Crossing the Blood–Brain Barrier: Can Shuttle Peptides Deliver Kv1.3 Inhibitors to the Brain?
Abstract
Peptide therapeutics are increasingly explored for the treatment of neurological disorders due to their high potency and target specificity. However, delivering peptide drugs to the brain remains a major challenge because the blood–brain barrier (BBB) restricts the entry of most large and hydrophilic molecules. One promising strategy to overcome this limitation involves the use of BBB shuttle peptides that facilitate receptor-mediated transport across brain endothelial cells. Recent studies have investigated whether conjugating shuttle peptides to therapeutic cargos can enhance brain delivery. In particular, the potent Kv1.3 potassium channel inhibitor HsTX1[R14A], a disulfide-rich peptide derived from scorpion venom, has been examined as a candidate for targeting neuroinflammation. By attaching BBB shuttle peptides such as MTfpep or Angiopep-2 using click chemistry, researchers aimed to improve the peptide’s brain permeability while preserving its biological activity. Although these conjugates maintained strong Kv1.3 inhibitory potency, experimental results suggest that shuttle peptide conjugation alone may not significantly enhance BBB penetration, highlighting the complexity of peptide-based CNS drug delivery.
Introduction: The Challenge of Delivering Peptide Drugs to the Brain
Peptide therapeutics have become an increasingly important class of molecules in modern biomedical research and drug development. Compared with small-molecule drugs, peptides often offer higher specificity, stronger target affinity, and lower off-target toxicity, making them attractive candidates for treating complex diseases. In recent years, peptide-based approaches have gained significant attention in the study of central nervous system (CNS) disorders, including Alzheimer’s disease, Parkinson’s disease, and other neurodegenerative conditions linked to chronic inflammation. As researchers continue to discover new bioactive peptides and venom-derived molecules with powerful pharmacological activity, peptide therapeutics are emerging as promising tools for targeting neurological pathways.
Despite their therapeutic potential, peptide drugs face a major biological obstacle when targeting the brain: the blood–brain barrier (BBB). The BBB is a specialized vascular interface composed of tightly connected endothelial cells that regulate the passage of molecules from the bloodstream into the brain. This protective barrier plays a critical role in maintaining neural homeostasis by preventing toxins, pathogens, and large molecules from entering brain tissue. However, the same protective mechanism also significantly limits the delivery of many therapeutic compounds.
Most peptides are large, hydrophilic molecules that cannot easily diffuse across cell membranes. Even peptides with high stability and strong biological activity may struggle to reach effective concentrations in the brain when administered systemically. Enzymatic degradation in the bloodstream and rapid clearance from circulation can further reduce the amount of peptide that reaches the CNS.
To address this challenge, researchers are actively exploring innovative delivery strategies designed to enhance peptide transport into the brain. Among the most promising approaches is the use of blood–brain barrier shuttle peptides, short targeting sequences that can interact with specific receptors on brain endothelial cells and facilitate receptor-mediated transport across the BBB.
One peptide that has attracted particular interest in neuroinflammation research is HsTX1[R14A], a potent inhibitor of the voltage-gated potassium channel Kv1.3. Scientists have recently investigated whether conjugating this peptide to BBB shuttle peptides could improve its ability to reach brain tissue. Understanding how such delivery strategies perform is essential for advancing peptide-based therapies for neurological diseases.
Kv1.3 Channels: A Promising Target for Neuroinflammation
Neuroinflammation is increasingly recognized as a central contributor to the progression of many neurodegenerative diseases. In disorders such as Alzheimer’s disease and Parkinson’s disease, chronic activation of immune cells in the brain can trigger inflammatory cascades that damage neurons and disrupt normal neural function. Among the various cell types involved in this process, microglia play a particularly important role. These resident immune cells of the central nervous system respond to injury or pathological stimuli by releasing inflammatory mediators.
Recent research has highlighted the voltage-gated potassium channel Kv1.3 as a key regulator of microglial activation. Kv1.3 channels are upregulated in pro-inflammatory microglia and are involved in controlling membrane potential and signaling pathways associated with immune responses. Because of this role, blocking Kv1.3 activity has been shown to suppress microglial activation and reduce neuroinflammatory signaling in several experimental models.
Targeting Kv1.3 therefore represents a promising strategy for developing therapies aimed at neurodegenerative diseases. While small-molecule inhibitors of Kv1.3 have been explored, peptide inhibitors derived from venom toxins have demonstrated particularly strong potential. These peptides often exhibit extremely high potency and remarkable selectivity, enabling precise targeting of ion channels while minimizing interactions with related proteins.
One of the most potent Kv1.3 inhibitors discovered to date is HsTX1[R14A], a peptide derived from the venom of the scorpion Heterometrus spinifer. This peptide consists of 34 amino acids stabilized by four disulfide bonds, which confer strong structural stability and resistance to degradation. HsTX1[R14A] has been reported to inhibit Kv1.3 channels with picomolar potency, making it one of the most powerful peptide blockers of this channel.
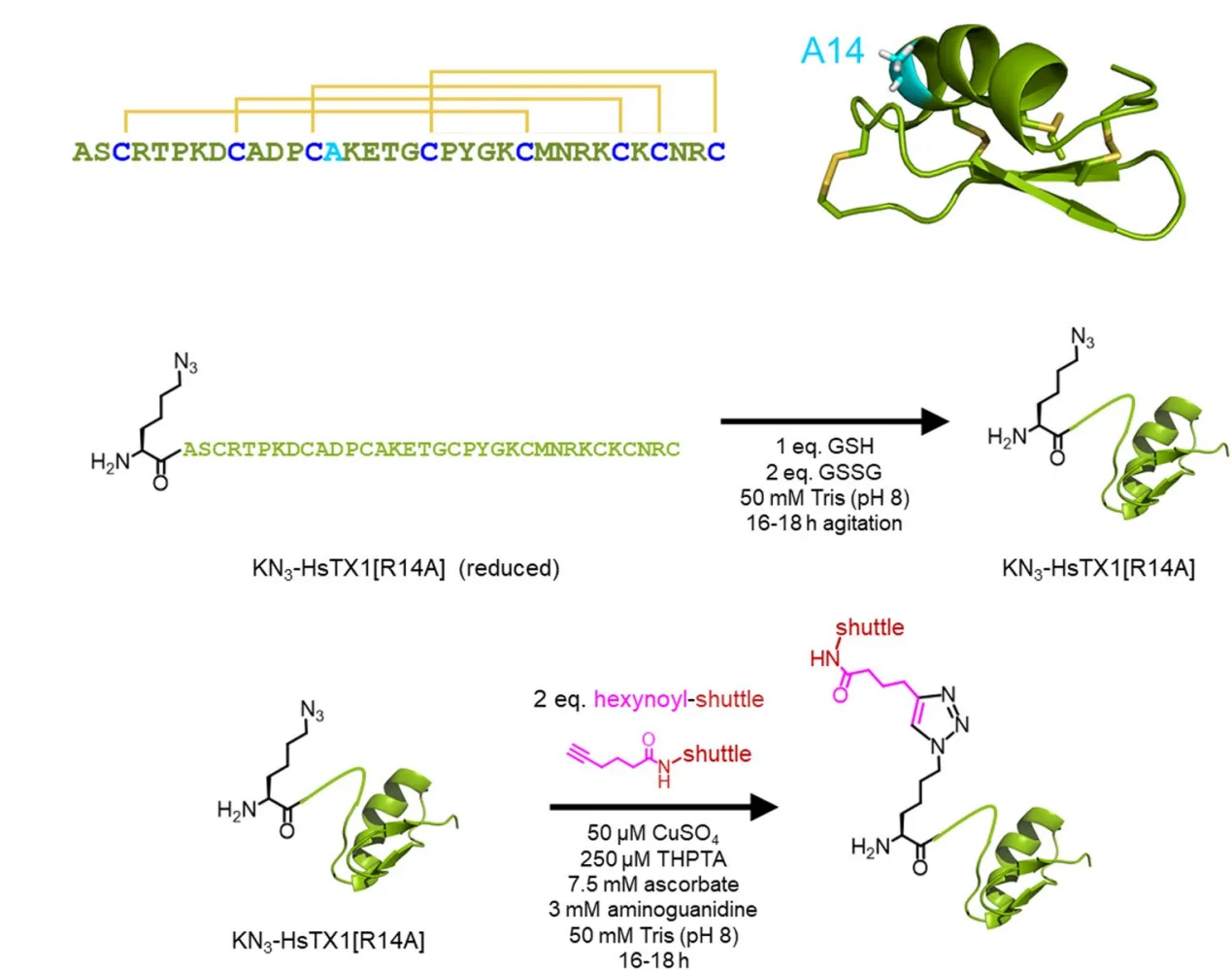
However, despite its exceptional activity and selectivity, HsTX1[R14A] faces a familiar challenge shared by many peptide therapeutics: limited ability to cross the blood–brain barrier. As a relatively large and structured peptide, it cannot easily penetrate the BBB through passive diffusion. Improving the brain delivery of such peptides is therefore an important step toward translating these molecules into potential therapies for neurological diseases.
BBB Shuttle Peptides: A Strategy for Brain Delivery
To overcome the transport limitations imposed by the blood–brain barrier, researchers have explored various strategies that take advantage of natural transport pathways within the BBB. One promising approach involves BBB shuttle peptides, short peptide sequences capable of interacting with specific receptors expressed on brain endothelial cells.
Unlike passive diffusion, these shuttle peptides utilize receptor-mediated transcytosis, a process in which molecules bind to receptors on the surface of endothelial cells, are internalized, and then transported across the cell to the opposite side of the barrier. By attaching therapeutic molecules to these shuttle peptides, it may be possible to deliver otherwise impermeable compounds into the brain.
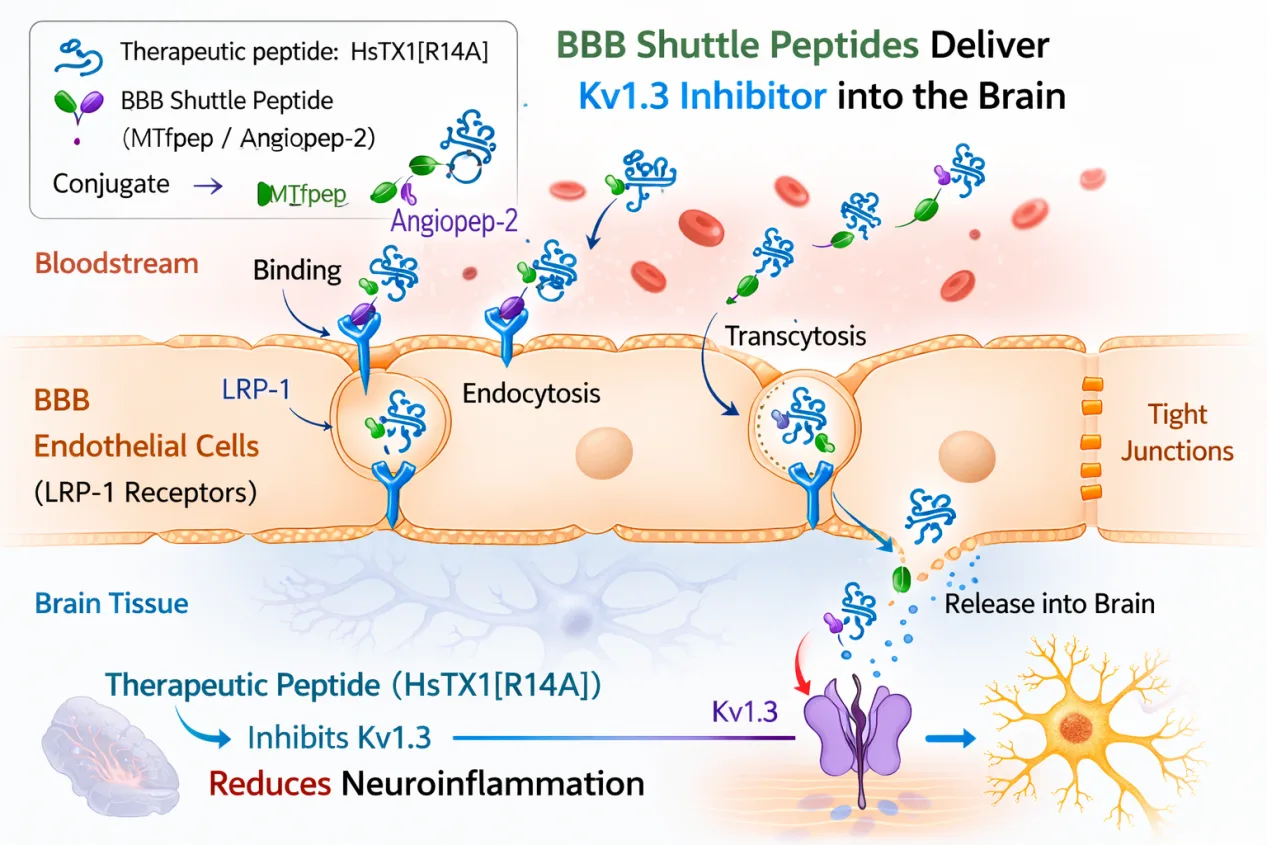
Two BBB shuttle peptides that have received significant attention in drug delivery research are MTfpep and Angiopep-2.
MTfpep is a short peptide fragment derived from melanotransferrin, a protein known to interact with the low-density lipoprotein receptor-related protein-1 (LRP-1) expressed on BBB endothelial cells. Previous studies have shown that MTfpep can enhance the brain uptake of large molecules such as antibodies, enzymes, and nucleic acids when chemically conjugated to these cargos.
Another widely studied shuttle peptide is Angiopep-2, a synthetic peptide designed to target the same LRP-1 receptor pathway. Angiopep-2 has been used to deliver various therapeutic cargos across the BBB, including small molecules, proteins, and nanoparticles. Notably, drug conjugates incorporating Angiopep-2 have progressed into clinical trials for certain brain cancers, demonstrating the translational potential of this delivery strategy.
Given the reported success of these shuttle peptides in transporting large biomolecules into the brain, researchers hypothesized that attaching MTfpep or Angiopep-2 to the Kv1.3-blocking peptide HsTX1[R14A] might improve its ability to cross the BBB and reach brain tissue.
Engineering Shuttle–Peptide Conjugates with Click Chemistry
Creating functional peptide conjugates requires precise chemical methods that preserve both the structure of the therapeutic peptide and the activity of the targeting ligand. One powerful approach for achieving this is click chemistry, a class of highly selective chemical reactions widely used in bioconjugation.
In the development of shuttle-toxin conjugates, researchers employed copper-catalyzed azide–alkyne cycloaddition (CuAAC), one of the most commonly used click chemistry reactions. This reaction enables the efficient joining of two molecules containing complementary chemical groups—an azide and an alkyne—under mild conditions compatible with peptides.
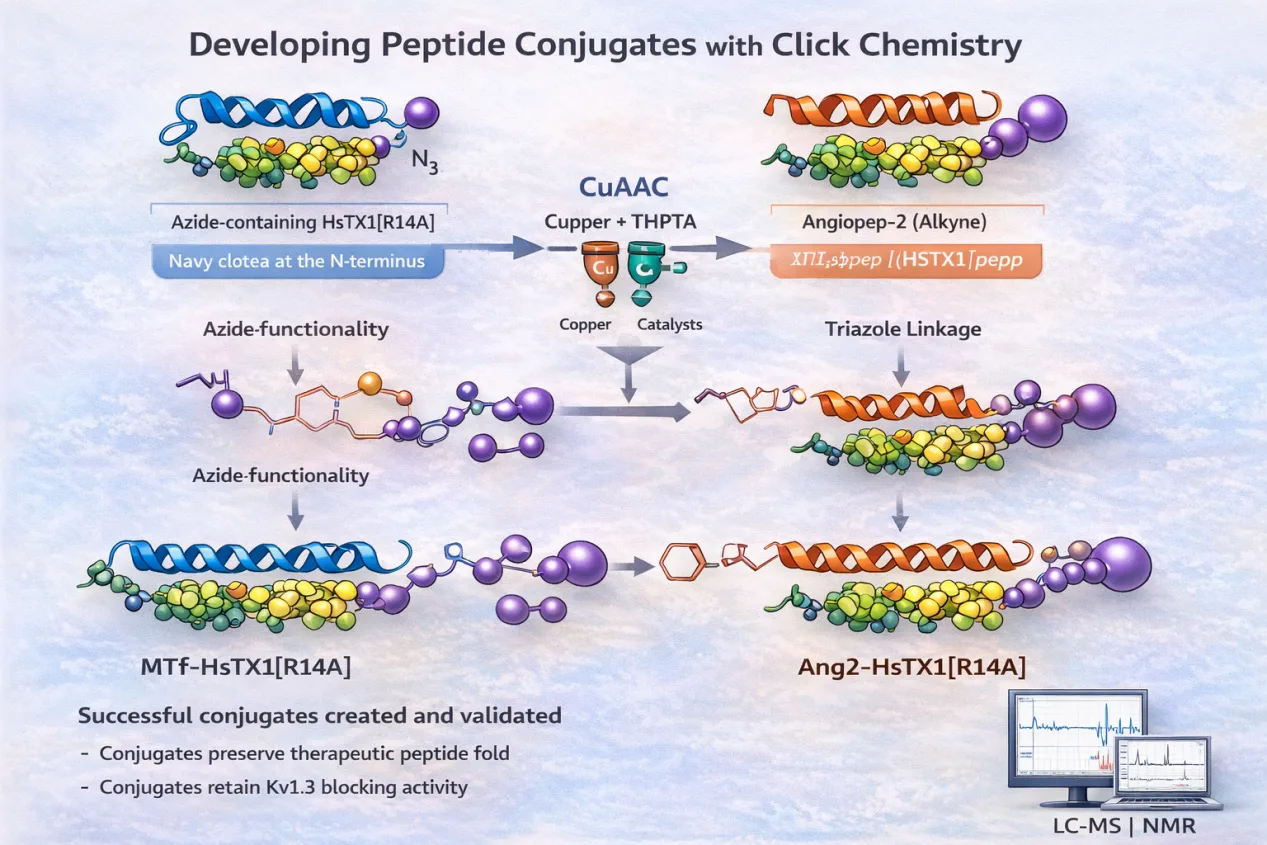
In this strategy, the Kv1.3 inhibitor HsTX1[R14A] was first modified to include an azide-containing amino acid at its N-terminus. Separately, the shuttle peptides MTfpep and Angiopep-2 were synthesized with alkyne functional groups attached. When these two components were combined under CuAAC conditions, the azide and alkyne groups reacted to form a stable triazole linkage, producing the desired peptide conjugates.
This modular approach generated two new molecules:
- MTf-HsTX1[R14A]
- Ang2-HsTX1[R14A]
Analytical techniques such as liquid chromatography–mass spectrometry (LC-MS) and nuclear magnetic resonance (NMR) spectroscopy confirmed that the conjugates were successfully synthesized and retained the correct folded structure of the toxin peptide.
Functional testing using electrophysiology assays further demonstrated that the conjugates remained active inhibitors of Kv1.3 channels. Although their potency was slightly reduced compared with the original toxin, both conjugates still exhibited nanomolar inhibitory activity, indicating that the conjugation process preserved their ability to interact with the target channel.
Did Shuttle Peptides Improve Brain Delivery?
While the shuttle-toxin conjugates maintained biological activity, the key question remained: could these shuttle peptides actually improve brain delivery of the Kv1.3 inhibitor?
To investigate this, researchers evaluated the permeability of the peptides using both in vitro blood–brain barrier models and in vivo mouse experiments. In vitro studies employed cultured brain endothelial cell layers designed to mimic the BBB. These experiments measured how much peptide was able to pass from one side of the barrier to the other over time.
The results showed extremely low permeability for both the original toxin peptide and the MTf-HsTX1[R14A] conjugate. Even after extended incubation periods, only a very small fraction of the peptides crossed the cell barrier, and no significant difference was observed between the modified and unmodified versions.
Further testing in mice produced similar findings. After intravenous administration of the MTf-HsTX1[R14A] conjugate, measurable concentrations of the peptide were detected in plasma for up to two hours. However, analysis of brain tissue revealed no detectable peptide accumulation, suggesting that the shuttle peptide did not significantly enhance transport across the BBB.
These findings highlight an important lesson in peptide drug delivery: BBB shuttle strategies may not work universally for all cargos. The success of receptor-mediated transport can depend on many factors, including peptide size, structure, conjugation site, and receptor interactions. Even shuttle peptides that perform well with certain cargos may not provide the same benefits when attached to other molecules.
Nevertheless, the modular conjugation and analytical methods developed in this research provide valuable tools for exploring new strategies to improve brain delivery of peptide therapeutics. Continued advances in peptide engineering, targeting ligands, and delivery technologies will likely play an essential role in unlocking the potential of peptide drugs for treating neurological diseases.
Reference
Wai, D. C., Koehn, L. M., Kreutzer, E., Runwal, P., Naseem, M. U., Panyi, G., … & Norton, R. S. (2026). Brain Delivery of a Kv1. 3-Blocking Peptide is not Enhanced by Conjugation to Blood–Brain Barrier Shuttle Peptides. International Journal of Peptide Research and Therapeutics, 32(2), 30. https://doi.org/10.1007/s10989-026-10810-w
Anami, Y., Xiong, W., Yamaguchi, A., Yamazaki, C. M., Zhang, N., An, Z., & Tsuchikama, K. (2022). Homogeneous antibody–angiopep 2 conjugates for effective brain targeting. RSC advances, 12(6), 3359-3364. https://doi.org/10.1039/D1RA08131D
Bartok, A., Fehér, K., Bodor, A., Rákosi, K., Tóth, G. K., Kövér, K. E., … & Varga, Z. (2015). An engineered scorpion toxin analogue with improved Kv1. 3 selectivity displays reduced conformational flexibility. Scientific reports, 5(1), 18397. https://doi.org/10.1038/srep18397
Eyford, B. A., Singh, C. S., Abraham, T., Munro, L., Choi, K. B., Hill, T., … & Jefferies, W. A. (2021). A nanomule peptide carrier delivers siRNA across the intact blood-brain barrier to attenuate ischemic stroke. Frontiers in Molecular Biosciences, 8, 611367. https://doi.org/10.3389/fmolb.2021.611367
 LinkPeptide
LinkPeptide