The Future of Immune Checkpoint Inhibition – Small Molecule and Peptide-Based Approaches
Abstract
Immune checkpoint inhibitors have revolutionized cancer immunotherapy, yet monoclonal antibodies (mAbs) face limitations such as poor oral bioavailability, high immunogenicity, and restricted patient response rates. To address these challenges, small molecule and peptide-based checkpoint inhibitors have emerged as promising alternatives, offering enhanced tumor penetration, flexible pharmacokinetics, and lower production costs. Advances in high-throughput screening, structure-based drug design, and targeted protein degradation are accelerating the development of novel small molecule inhibitors. Similarly, peptide-based inhibitors are leveraging computational modeling, phage display, and macrocyclic peptide discovery for enhanced efficacy and specificity. Additionally, nanotechnology-driven drug delivery is optimizing immune checkpoint modulation by improving drug stability, selectivity, and targeted release. This review explores the latest advancements in small molecule, peptide, and nanomedicine-based checkpoint inhibitors, highlighting their potential to overcome the limitations of mAb-based therapies and shape the next generation of cancer immunotherapy.
Beyond Monoclonal Antibodies – The Next Era of Immune Checkpoint Inhibition
The advent of immune checkpoint inhibitors (ICIs) has revolutionized the landscape of cancer immunotherapy by leveraging the body’s intrinsic immune surveillance mechanisms to combat malignancies. Immune checkpoints, such as programmed cell death protein 1 (PD-1) and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), function as critical regulators of immune homeostasis, preventing excessive immune activation and autoimmunity. However, malignant cells exploit these pathways to evade immune detection, necessitating the development of targeted interventions that restore effective antitumor immunity.
Monoclonal antibodies (mAbs) have emerged as the predominant therapeutic modality for immune checkpoint blockade, with FDA-approved agents such as pembrolizumab, nivolumab, and ipilimumab demonstrating clinical efficacy across multiple tumor types. Despite their success, these biologics present several inherent limitations, including poor oral bioavailability, high production costs, and immunogenicity-related adverse effects. Additionally, their prolonged systemic half-life often leads to sustained immune modulation, increasing the risk of immune-related adverse events (irAEs). Furthermore, the therapeutic response to mAbs is restricted to a subset of patients, underscoring the need for alternative checkpoint inhibition strategies that are more broadly applicable and pharmacologically versatile.
To address these challenges, researchers are actively exploring small molecule inhibitors, peptide-based therapeutics, and nanotechnology-driven drug delivery systems as next-generation checkpoint inhibitors. Small molecule inhibitors offer distinct advantages, such as enhanced tumor penetration, improved pharmacokinetic profiles, and reduced immunogenicity, making them promising candidates for overcoming the limitations of mAb-based therapies. Similarly, peptide-based therapeutics, with their ability to disrupt protein-protein interactions with high specificity, represent an emerging class of modulators with potential for improved bioavailability and safety. Concurrently, nanotechnology is being integrated into immunotherapeutic development, providing a platform for targeted delivery, increased drug stability, and enhanced immune activation.
This review will examine the latest advancements in small molecule- and peptide-based checkpoint inhibitors, focusing on their mechanisms of action, advantages over mAbs, and their potential to reshape the future of cancer immunotherapy. By leveraging rational drug design, high-throughput screening, and nanomedicine innovations, these novel approaches may overcome existing limitations and drive the next wave of immunotherapeutic breakthroughs.
Small Molecule Immune Checkpoint Inhibitors: Advancing Precision and Pharmacological Versatility
Monoclonal antibodies (mAbs) have long been the mainstay of immune checkpoint blockade, yet their inherent limitations—high production costs, limited tumor penetration, prolonged systemic half-life, and immunogenicity-related toxicities—have necessitated the exploration of alternative therapeutic modalities. In this context, small molecule inhibitors are emerging as a promising class of immune checkpoint modulators, offering distinct advantages such as oral bioavailability, tunable pharmacokinetics, and enhanced tissue penetration.
Advantages of Small Molecule Inhibitors over mAbs
Unlike mAbs, which are large, structurally complex biomolecules requiring parenteral administration, small molecule inhibitors possess lower molecular weights, facilitating their diffusion across biological barriers and enabling efficient penetration into solid tumors. Furthermore, their chemical properties allow for rational pharmacokinetic optimization, enabling flexible dosing regimens that can mitigate the risk of immune-related adverse events (irAEs) often associated with prolonged checkpoint blockade. Additionally, small molecules circumvent the immunogenicity challenges faced by biologics, reducing the likelihood of anti-drug antibody (ADA) formation, which can compromise therapeutic efficacy.
Drug Discovery Strategies for Small Molecule Immune Checkpoint Inhibitors
The development of small molecule-based checkpoint inhibitors has been driven by high-throughput screening (HTS), computational modeling, and targeted protein degradation strategies. These methodologies have accelerated the identification and optimization of candidate compounds targeting key immune checkpoints such as PD-1, PD-L1, TIGIT, and VISTA.
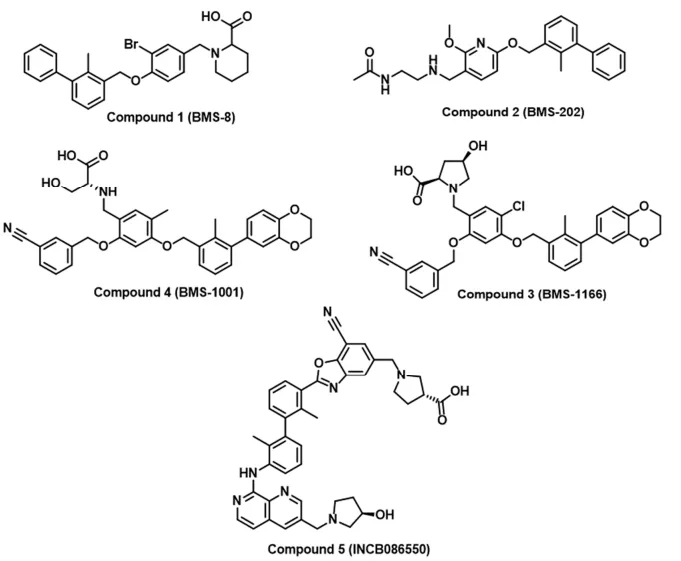
- High-Throughput Screening (HTS):
- HTS enables the rapid screening of large chemical libraries to identify lead compounds that disrupt immune checkpoint interactions.
- This approach is particularly valuable for identifying non-peptidic small molecules that can bind to checkpoint proteins with high specificity and affinity.
- Computational and Structure-Based Drug Design:
- Virtual screening and molecular docking facilitate the rational selection of small molecules with optimal binding properties.
- Advanced computational techniques, such as molecular dynamics simulations and pharmacophore modeling, enhance the precision of hit identification and lead optimization.
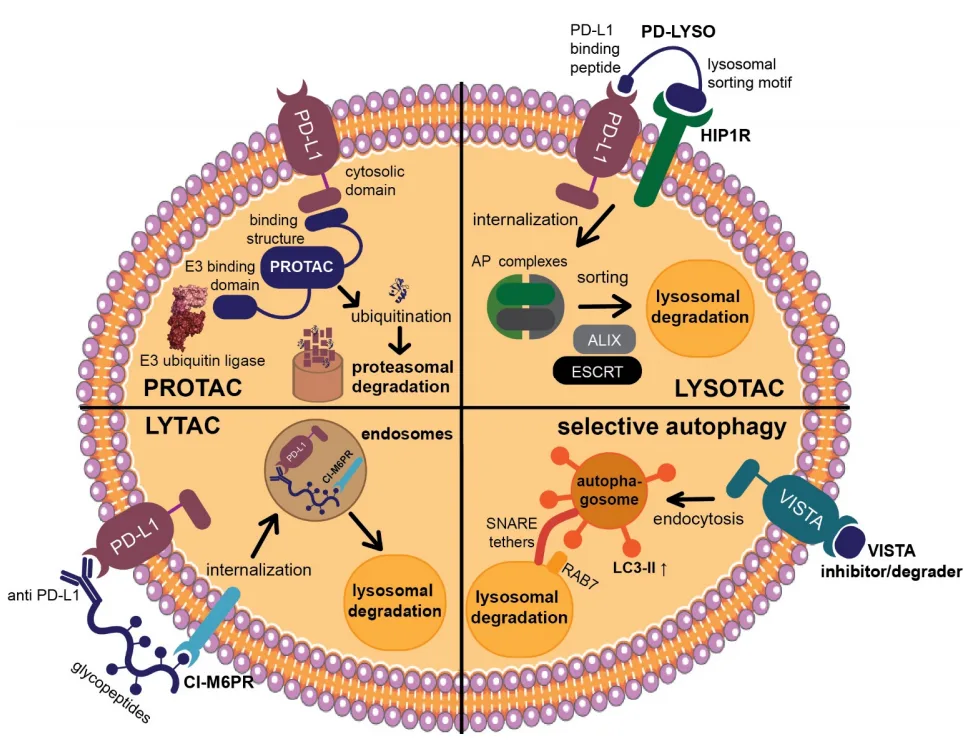
- Targeted Protein Degradation Strategies:
- PROteolysis-TArgeting Chimeras (PROTACs) represent a novel approach to immune checkpoint modulation, leveraging the ubiquitin-proteasome system to selectively degrade checkpoint proteins such as PD-L1.
- Lysosome-targeting chimeras (LYTACs) and autophagy-based degraders offer alternative mechanisms for eliminating immune checkpoint proteins, broadening the therapeutic potential of small molecule-based interventions.
Key Advances in Small Molecule Checkpoint Inhibitors
Several small molecule inhibitors have demonstrated promising preclinical efficacy by disrupting immune checkpoint interactions. Notably:
- PD-1/PD-L1 inhibitors: Multiple pharmaceutical companies have developed small molecules that bind to PD-L1, preventing its interaction with PD-1. Some candidates have shown efficacy in preclinical models and are undergoing clinical evaluation.
- TIGIT inhibitors: Given the increasing recognition of TIGIT as a co-inhibitory receptor in T-cell exhaustion, small molecule inhibitors targeting TIGIT/CD155 interactions are being developed to enhance T-cell activation.
- VISTA inhibitors: As an emerging checkpoint, V-domain Ig suppressor of T-cell activation (VISTA) has been identified as a therapeutic target, with small molecule VISTA inhibitors demonstrating immunomodulatory potential in preclinical models.
Future Directions and Clinical Prospects
The integration of small molecule immune checkpoint inhibitors into clinical oncology is poised to broaden the scope of immunotherapy, offering treatment options for patients who are unresponsive to mAb-based therapies. Combination regimens—integrating small molecules with existing biologics or other immunotherapeutic agents—may further enhance therapeutic efficacy and overcome resistance mechanisms. Additionally, the advent of next-generation inhibitors with improved selectivity and pharmacokinetic profiles will likely accelerate the transition of these agents from preclinical validation to clinical application.
In summary, small molecule inhibitors hold immense promise as next-generation immune checkpoint modulators, with their flexibility, cost-effectiveness, and enhanced pharmacological properties making them attractive alternatives to mAb-based therapies. Continued advances in drug discovery technologies, targeted protein degradation, and combination immunotherapy approaches will be instrumental in realizing their full clinical potential.
Peptide-Based Checkpoint Inhibitors: Harnessing Molecular Specificity for Targeted Immune Modulation
The development of peptide-based immune checkpoint inhibitors represents an emerging frontier in cancer immunotherapy, leveraging the unique ability of peptides to disrupt protein-protein interactions (PPIs) with high specificity. Unlike monoclonal antibodies (mAbs), which face challenges such as poor tissue penetration, prolonged half-life, and high immunogenicity, peptides offer advantages such as lower immunogenicity, synthetic flexibility, and enhanced tumor penetration. Moreover, peptides can be engineered to exhibit high target specificity and affinity, making them attractive candidates for modulating immune checkpoints such as PD-1, PD-L1, CTLA-4, TIGIT, and VISTA.
Peptide Discovery Strategies for Immune Checkpoint Modulation
Peptide drug discovery has evolved through computational, biological, and high-throughput screening methodologies, facilitating the identification of novel inhibitors with improved therapeutic potential.
- Rational Peptide Design:
- Computational modeling and molecular docking techniques enable the design of peptides that mimic endogenous checkpoint ligands or disrupt key interactions.
- Molecular dynamics simulations refine peptide structures to enhance stability, affinity, and pharmacokinetics.
- Phage Display Screening:
- This high-throughput technique enables the selection of peptides with strong binding affinity to immune checkpoint proteins.
- PD-L1-binding peptides, identified through phage display, have demonstrated potent immune activation by blocking PD-1/PD-L1 interactions and restoring T-cell function.
- RaPID (Random Nonstandard Peptide Integrated Discovery) Platform:
- This technology facilitates the identification of macrocyclic peptides with enhanced stability and resistance to proteolytic degradation.
- RaPID-derived peptides targeting CD47-SIRPα and other immune checkpoints have shown promising immunotherapeutic potential.
Key Peptide Inhibitors in Immune Checkpoint Therapy
Several peptide-based checkpoint inhibitors have demonstrated preclinical efficacy:
- PD-L1-binding peptides (e.g., PD-L1Pep-1 and PD-L1Pep-2) have shown high affinity for PD-L1, effectively restoring T-cell activation.
- CTLA-4-targeting peptides have been designed to disrupt CTLA-4/CD80 interactions, enhancing antitumor immune responses.
- D-peptides (e.g., DTBP-3 for TIGIT blockade) exhibit resistance to proteolytic degradation, offering improved in vivo stability.
Challenges and Future Directions
Despite their advantages, peptide therapeutics face challenges such as short half-life, rapid renal clearance, and proteolytic degradation. However, these limitations can be mitigated through:
- Cyclization and backbone modification to enhance stability.
- Nanoparticle-based peptide delivery for sustained bioavailability.
- Peptide-dendrimer conjugates to increase potency and duration of action.
In conclusion, peptide-based immune checkpoint inhibitors represent a highly promising class of therapeutics with the potential to complement or replace mAbs in cancer immunotherapy. Advances in computational design, high-throughput screening, and formulation strategies will be instrumental in accelerating their clinical translation.
Nanotechnology in Immunotherapy: Engineering Smart Drug Delivery and Cellular Modulation
Nanotechnology is emerging as a transformative platform in cancer immunotherapy, addressing key challenges associated with small molecule and peptide-based immune checkpoint inhibitors. By leveraging nanomedicine, it is possible to enhance drug stability, improve tumor selectivity, and optimize pharmacokinetics, thereby overcoming limitations related to poor bioavailability, rapid degradation, and off-target toxicity.
Lipid Nanoparticles for RNA-Based Immunotherapy
Lipid-based nanoparticles (LNPs) have gained significant attention for RNA-based immunotherapy, particularly for enhancing immune checkpoint blockade. Lipid-encapsulated antigen-encoding mRNA has been designed to promote tumor antigen presentation, leading to robust T-cell activation and tumor-specific immune responses. Clinical studies have demonstrated that LNP-formulated mRNA can enhance immune checkpoint inhibition while reducing systemic toxicity compared to conventional biologics.
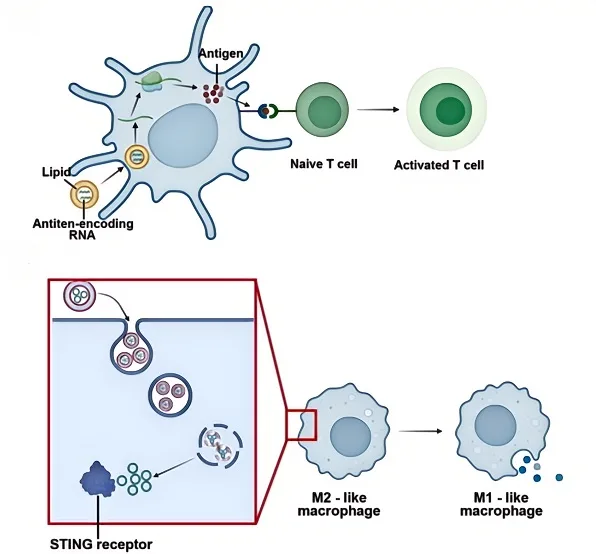
Liposome-Encapsulated STING Agonists
Stimulator of Interferon Genes (STING) agonists play a crucial role in innate immune activation, reprogramming tumor-associated macrophages (TAMs) from an immunosuppressive M2 phenotype to a pro-inflammatory M1 state. However, direct administration of STING agonists often leads to systemic inflammatory responses. Liposome-based delivery systems provide controlled and tumor-selective activation, thereby enhancing antitumor immunity while minimizing toxicity.
Nanoparticle-Based CAR-T Cell Therapy
The application of nanoparticles extends beyond drug delivery to genetic reprogramming of immune cells. Nanoparticle-mediated in vivo CAR-T engineering has been explored as an alternative to traditional ex vivo T-cell modification, offering a less invasive and cost-effective approach to adoptive cell therapy. Nanoparticles encapsulating CAR-encoding genes have demonstrated the ability to convert circulating T cells into tumor-targeting CAR-T cells in vivo, providing a promising next-generation strategy for immune checkpoint modulation.
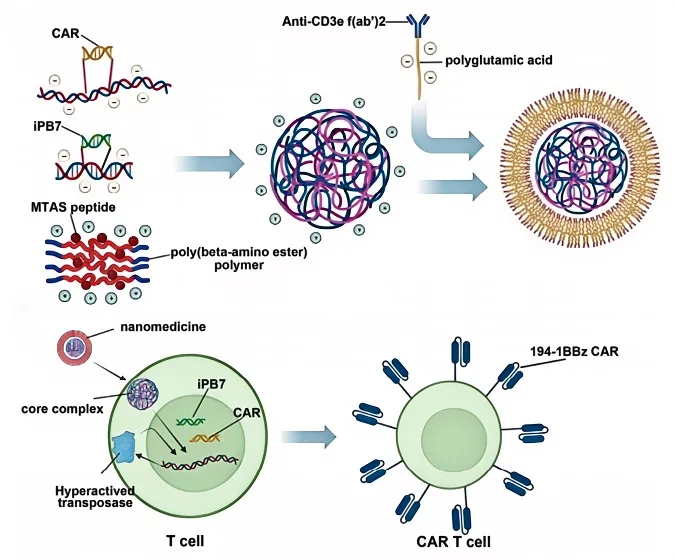
Future Directions in Nanoparticle-Based Immunotherapy
Nanotechnology is expected to play an integral role in the next generation of immune checkpoint inhibitors, enabling controlled drug release, improved tumor targeting, and combination immunotherapy strategies. The integration of nanomedicine with small molecule and peptide-based inhibitors may enhance therapeutic efficacy and pave the way for more precise and patient-specific immunotherapies.
Integrating Next-Generation Checkpoint Inhibitors into the Future of Cancer Immunotherapy
The development of small molecule and peptide-based immune checkpoint inhibitors represents a paradigm shift in cancer immunotherapy, offering advantages over monoclonal antibodies (mAbs) such as improved bioavailability, reduced immunogenicity, and greater tumor penetration. These next-generation therapeutics, coupled with nanotechnology-driven drug delivery systems, have the potential to overcome the limitations of current mAb-based approaches and expand treatment options for a broader patient population.
Small molecules provide flexibility in dosing and pharmacokinetic optimization, while peptides offer specificity in targeting protein-protein interactions. Advances in high-throughput screening, structure-based drug design, and targeted protein degradation strategies are accelerating the identification of novel checkpoint inhibitors. Meanwhile, nanomedicine is enhancing drug stability, selectivity, and delivery efficiency, further optimizing immune checkpoint blockade.
As research progresses, combination approaches integrating small molecules, peptides, and nanotechnology will likely improve response rates, minimize adverse effects, and combat resistance mechanisms. Future efforts should focus on clinical translation, biomarker-driven patient selection, and regulatory advancements to establish these therapies as mainstream immuno-oncology treatments. By harnessing innovative drug discovery platforms, the next decade of immune checkpoint inhibition holds immense potential to reshape the landscape of cancer immunotherapy and improve patient outcomes worldwide.
 LinkPeptide
LinkPeptide