Cleaner, Smarter, Greener: Redefining Peptide Manufacturing for the 21st Century
Abstract
Peptide therapeutics have become vital tools in modern medicine, yet their chemical synthesis remains environmentally burdensome. This article explores Stephen B. H. Kent’s 2025 perspective on redefining Solid-Phase Peptide Synthesis (SPPS) through the lens of green chemistry. It examines why traditional Fmoc and Boc methods fall short of sustainability goals and outlines Kent’s blueprint for transformation using N-carboxyanhydride (NCA) monomers, aqueous-compatible resins, and benign deprotection strategies. The discussion also highlights the growing importance of convergent synthesis and Native Chemical Ligation (NCL) in reducing waste and improving efficiency. Together, these innovations represent a fundamental shift toward sustainable peptide manufacturing — one that preserves SPPS’s precision while aligning with environmental and industrial imperatives.
From Breakthrough to Bottleneck: The Environmental Cost of SPPS
Peptide therapeutics are transforming modern medicine. From cancer immunotherapies to metabolic and neurological treatments, peptides stand at the frontier of targeted, high-precision therapeutics. Their remarkable specificity and safety profiles have made them indispensable tools in both research and clinical innovation.
Yet behind this scientific progress lies a quiet contradiction — the chemistry powering peptide production remains deeply unsustainable. For more than half a century, the field has relied on Solid-Phase Peptide Synthesis (SPPS), a method that reshaped peptide chemistry and made complex sequences accessible to laboratories and industries worldwide.
Pioneered by Bruce Merrifield in 1963, SPPS introduced a revolutionary idea: assemble peptides step by step on an insoluble polymer resin. This approach eliminated laborious solution-phase purifications, enabled automation, and made synthetic peptides commercially viable. It is no exaggeration to say that SPPS fueled the peptide era.
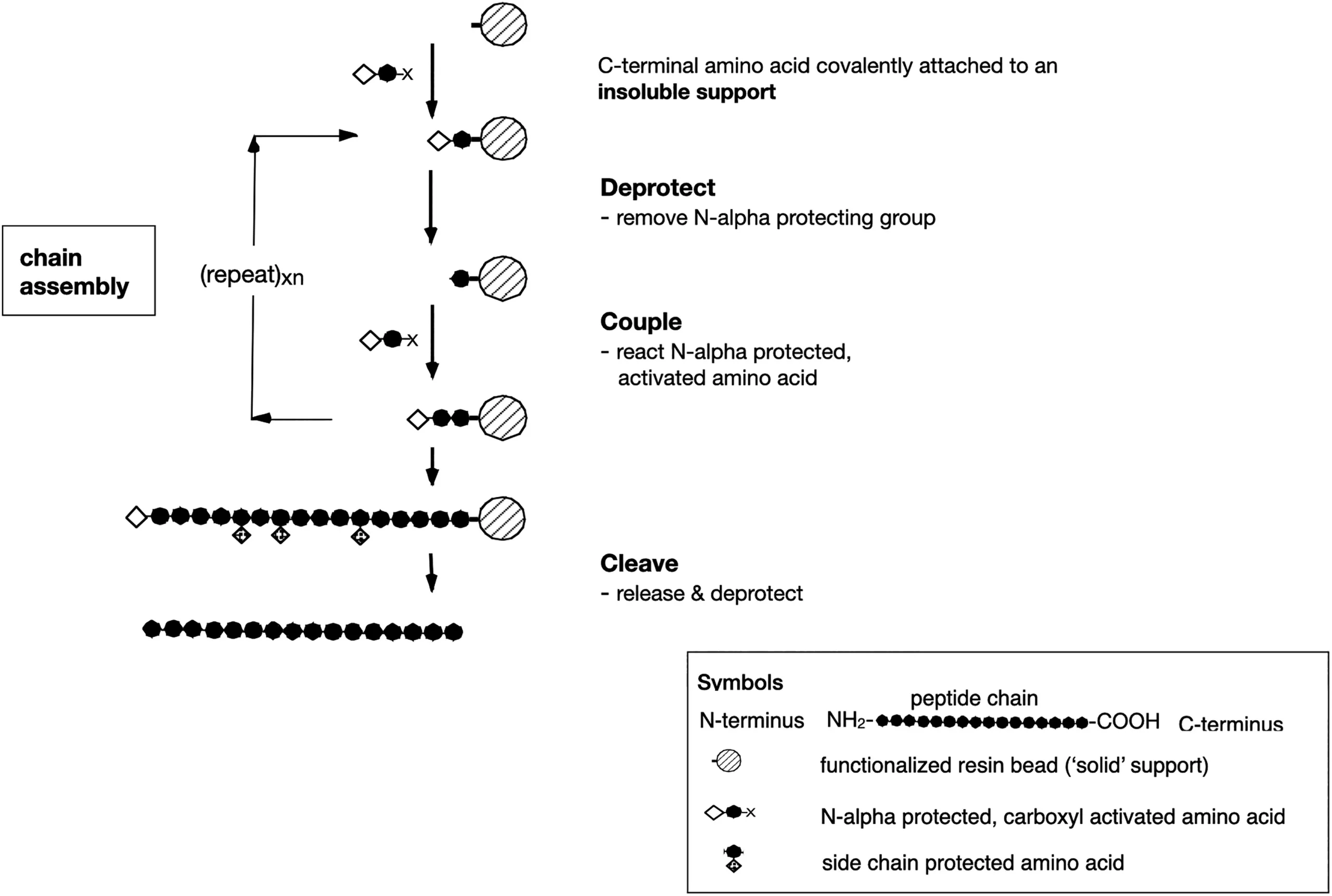
However, success has come at a cost. Conventional SPPS consumes toxic solvents such as DMF and piperidine, depends on bulky protecting groups that generate large waste streams, and requires extensive chromatographic purification to achieve pharmaceutical-grade purity. The result is an enormous environmental footprint — one that clashes with the very ideals of green chemistry.
Recognizing this paradox, Stephen B. H. Kent, in his 2025 Journal of Peptide Science essay, calls for a radical rethinking of peptide synthesis. Rather than optimizing the old system, Kent argues for rebuilding it — combining the molecular precision of SPPS with the environmental responsibility of green chemical design. His vision marks a turning point for the field: a shift from efficiency at any cost to sustainability by design.
Why Traditional SPPS Can’t Be Called Green — Yet Still Rules the Lab
Solid-Phase Peptide Synthesis (SPPS) remains the backbone of peptide chemistry, both in academic research and large-scale drug manufacturing. Its principle is elegantly simple: peptides are assembled step by step on a solid resin, with each amino acid added sequentially to the growing chain. Two major chemistries dominate this process — Fmoc-SPPS, which uses a base-labile fluorenylmethyloxycarbonyl group, and Boc-SPPS, which employs an acid-labile tert-butyloxycarbonyl group. Each offers precise control over peptide sequence and structure, yet both come with substantial environmental costs.
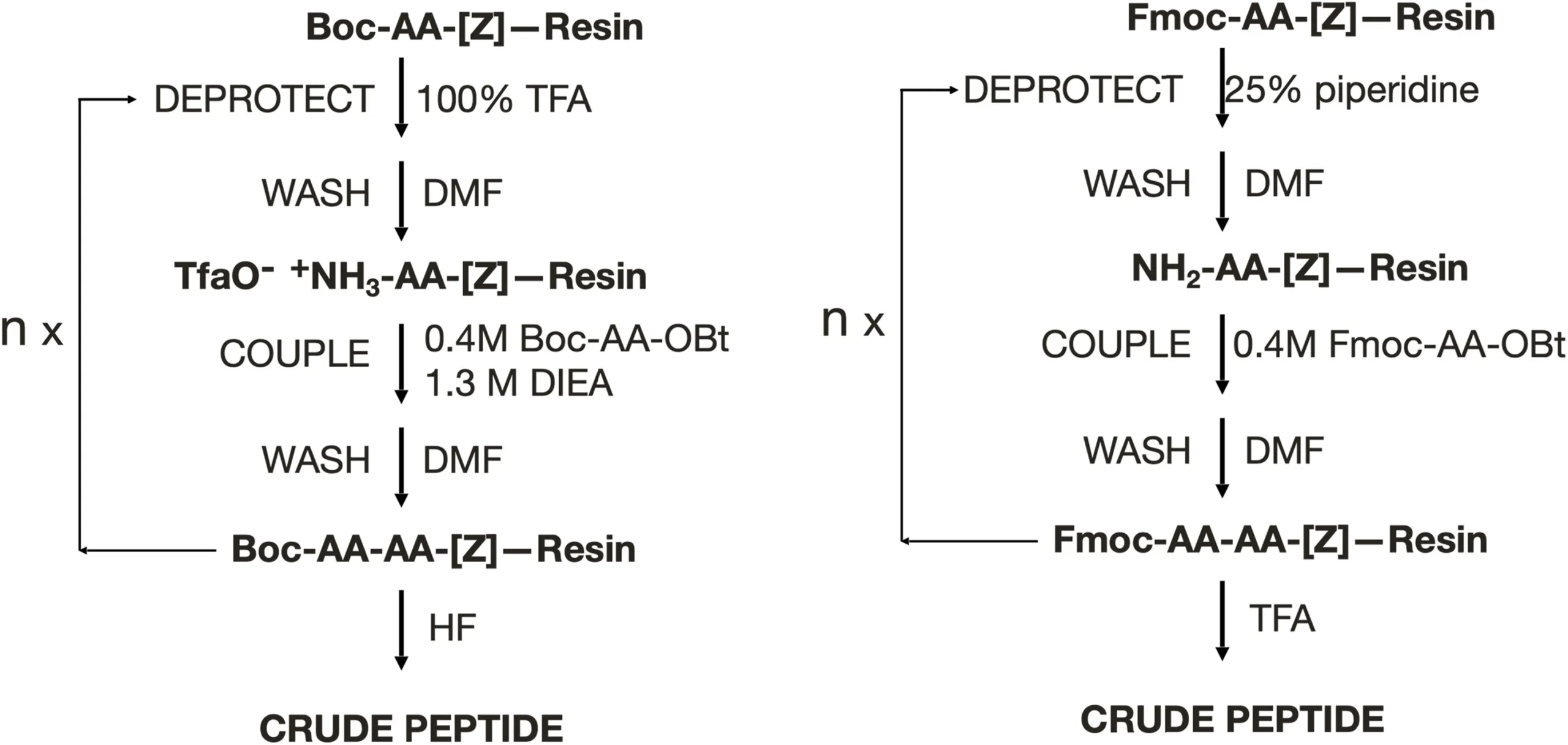
Kent points out that neither Fmoc nor Boc chemistry aligns with green chemistry principles. The reagents are toxic, the protecting groups heavy, and the solvents—particularly DMF, DCM, and piperidine—pose significant disposal challenges. Atom economy, a key measure of sustainable design, is especially poor: the Fmoc group alone has a mass nearly twice that of a typical amino acid residue, meaning most of the reaction mass ends up as waste. Moreover, to ensure complete coupling, chemists routinely use three- to fivefold excesses of each amino acid, compounding inefficiency and cost.
Despite these drawbacks, SPPS remains indispensable. Its resin-based platform offers unique physicochemical advantages: within solvent-swollen beads, peptide chains remain highly solvated, minimizing aggregation and ensuring near-solution reaction kinetics. This built-in solvation effect, combined with automation, reproducibility, and scalability, explains why SPPS continues to dominate peptide synthesis today.
Kent’s challenge is therefore not to abandon SPPS but to reinvent it—to preserve its precision and versatility while redesigning it for a cleaner, more sustainable future.
Rethinking the Chemistry: Building a Greener Foundation for SPPS
If Solid-Phase Peptide Synthesis (SPPS) has transformed peptide science, it must now evolve to meet the demands of sustainability. In his 2025 essay, Stephen B. H. Kent argues that greening SPPS requires more than swapping out a few reagents — it demands a rethinking of the chemistry itself. His proposed blueprint focuses on atom economy, solvent choice, resin design, and purification efficiency, all guided by the twelve principles of green chemistry.

1. N-Carboxyanhydrides: A Smarter Building Block
At the heart of Kent’s vision lies the use of amino acid N-carboxyanhydrides (NCAs) as next-generation monomers. Unlike Fmoc or Boc derivatives, NCAs are self-activated and N-protected, releasing only carbon dioxide as a by-product when forming a peptide bond. This means no external activators, no toxic waste, and dramatically improved atom economy. NCAs can also react in aqueous environments, opening the door to water-based peptide synthesis — a milestone for green chemistry.
2. Designing Aqueous-Compatible Resins
To use NCAs effectively, the polymer support must withstand both water and organic solvents. Kent highlights the NH₂–(TTD-Succ)n–(S-DVB) resin, a chemically defined support derived from common reagents. It offers high loading capacity (~1 mmol/g) and promotes clean, efficient coupling reactions while minimizing side products.
3. Minimizing Protection and Waste
Under mild NCA conditions, only a few amino acids — histidine, lysine, and cysteine — require side-chain protection. This selective protection drastically reduces reagent use and waste. For final cleavage, Kent suggests photolytic or nucleophilic methods rather than corrosive acids like TFA, aligning the process with environmentally safe protocols.
4. Smarter Purification
Finally, Kent emphasizes purification strategies that cut energy and solvent demands — such as tag-assisted isolation and sample-displacement HPLC — ensuring that green chemistry principles extend beyond synthesis to product recovery.
Together, these innovations outline a future where SPPS maintains its precision but sheds its environmental burden — a reinvention of peptide synthesis at the molecular level.
Beyond Stepwise Synthesis: Convergent Chemistry and the Power of Native Ligation
While stepwise SPPS has long been the workhorse of peptide chemistry, its linear nature makes it inherently wasteful. Each coupling cycle adds not only a new amino acid but also potential impurities, incomplete reactions, and excess reagents. Over dozens of cycles, even minor inefficiencies compound, demanding massive solvent use and extensive purification.
To move beyond these limits, Stephen Kent proposes returning to a principle as old as organic synthesis itself — convergent assembly. Instead of building one long chain residue by residue, convergent synthesis constructs shorter, purified peptide segments separately and then joins them efficiently. This approach minimizes exposure to harsh reagents, improves yields, and dramatically reduces waste.
The key to this transformation is Native Chemical Ligation (NCL), a technique first developed in Kent’s own laboratory. NCL enables the covalent joining of unprotected peptides in aqueous solutions under near-neutral conditions. The process operates without protecting groups, uses water as the solvent, and proceeds at room temperature — an embodiment of green chemistry. Each ligation step forms a natural peptide bond, producing no extraneous by-products and offering near-perfect atom economy.
Kent illustrates this efficiency with a striking comparison: the 46-residue protein Crambin synthesized stepwise by SPPS yielded around 12%, whereas the same molecule assembled via convergent NCL achieved 24% yield — twice the output with fewer purification steps and lower environmental cost.
Supporting technologies, such as peptide hydrazides and thioesters, further expand the power of NCL by enabling modular, scalable ligation of multiple segments. Together, these advances redefine what “green synthesis” means — not incremental improvement, but a paradigm shift toward efficiency, precision, and sustainability.
Convergent chemistry, Kent concludes, is more than an academic concept; it’s the foundation for the next generation of eco-conscious peptide manufacturing.
Toward a Sustainable Future: The New Blueprint for Peptide Manufacturing
In the final section of his 2025 essay, Stephen B. H. Kent envisions a peptide synthesis landscape where efficiency and sustainability coexist. His proposed framework — summarized in his “Green SPPS” model — reimagines every stage of the process. Peptides would be built using N-carboxyanhydrides (NCAs) in aqueous or mild aqueous–organic solvents, supported by resins designed for environmental compatibility. Corrosive acids like trifluoroacetic acid (TFA) would be replaced with photolytic or nucleophilic cleavage methods, while tag-assisted purification and sample-displacement chromatography would dramatically lower solvent and energy consumption.
This approach represents more than a procedural improvement; it’s a philosophical shift. By emphasizing atom economy, benign reagents, and low process mass intensity, Kent’s model aligns peptide manufacturing with the broader goals of green chemical engineering. For industrial peptide therapeutics, this could mean cleaner production pipelines, reduced carbon footprints, and faster compliance with environmental regulations — all while maintaining analytical rigor and product quality.
The road to fully sustainable peptide synthesis will demand creativity, material innovation, and cross-disciplinary collaboration. Yet Kent’s blueprint offers a clear direction: a future where chemical precision meets environmental responsibility, and where green SPPS becomes not an aspiration, but the new standard for peptide science.
Reference
Kent, S. B. (2025). Fundamental aspects of SPPS and green chemical peptide synthesis. Journal of Peptide Science, 31(5), e70013.
https://doi.org/10.1002/psc.70013
Bryan, M. C., Dunn, P. J., Entwistle, D., Gallou, F., Koenig, S. G., Hayler, J. D., … & Weiberth, F. J. (2018). Key Green Chemistry research areas from a pharmaceutical manufacturers’ perspective revisited. Green Chemistry, 20(22), 5082-5103.
https://doi.org/10.1039/C8GC01276H
Tran, T. V., Shen, Y., Nguyen, H. D., Deng, S., Roshandel, H., Cooper, M. M., … & Do, L. H. (2022). N-Carboxyanhydrides directly from amino acids and carbon dioxide and their tandem reactions to therapeutic alkaloids. Green Chemistry, 24(23), 9245-9252.
https://doi.org/10.1039/D2GC03507C
Hirschmann, R., Schwam, H., Strachan, R. G., Schoenewaldt, E. F., Barkemeyer, H., Miller, S. M., … & Denkewalter, R. G. (1971). Controlled synthesis of peptides in aqueous medium. VIII. Preparation and use of novel. alpha.-amino acid N-carboxyanhydrides. Journal of the American Chemical Society, 93(11), 2746-2754.
 LinkPeptide
LinkPeptide