, revealing how CPP secondary structures change when they encounter lipid membranes. Many CPPs shift from disordered states to well-defined helices upon binding—a transition that often correlates with improved uptake or membrane insertion.
For deeper structural insight, NMR spectroscopy and electron microscopy (TEM, SEM, cryo-EM) play crucial roles. NMR can map peptide orientation and interactions within membrane-mimicking environments, while electron microscopy can visualize CPP-nanoparticle complexes, membrane deformation events, and intracellular vesicles.
Together, these techniques build a comprehensive picture of how CPPs behave, guiding the rational design of safer, more effective delivery peptides.
From Lab to Clinic: Where CPPs Are Making an Impact—and What Challenges Remain
With their ability to transport diverse molecules across cellular barriers, cell-penetrating peptides are rapidly making their way from fundamental research into real therapeutic and diagnostic applications. Their versatility is particularly valuable in fields where delivery remains the bottleneck—such as gene therapy, precision oncology, and molecular imaging.
In drug delivery, CPPs have been used to ferry peptides, proteins, antibodies, and even CRISPR components directly into cells. They are especially promising for nucleic acid therapeutics such as antisense oligonucleotides and siRNA, which typically exhibit poor membrane permeability. CPP–PMO conjugates for Duchenne muscular dystrophy represent one of the most advanced examples, showcasing how rational peptide design can dramatically improve intracellular delivery.
The diagnostic space is equally active. Researchers are developing fluorescent, PET, and MRI imaging agents that use CPPs to home in on tumors or inflamed tissues, improving contrast and enabling more accurate detection. Activatable CPPs (ACPPs)—peptides that switch “on” in response to pH changes, specific enzymes, or external triggers—are emerging as powerful tools for targeted imaging and controlled drug release. Some have already entered clinical trials for fluorescence-guided surgery and cancer diagnostics.
Despite this progress, several challenges remain. CPPs must contend with rapid degradation, potential immunogenicity, dose-dependent toxicity, and the persistent issue of endosomal entrapment, which can dramatically reduce the effective dose reaching the cytosol. Achieving cell-type specificity is another ongoing hurdle, as many CPPs naturally enter a wide range of tissues.
Even so, the momentum behind CPP research is unmistakable. With continued advances in peptide engineering, targeting strategies, and mechanistic insight, CPPs are poised to become foundational tools in the next generation of precision therapeutics and molecular imaging platforms.
Reference
Gori, A., Lodigiani, G., Colombarolli, S. G., Bergamaschi, G., & Vitali, A. (2023). Cell penetrating peptides: classification, mechanisms, methods of study, and applications. ChemMedChem, 18(17), e202300236.
https://doi.org/10.1002/cmdc.202300236
Tietz, O., Cortezon-Tamarit, F., Chalk, R., Able, S., & Vallis, K. A. (2022). Tricyclic cell-penetrating peptides for efficient delivery of functional antibodies into cancer cells. Nature chemistry, 14(3), 284-293.
https://doi.org/10.1038/s41557-021-00866-0
Kondo, E., Iioka, H., & Saito, K. (2021). Tumor‐homing peptide and its utility for advanced cancer medicine. Cancer science, 112(6), 2118-2125.
https://doi.org/10.1111/cas.14909
Lim, S., Kim, W. J., Kim, Y. H., Lee, S., Koo, J. H., Lee, J. A., … & Choi, J. M. (2015). dNP2 is a blood–brain barrier-permeable peptide enabling ctCTLA-4 protein delivery to ameliorate experimental autoimmune encephalomyelitis. Nature communications, 6(1), 8244.
 LinkPeptide
LinkPeptide