Cholecystokinin: A Multifunctional Peptide at the Intersection of Metabolism, Neuroscience, and Oncology
Abstract
Cholecystokinin (CCK) is a multifunctional peptide that plays a pivotal role in gastrointestinal regulation, neuroendocrine signaling, metabolic homeostasis, and oncogenesis. Initially identified as a digestive hormone, CCK has since been recognized as a key neuromodulator and cytokine-like peptide with diverse physiological effects. Its bioactivity is mediated through CCK1 and CCK2 receptors, which regulate digestion, satiety, neurotransmission, and tumor growth. Recent advances in peptide therapeutics highlight CCK as a promising target for obesity treatment, neuropsychiatric disorders, and oncology. However, challenges remain in achieving receptor specificity and optimizing drug efficacy. The potential discovery of a novel CCK3 receptor further expands therapeutic opportunities. This review explores the molecular diversity, physiological significance, and emerging pharmacological applications of CCK, emphasizing its evolving role in precision medicine. As research progresses, CCK-based interventions may offer novel strategies for metabolic, neurological, and oncological therapies.
Cholecystokinin as a Multifunctional Peptide in Human Physiology
Cholecystokinin (CCK) was first identified in 1928 as a gastrointestinal hormone responsible for gallbladder contraction and pancreatic enzyme secretion. Over subsequent decades, advancements in peptide biochemistry and neuroendocrinology have revealed CCK to be far more than a digestive regulator. It is now recognized as a multifunctional peptide messenger with a significant role in neurotransmission, satiety signaling, inflammation modulation, and oncogenesis.
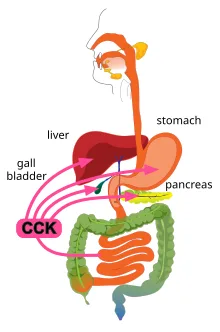
CCK exists in multiple bioactive forms, all derived from a single precursor peptide that undergoes extensive post-translational modifications. It acts primarily through two G-protein-coupled receptors—CCK1 and CCK2—each exhibiting distinct physiological functions. In the gastrointestinal system, CCK modulates digestive enzyme release, gastric motility, and bile flow. In the central nervous system, it functions as a major excitatory neuropeptide, influencing cognitive processes, feeding behavior, and mood regulation. Additionally, CCK exhibits cytokine-like properties, impacting immune function and inflammatory pathways.
Given its diverse physiological roles, CCK has emerged as a critical target for pharmaceutical research. CCK receptor agonists are being investigated for obesity treatment, while antagonists show promise in psychiatric and neurodegenerative disorders. Furthermore, the expression of CCK peptides in tumors highlights their potential as cancer biomarkers and therapeutic targets. The expanding scope of CCK biology underscores its significance in peptide-based drug development.
Molecular Diversity and Tissue-Specific Expression of CCK Peptides
Cholecystokinin (CCK) is encoded by a single gene that gives rise to a family of bioactive peptides through complex post-translational processing. The CCK precursor, preproCCK, undergoes enzymatic modifications, including proteolytic cleavage, O-sulfation, and C-terminal amidation, resulting in multiple biologically active isoforms—CCK-83, CCK-58, CCK-33, CCK-22, CCK-8, and CCK-5. These variants exhibit differential receptor affinities and tissue-specific distributions, influencing a wide range of physiological processes.
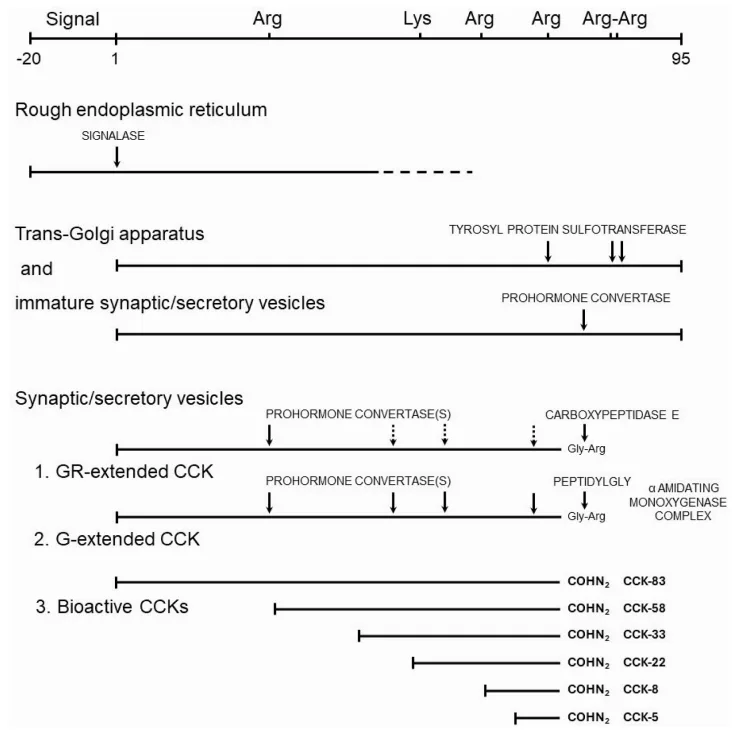
Post-Translational Modifications and Bioactivity
The sulfation of tyrosine residues within CCK peptides is a critical determinant of receptor specificity. Sulfated forms preferentially activate the CCK1 receptor, which is predominantly expressed in the gastrointestinal tract and mediates digestive and satiety-related functions. In contrast, both sulfated and non-sulfated CCK peptides act on the CCK2 receptor, which is widely distributed in the central nervous system and plays a key role in neurotransmission, anxiety regulation, and neuroplasticity. Amidation at the C-terminal phenylalanine is essential for full receptor activation, ensuring the stability and potency of CCK peptides in physiological signaling.
Tissue-Specific Expression and Functional Implications
The expression of CCK peptides follows a highly cell-specific pattern, reflecting their diverse physiological roles:
- Neuronal CCK: The predominant forms in the central and peripheral nervous systems are sulfated CCK-8 and non-sulfated CCK-5. These peptides function as neuromodulators, co-released with classical neurotransmitters such as glutamate and GABA, influencing cognition, mood, and feeding behavior.
- Gastrointestinal CCK: In enteroendocrine I-cells, larger molecular forms (CCK-83, -58, -33, -22) coexist with smaller peptides, modulating pancreatic enzyme secretion, gallbladder contraction, and gastric motility.
- Endocrine and Immune CCK: CCK is also produced in endocrine tissues such as the pituitary, thyroid, adrenal medulla, and testes, where it regulates hormone release and cell proliferation. Additionally, its expression in immune cells suggests a role in modulating inflammatory responses and cytokine signaling.
The molecular diversity and tissue-specific expression of CCK peptides highlight their physiological complexity and therapeutic potential, positioning CCK as a key target in peptide-based drug development.
Physiological Roles and Pathophysiological Implications of CCK Signaling
Cholecystokinin (CCK) exerts multifaceted effects across various physiological systems, acting as a hormone, neurotransmitter, and cytokine-like modulator. Originally recognized for its role in digestion, CCK is now known to influence neuroendocrine function, metabolic homeostasis, immune signaling, and oncogenesis. Dysregulation of CCK pathways has been implicated in a range of diseases, including gastrointestinal disorders, neuropsychiatric conditions, and cancer, underscoring its importance as a therapeutic target.
Endocrine Functions: Regulation of Digestion and Metabolic Homeostasis
CCK plays a critical role in digestive physiology through its endocrine release from enteroendocrine I-cells in the duodenal mucosa. It acts on CCK1 receptors in the pancreas to stimulate enzyme secretion, facilitating protein and lipid digestion. Additionally, CCK triggers gallbladder contraction and relaxation of the sphincter of Oddi, promoting bile release for lipid emulsification. Notably, CCK slows gastric emptying, a function that contributes to postprandial satiety and glucose regulation. The discovery of non-sulfated CCK peptides in intestinal cells suggests additional autocrine and paracrine functions in local gastrointestinal signaling, with potential implications for metabolic diseases such as diabetes and obesity.
Neurotransmitter Role: Modulation of Brain Function and Mood
CCK is one of the most abundant neuropeptides in the mammalian brain, where it acts as an excitatory neuromodulator. CCK peptides are co-released with classical neurotransmitters such as GABA and glutamate, modulating synaptic transmission in the hippocampus, cortex, and amygdala. These interactions influence cognitive processes, anxiety regulation, and stress responses. Aberrant CCK signaling has been implicated in neuropsychiatric disorders, including schizophrenia, depression, and anxiety. CCK2 receptor antagonists are under investigation as potential anxiolytic and antidepressant agents, highlighting the clinical significance of this peptide in neuropharmacology.
Metabolic and Appetite Regulation: Implications for Obesity Treatment
As a key satiety factor, CCK is released in response to nutrient ingestion and acts on vagal afferents via CCK1 receptors to suppress food intake. This function is integral to the gut-brain axis, regulating appetite and energy balance. Preclinical studies suggest that CCK agonists could serve as potential therapeutics for obesity by enhancing satiety signaling. However, challenges remain in optimizing receptor selectivity and overcoming compensatory mechanisms that counteract weight loss.
Immunomodulatory Effects: CCK as a Cytokine-Like Peptide
Recent evidence suggests that CCK peptides participate in immune modulation, functioning as cytokine-like regulators of inflammation. CCK is expressed in mononuclear immune cells, where it influences cytokine release and leukocyte activity. Studies indicate that CCK plays a role in gut-associated immune responses, contributing to both protective and pathological inflammatory processes. The precise mechanisms underlying these effects warrant further investigation, particularly in the context of inflammatory bowel disease and autoimmune disorders.
CCK in Disease States: A Potential Biomarker and Therapeutic Target
Given its widespread expression and physiological influence, CCK has emerged as a potential biomarker and therapeutic target in multiple diseases:
- Neurological Disorders: Altered CCK signaling has been observed in schizophrenia, Parkinson’s disease, and mood disorders. Low cerebrospinal fluid CCK levels in these patients suggest potential diagnostic and therapeutic relevance.
- Gastrointestinal and Metabolic Disorders: Dysregulated CCK secretion has been linked to functional dyspepsia, irritable bowel syndrome, and obesity.
- Cancer Progression and Tumor Biomarker Potential: CCK peptides have been identified in various malignancies, including gastrointestinal, thyroid, and pancreatic tumors. CCK2 receptor overexpression in certain cancers presents opportunities for targeted therapies and diagnostic imaging using radiolabeled CCK analogs.
As the field of peptide therapeutics advances, CCK continues to be a compelling focus for research, with implications spanning endocrinology, neuroscience, immunology, and oncology.
Targeting CCK Receptors: Therapeutic Innovations and Challenges
The diverse physiological functions of cholecystokinin (CCK) and its receptor-mediated signaling have positioned it as an attractive target for drug development. CCK-based therapeutics are being explored across metabolic disorders, neuropsychiatric diseases, and oncology. Understanding the pharmacology of CCK receptors is critical for optimizing the therapeutic potential of this peptide system.
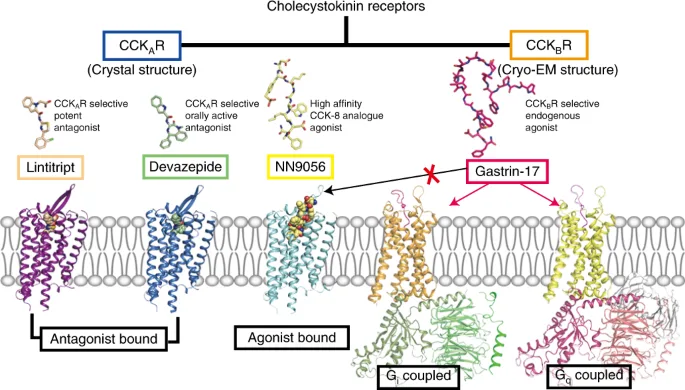
CCK Receptor Pharmacology and Therapeutic Targeting
CCK exerts its effects primarily through two well-characterized G-protein-coupled receptors, CCK1 and CCK2, each with distinct tissue distribution and functional roles. CCK1 receptors are predominantly found in the gastrointestinal tract and vagal afferents, mediating digestive enzyme secretion, gallbladder contraction, and satiety. CCK2 receptors, highly expressed in the central nervous system and various tumors, regulate neuronal excitability, anxiety, and cell proliferation. Recent evidence suggests the possible existence of a third receptor, CCK3 (GPR173), which could offer additional therapeutic avenues. The selective modulation of CCK receptors remains a key challenge in drug discovery, requiring a balance between efficacy and receptor specificity to minimize off-target effects.
Clinical and Preclinical Drug Development
Given its role in appetite regulation, CCK1 receptor agonists have been investigated as potential anti-obesity agents. However, while they demonstrate promising satiety-inducing effects, compensatory mechanisms such as ghrelin secretion often limit long-term efficacy. Combination therapies targeting multiple appetite-regulating pathways may enhance therapeutic success.
Conversely, CCK2 receptor antagonists are being explored for their anxiolytic and antidepressant properties, given CCK’s involvement in stress and mood regulation. Preclinical studies indicate that blocking CCK2 receptors may reduce anxiety symptoms, positioning these compounds as potential alternatives to benzodiazepines with fewer sedative effects.
In oncology, CCK-based diagnostics and therapeutics have gained traction due to the overexpression of CCK2 receptors in various malignancies, including medullary thyroid carcinoma and gastrointestinal tumors. Radiolabeled CCK analogs are being developed for tumor imaging, while receptor-targeted therapies aim to inhibit CCK-driven tumor proliferation.
Challenges and Future Directions
Despite its therapeutic promise, several challenges hinder the clinical translation of CCK-based drugs. Achieving receptor selectivity remains a major obstacle, as cross-reactivity between CCK1 and CCK2 receptors can lead to unintended physiological effects. Additionally, the dynamic and context-dependent nature of CCK signaling complicates the development of universally effective pharmacological agents.
Future research should focus on precision medicine approaches, leveraging advanced molecular modeling and receptor pharmacology to design highly selective CCK modulators. Additionally, the potential role of CCK3 receptors in physiological and pathological processes warrants further investigation. As peptide-based drug development continues to evolve, the strategic targeting of CCK pathways holds great promise for advancing treatments in metabolism, neuropsychiatry, and oncology.
Future Directions: Unlocking the Therapeutic Potential of CCK in Precision Medicine
Cholecystokinin (CCK) has evolved from a classical gut hormone to a multifunctional peptide with critical roles in digestion, neurotransmission, metabolism, immunity, and cancer biology. Its receptor-mediated signaling presents significant therapeutic potential across obesity, neuropsychiatric disorders, and oncology. Despite challenges in receptor selectivity and regulatory complexity, advancements in peptide pharmacology and precision medicine offer promising avenues for CCK-based therapeutics. Future research should focus on refining receptor-specific modulators and exploring novel CCK analogs to optimize clinical efficacy. As the field of peptide-based drug development advances, CCK remains a compelling target for next-generation therapeutics in diverse medical applications.
 LinkPeptide
LinkPeptide