Peptide-Based Immune Checkpoint Inhibitors: The Next Frontier in Cancer Immunotherapy
Abstract
Peptide-based immune checkpoint inhibitors (ICIs) are emerging as a promising alternative to monoclonal antibodies and small-molecule inhibitors in cancer immunotherapy. These peptides effectively disrupt immune checkpoint interactions, such as PD-1/PD-L1, TIGIT/PVR, and LAG-3/MHC-II, to restore antitumor immune responses. Recent advancements in peptide engineering, including cyclization, D-amino acid incorporation, and peptide-drug conjugates (PDCs), have improved their stability, bioavailability, and therapeutic potential. Additionally, dual-function peptides that simultaneously target multiple immune pathways are expanding the scope of peptide therapeutics. Despite challenges in enzymatic degradation and oral delivery, innovative nanoparticle formulations and chemical modifications are addressing these limitations. Peptide-based ICIs offer enhanced selectivity, better tumor penetration, and ease of synthesis, making them a compelling alternative for next-generation cancer immunotherapy. With continued research and clinical validation, peptide therapeutics could play a pivotal role in reshaping immuno-oncology.
Peptide-Based Immunotherapy: A New Era in Cancer Treatment
Cancer immunotherapy has revolutionized oncology by leveraging the immune system to target malignant cells. Unlike chemotherapy or radiotherapy, which directly kill tumor cells, immunotherapy enhances immune responses to eliminate cancer. A major breakthrough in this field is the development of immune checkpoint inhibitors (ICIs), which block inhibitory pathways that suppress immune activation. Among them, PD-1/PD-L1 inhibitors have demonstrated significant clinical success in multiple cancer types.
Despite their effectiveness, monoclonal antibody-based ICIs have notable limitations, including high molecular weight, long half-life, high production costs, and potential immune-related toxicities. Small-molecule inhibitors, although easier to manufacture, often lack specificity and can cause off-target effects.
Peptide-based therapeutics have emerged as a promising alternative, offering advantages such as high selectivity, efficient tumor penetration, and ease of synthesis. However, their clinical development is hindered by enzymatic degradation and poor oral bioavailability, requiring innovative formulation strategies.
This article explores peptide immune checkpoint inhibitors, detailing their mechanisms, advantages, and recent advancements. It will also discuss engineering strategies such as cyclization, D-amino acid incorporation, and drug conjugation to enhance peptide stability and therapeutic efficacy. With continuous innovation, peptide-based ICIs may overcome the limitations of current immunotherapies, offering a next-generation approach to cancer treatment.
How Peptide Inhibitors Work: Mechanisms and Key Advantages
Peptides are emerging as a powerful class of immune checkpoint inhibitors (ICIs) due to their ability to disrupt protein-protein interactions (PPIs) critical for immune suppression in tumors. Unlike monoclonal antibodies, which function through prolonged receptor blockade, peptide inhibitors can selectively target immune checkpoints with greater flexibility and tunability.
The most well-characterized immune checkpoint is the PD-1/PD-L1 axis, where tumor cells exploit PD-L1 expression to suppress T-cell activity. Peptide inhibitors designed through high-throughput screening or structure-based design can effectively disrupt this interaction, reactivating T-cell responses against tumors. Additionally, peptides have been developed to target alternative immune checkpoints such as TIGIT/PVR, LAG-3/MHC-II, and VISTA/PSGL-1, expanding their therapeutic potential beyond PD-1/PD-L1 blockade.
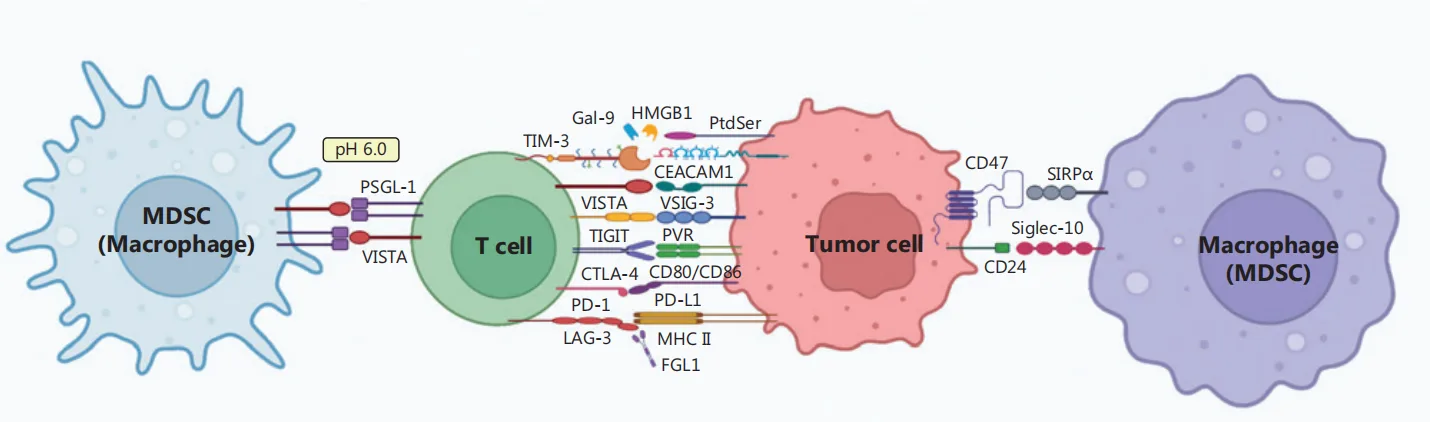
Peptides offer several advantages over monoclonal antibodies and small molecules. Their high selectivity minimizes off-target effects, while their small size allows for better tumor penetration compared to large antibody drugs. Furthermore, peptides are easier and more cost-effective to synthesize, enabling rapid development and optimization.
Despite these advantages, peptide drugs face two major challenges: enzymatic degradation and poor oral bioavailability. Peptides are susceptible to proteolytic enzymes in the body, leading to rapid clearance and reduced efficacy. To address this, researchers have developed strategies such as peptide cyclization, incorporation of D-amino acids, and conjugation with stabilizing carriers. Additionally, new formulation approaches, including hydrogels and nanoparticle delivery systems, are improving peptide stability and bioavailability.
With these advancements, peptide-based ICIs are positioned to become a versatile and effective modality in cancer immunotherapy, offering a promising alternative to traditional antibody-based therapies.
Next-Generation Peptide ICIs: Innovations and Clinical Prospects
Recent advancements in peptide-based immune checkpoint inhibitors (ICIs) have led to the development of several promising candidates targeting PD-1/PD-L1 and alternative immune checkpoint pathways. These novel peptides are designed to enhance stability, improve efficacy, and expand therapeutic applications beyond conventional monoclonal antibodies.
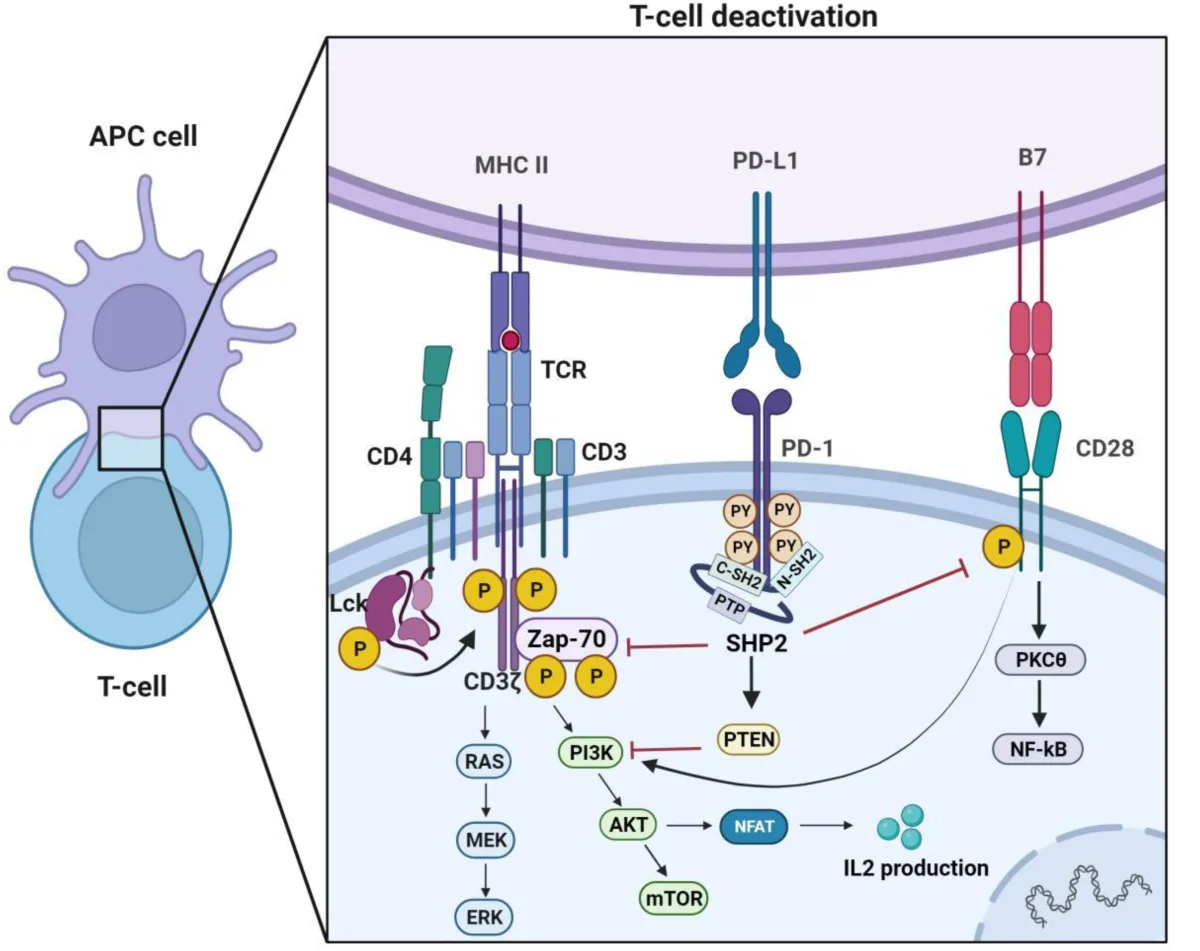
One notable example is BMS-986189, a macrocyclic peptide developed by Bristol-Myers Squibb. This peptide targets PD-L1 and has demonstrated promising results in early-phase clinical trials, highlighting the feasibility of peptide-based checkpoint inhibition. Additionally, DPPA-1, a fully D-amino acid peptide, has been engineered to resist enzymatic degradation while effectively blocking PD-L1. Similarly, OPBP-1, another PD-L1-targeting peptide, has been formulated using trimethyl chitosan hydrogel, enhancing its oral bioavailability and extending its half-life. These innovations mark a significant step toward oral peptide-based cancer immunotherapy, a major improvement over injectable antibodies.
Beyond PD-1/PD-L1, researchers have designed peptide inhibitors targeting alternative immune checkpoints that contribute to tumor immune evasion. DTBP-3 is the first D-peptide inhibitor that blocks the TIGIT/PVR interaction, an emerging immunosuppressive pathway. This peptide has shown efficacy in both PD-1-responsive and PD-1-resistant cancer models, demonstrating its potential as a complementary therapy. Another promising candidate is C25, a cyclic peptide that disrupts the LAG-3/MHC-II interaction, leading to enhanced CD8+ T cell activation and reduced regulatory T cells (Tregs) in the tumor microenvironment.
Peptides are also being developed to target macrophage-associated immune checkpoints, such as CD47/SIRPα and CD24/Siglec10, which regulate the “don’t eat me” signal and prevent immune clearance of tumors. Pep-20 and its D-amino acid variant pep-20-D12 have been shown to activate tumor-associated macrophages (TAMs) while avoiding toxicity to red blood cells, overcoming a major limitation of antibody-based CD47 inhibitors.
These advancements highlight the growing therapeutic potential of peptide ICIs. With improvements in peptide engineering, stability, and delivery, these drugs are poised to play a crucial role in next-generation cancer immunotherapy.
Expanding Peptide Therapy: Dual-Function Peptides and Peptide-Drug Conjugates
The evolution of peptide-based immune checkpoint inhibitors (ICIs) has expanded beyond single-target therapies, leading to the development of dual-function peptides and peptide-drug conjugates (PDCs). These innovations aim to enhance immune activation, improve pharmacokinetics, and maximize antitumor efficacy while overcoming current limitations in immunotherapy.
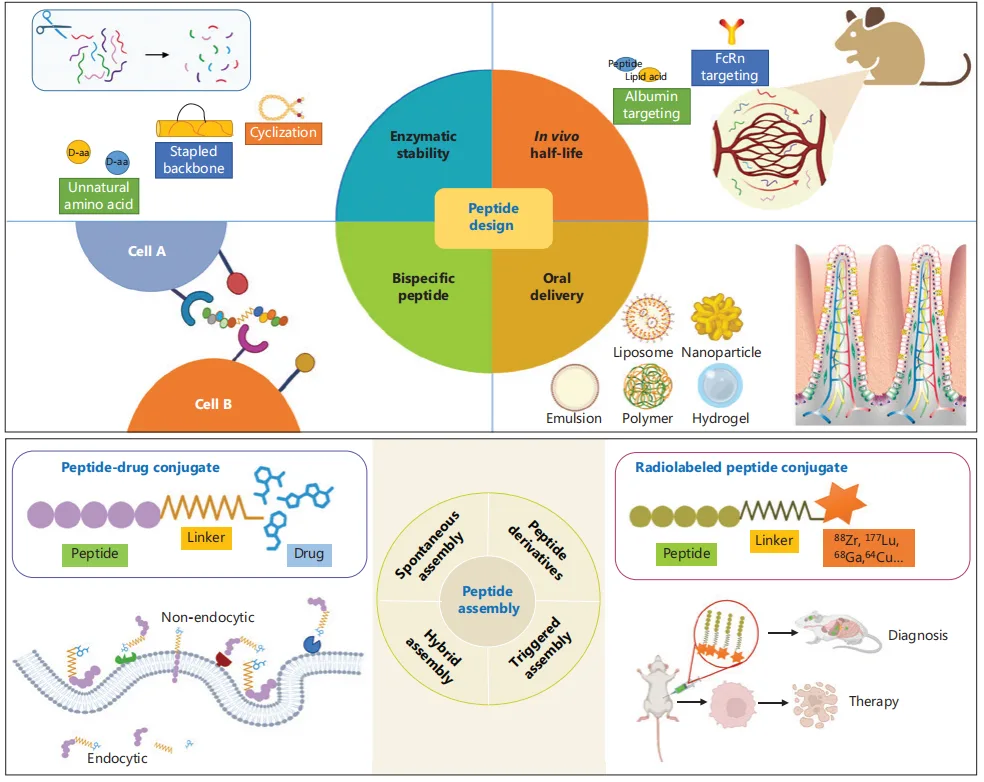
Dual-Function Peptides: A New Paradigm in Immunotherapy
Dual-function peptides, akin to bispecific antibodies, are designed to simultaneously target multiple immune checkpoints or complementary pathways. A key example is Pal-DMPOP, a chimeric peptide that combines PD-1/PD-L1 blockade with CD47/SIRPα inhibition, thereby activating both T cells and macrophages. This dual engagement enhances antitumor immunity through complementary mechanisms, leading to a more robust and sustained immune response.
Another innovative strategy involves targeting tumor vasculature in addition to immune modulation. DSPOGS, a long-acting peptide conjugate, combines PD-1/PD-L1 blockade with anti-angiogenesis effects, effectively countering immune evasion and tumor-driven neovascularization. Such multifunctional peptides represent a promising alternative to combination therapy with separate agents.
Peptide-Drug Conjugates (PDCs) and Nanoparticle Delivery
Beyond dual-function peptides, PDCs are being developed to enhance the stability, targeting, and therapeutic efficacy of peptide ICIs. In contrast to antibody-drug conjugates (ADCs), which rely on toxin-based payloads, PDCs utilize small-molecule inhibitors, chemotherapy agents, or immunomodulatory compounds to amplify their effects.
Recent advancements in self-assembling nanoparticles have further improved peptide drug delivery. For example, peptides conjugated with radiosensitizers or Toll-like receptor (TLR) agonists have been formulated into nanoparticle systems that enhance both innate and adaptive immunity. Such approaches increase drug accumulation at tumor sites, reduce systemic toxicity, and optimize treatment efficacy.
With ongoing innovations in peptide engineering, drug conjugation, and targeted delivery, dual-function peptides and PDCs are poised to play a crucial role in the next generation of cancer immunotherapy.
The Future of Peptide ICIs: Overcoming Challenges and Unlocking New Potential
Peptide-based immune checkpoint inhibitors (ICIs) are rapidly emerging as a next-generation approach in cancer immunotherapy, offering unique advantages over monoclonal antibodies and small-molecule inhibitors. Their high selectivity, improved tumor penetration, and ease of synthesis make them attractive therapeutic candidates. Recent innovations, including cyclization, D-amino acid incorporation, dual-function peptides, and peptide-drug conjugates (PDCs), have significantly improved their stability, bioavailability, and therapeutic efficacy.
Despite these advancements, challenges remain, particularly in oral peptide delivery and prolonging in vivo half-life. Future research will focus on optimizing peptide modifications, hybrid nanoparticle delivery systems, and multifunctional peptide conjugates to enhance efficacy and reduce immunotherapy resistance.
As peptide therapeutics continue to evolve, their integration into combination regimens with vaccines, chemotherapy, and radiotherapy could reshape cancer treatment paradigms. With further clinical validation, peptide-based ICIs may become a cornerstone of modern oncology, offering more precise and effective cancer immunotherapies.
 LinkPeptide
LinkPeptide