Homoarginine Substitution in Antimicrobial Peptides: Improving Stability Without Losing Activity
Introduction: Antimicrobial Peptides as Next-Generation Antibiotics
Antimicrobial peptides (AMPs) are gaining increasing attention as potential alternatives to traditional antibiotics. These short peptides, usually composed of 10–60 amino acids, are part of the innate immune defense systems found in many organisms. They exhibit broad-spectrum antimicrobial activity against bacteria, fungi, viruses, and parasites.
Unlike conventional antibiotics that target specific metabolic pathways, antimicrobial peptides often act by disrupting microbial membranes or interfering with intracellular functions. Because of this mechanism, AMPs are generally less likely to trigger rapid resistance development.
Despite their strong antimicrobial potential, peptide therapeutics face a major challenge: poor stability in physiological environments. Proteases present in blood and tissues can quickly degrade peptides, significantly reducing their therapeutic effectiveness.
To address this limitation, researchers are increasingly exploring peptide engineering strategies, including the incorporation of non-canonical amino acids such as homoarginine.
The Importance of Arginine in Antimicrobial Peptides
Arginine residues play a crucial role in the activity of many antimicrobial peptides. The guanidinium group present in arginine allows strong electrostatic interactions with negatively charged bacterial membranes.
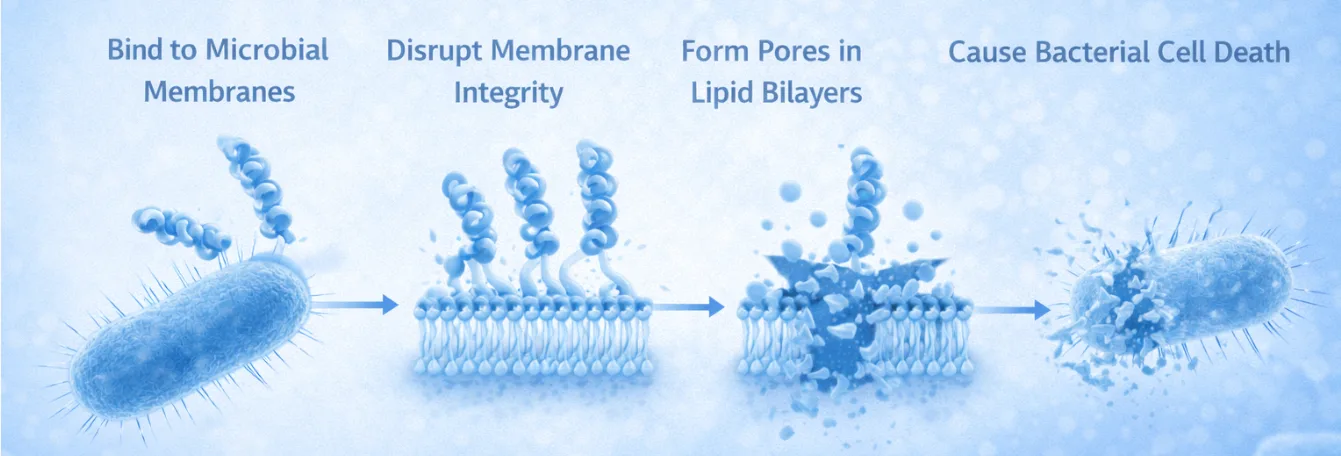
These interactions enable antimicrobial peptides to:
- bind efficiently to microbial membranes
- disrupt membrane integrity
- form pores in lipid bilayers
- ultimately cause bacterial cell death
Bacterial membranes typically carry a strong negative charge, while mammalian cell membranes are more neutral. This difference allows arginine-rich antimicrobial peptides to selectively target microbial cells.
However, the presence of multiple arginine residues also creates a weakness. Many proteases, particularly trypsin, specifically cleave peptide bonds near arginine residues, making these peptides susceptible to rapid degradation.
Homoarginine: A Promising Non-Canonical Amino Acid
Homoarginine is structurally similar to arginine but contains an additional methylene group in its side chain. Although this change may appear minor, it can significantly influence peptide stability and structure.
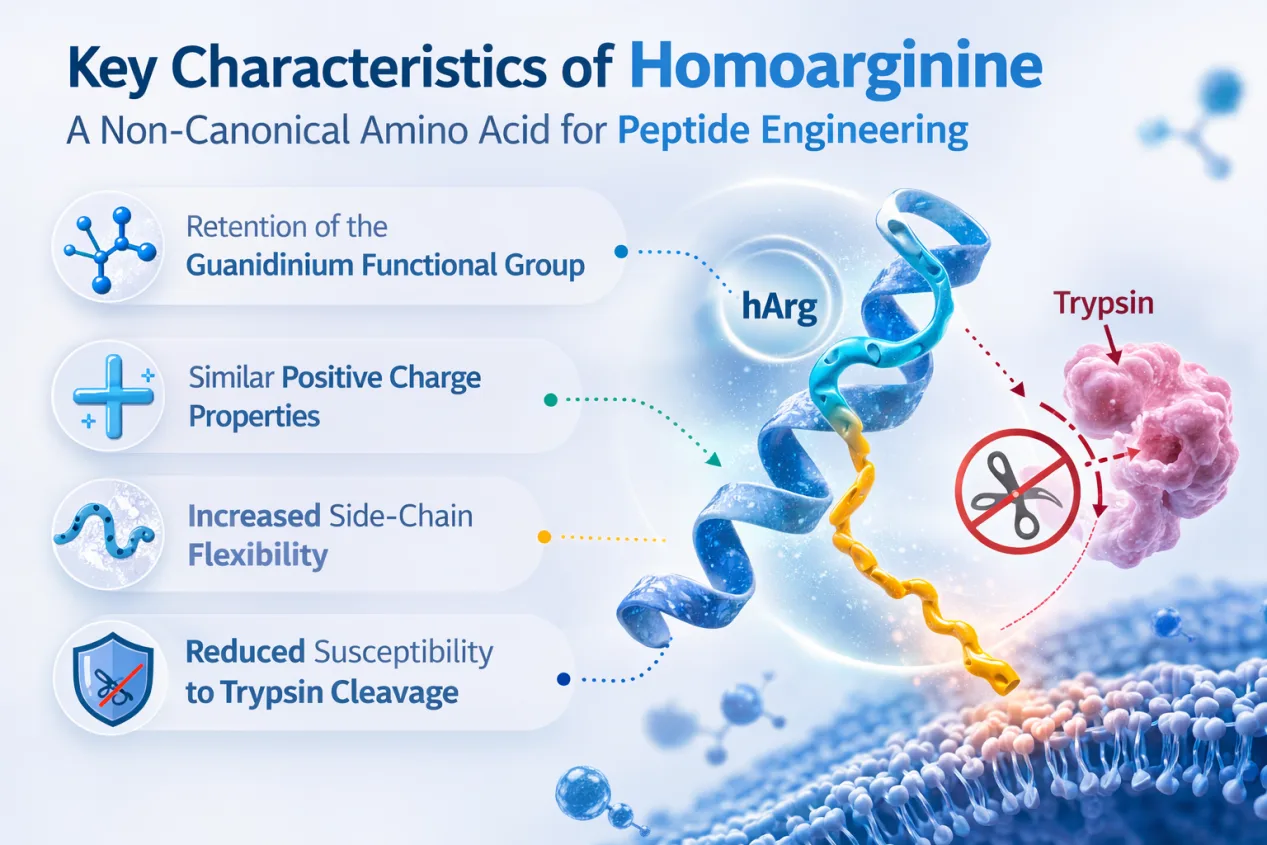
Key characteristics of homoarginine include:
- retention of the guanidinium functional group
- similar positive charge properties
- increased side-chain flexibility
- reduced susceptibility to trypsin cleavage
Because of these properties, homoarginine has attracted attention as a useful modification in peptide engineering. By replacing arginine residues with homoarginine, researchers aim to design antimicrobial peptides that maintain activity while becoming more resistant to enzymatic degradation.
Experimental Comparison of Arginine and Homoarginine Peptides
To evaluate the impact of this substitution, researchers have compared arginine-containing peptides with their homoarginine counterparts.
Two peptide models commonly used in these studies include:
- Mel4, an antimicrobial peptide derived from melimine
- PLR10, a poly-L-arginine peptide
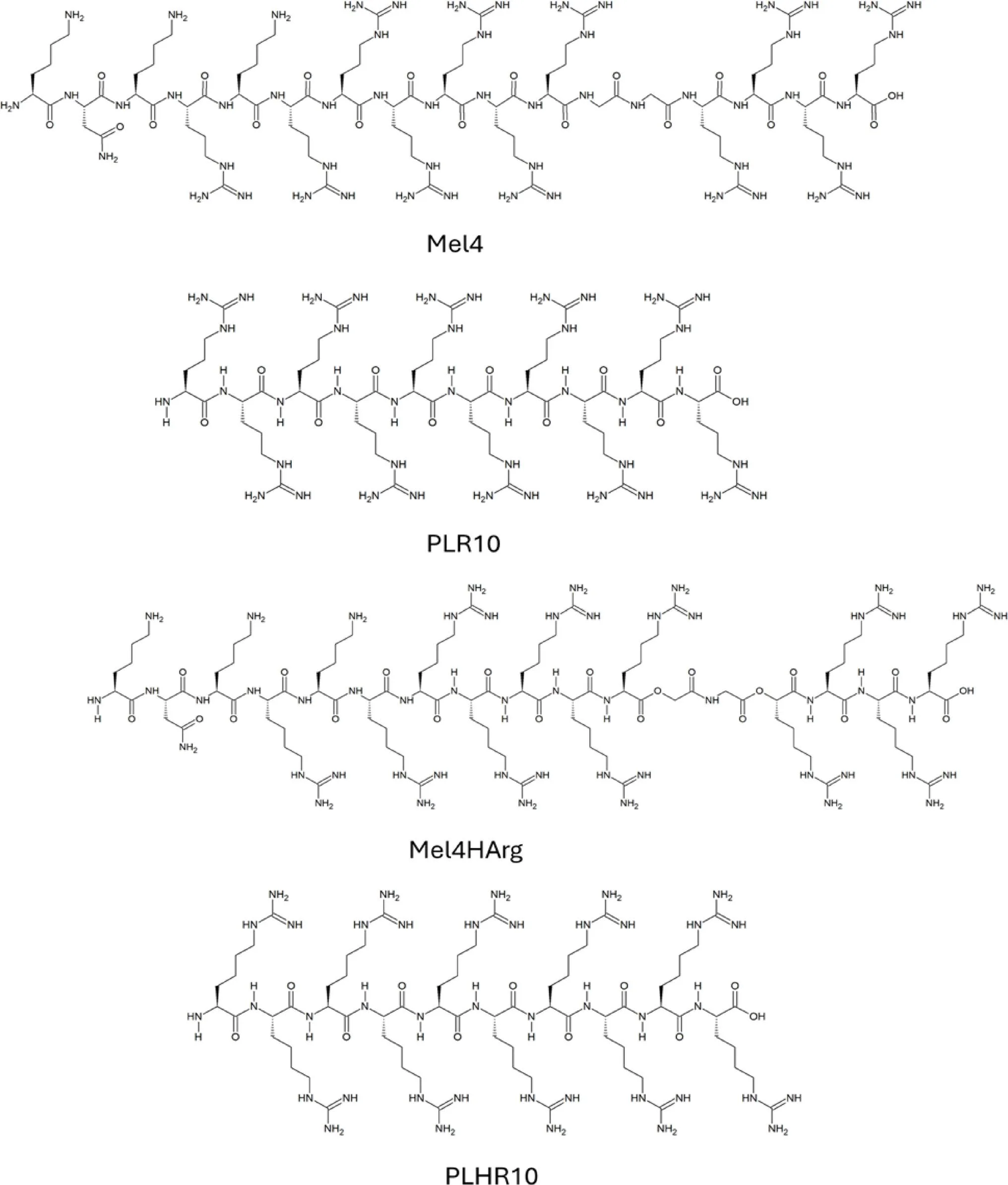
Their modified variants include:
- Mel4HArg – homoarginine substituted version of Mel4
- PLHR10 – poly-L-homoarginine peptide
These peptides were tested against several bacterial strains, including Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa.
The objective was to investigate how Arg→hArg substitution influences antimicrobial activity, structural conformation, and resistance to proteolytic enzymes.
Effects of Homoarginine Substitution on Antimicrobial Activity
Experimental results indicate that the impact of homoarginine substitution depends strongly on peptide sequence and structure.
For example:
- PLR10 and PLHR10 exhibited similar antimicrobial activity, particularly against S. aureus and E. coli.
- However, Mel4HArg showed reduced antimicrobial potency compared with Mel4 against some bacterial strains.
These findings highlight that amino acid substitutions can produce complex effects on peptide function. Even small structural modifications may influence charge distribution, folding behavior, and membrane interaction.
Therefore, careful optimization of peptide sequences is essential when incorporating non-canonical amino acids.
Structural Effects: Changes in Peptide Secondary Structure
Secondary structure plays an important role in how antimicrobial peptides interact with bacterial membranes.
Many AMPs adopt α-helical conformations when exposed to membrane environments. These structures allow peptides to insert into lipid bilayers and destabilize microbial membranes.
Circular dichroism analysis revealed important differences between arginine peptides and homoarginine variants.
In membrane-mimicking conditions:
- Arginine-containing peptides formed α-helical structures
- Homoarginine peptides largely lost this helical conformation
This structural change may explain why antimicrobial activity decreases in certain peptide sequences after substitution.
Although homoarginine maintains positive charge, the additional methylene group may slightly alter peptide folding and membrane interaction dynamics.
Enhanced Protease Resistance Through Homoarginine Incorporation
One of the most significant advantages of homoarginine substitution is improved resistance to enzymatic degradation.
Proteases such as trypsin normally cleave peptide bonds next to lysine and arginine residues. Because antimicrobial peptides often contain multiple arginine residues, they can be rapidly degraded under physiological conditions.
When arginine residues were replaced with homoarginine, researchers observed:
- reduced susceptibility to trypsin digestion
- increased stability of peptide fragments
- greater retention of antimicrobial activity after protease exposure
For instance, the homoarginine peptide PLHR10 retained significantly higher antimicrobial activity after trypsin treatment compared with PLR10.
These findings suggest that homoarginine substitution can extend the functional lifespan of antimicrobial peptides in biological environments.
Cytotoxicity and Safety Considerations
Another critical aspect of antimicrobial peptide development is ensuring that peptides selectively target microbes without harming host cells.
Cytotoxicity studies comparing these peptides showed that:
- polyarginine peptides demonstrated relatively high toxicity toward mammalian cells
- Mel4-derived peptides exhibited significantly lower cytotoxicity profiles
This emphasizes the importance of balancing antimicrobial potency with safety when designing peptide therapeutics.
Future Strategies for Antimicrobial Peptide Engineering
Incorporating non-canonical amino acids such as homoarginine represents just one of several strategies being explored to improve peptide therapeutics.
Additional approaches include:
- peptide cyclization
- lipidation or fatty acid modification
- incorporation of D-amino acids
- backbone modification
Combining these strategies with rational sequence design may lead to antimicrobial peptides with improved stability, potency, and selectivity.
Supporting Antimicrobial Peptide Research with Custom Synthesis
Advances in antimicrobial peptide research require access to reliable peptide synthesis technologies.
At LinkPeptide, researchers can obtain:
- custom antimicrobial peptide synthesis
- incorporation of non-canonical amino acids
- arginine-rich peptide design
- peptide modification and optimization services
These capabilities allow scientists to rapidly explore new peptide designs and accelerate antimicrobial peptide discovery.
Conclusion
Antimicrobial peptides represent a promising solution to the global challenge of antibiotic resistance. However, their practical application depends on overcoming challenges such as protease instability and cytotoxicity.
Homoarginine substitution offers a valuable strategy for improving peptide stability while preserving key functional properties of arginine-rich antimicrobial peptides. Although its effects vary depending on sequence context, this modification highlights the potential of non-canonical amino acids in peptide engineering.
As research continues to advance, innovative design strategies and custom peptide synthesis technologies will play a critical role in developing the next generation of antimicrobial therapeutics.
Reference
Enninful, G. N., Kuppusamy, R., Tiburu, E., Kumar, N., & Willcox, M. D. (2026). Assessing the Effect of Substitution of Arginine with Homoarginine in Two Antimicrobial Peptides. International Journal of Peptide Research and Therapeutics, 32(2), 24. https://doi.org/10.1007/s10989-026-10804-8
Adzhubei, A. A., Sternberg, M. J., & Makarov, A. A. (2013). Polyproline-II helix in proteins: structure and function. Journal of molecular biology, 425(12), 2100-2132. https://doi.org/10.1016/j.jmb.2013.03.018
Benfield, A. H., & Henriques, S. T. (2020). Mode-of-action of antimicrobial peptides: membrane disruption vs. intracellular mechanisms. Frontiers in Medical Technology, 2, 610997. https://doi.org/10.3389/fmedt.2020.610997
Decker, A. P., Mechesso, A. F., & Wang, G. (2022). Expanding the landscape of amino acid-rich antimicrobial peptides: Definition, deployment in nature, implications for peptide design and therapeutic potential. International Journal of Molecular Sciences, 23(21), 12874. https://doi.org/10.3390/ijms232112874
 LinkPeptide
LinkPeptide