Decoding Peptide Materials: How Sequence Context Shapes Supramolecular Assembly
Abstract
Peptide-based materials have emerged as a versatile platform for designing functional and adaptive soft matter, yet their self-assembly behavior often defies simple design rules. Traditional approaches that classify peptides by hydrophobicity, charge, or secondary-structure motifs frequently fail to predict material outcomes. Recent advances reveal that peptide assembly is governed by a context-dependent assembly code, in which amino-acid sequence, local residue arrangement, and environmental conditions act together to determine structure and dynamics. Subtle changes in sequence order or external parameters such as pH, salt concentration, and hydration can shift peptides across a continuum of material states, from soluble assemblies to liquid condensates, fibrous gels, and ordered crystals. Importantly, dynamic and disordered states are not design failures but functional architectures that resemble biological systems. Understanding peptide materials therefore requires a holistic, system-level perspective. Embracing sequence context as a design principle opens new opportunities for creating responsive, programmable, and biologically inspired peptide-based materials.
Peptide Materials Beyond Simple Rules
Peptide-based materials are gaining increasing attention as a powerful class of building blocks for biomaterials, nanotechnology, and adaptive soft matter. Composed of amino acids—the same molecular units used by living systems—peptides combine chemical precision, structural modularity, and intrinsic biocompatibility. These qualities make them especially attractive for applications such as tissue scaffolds, drug delivery systems, responsive gels, and functional nanostructures.
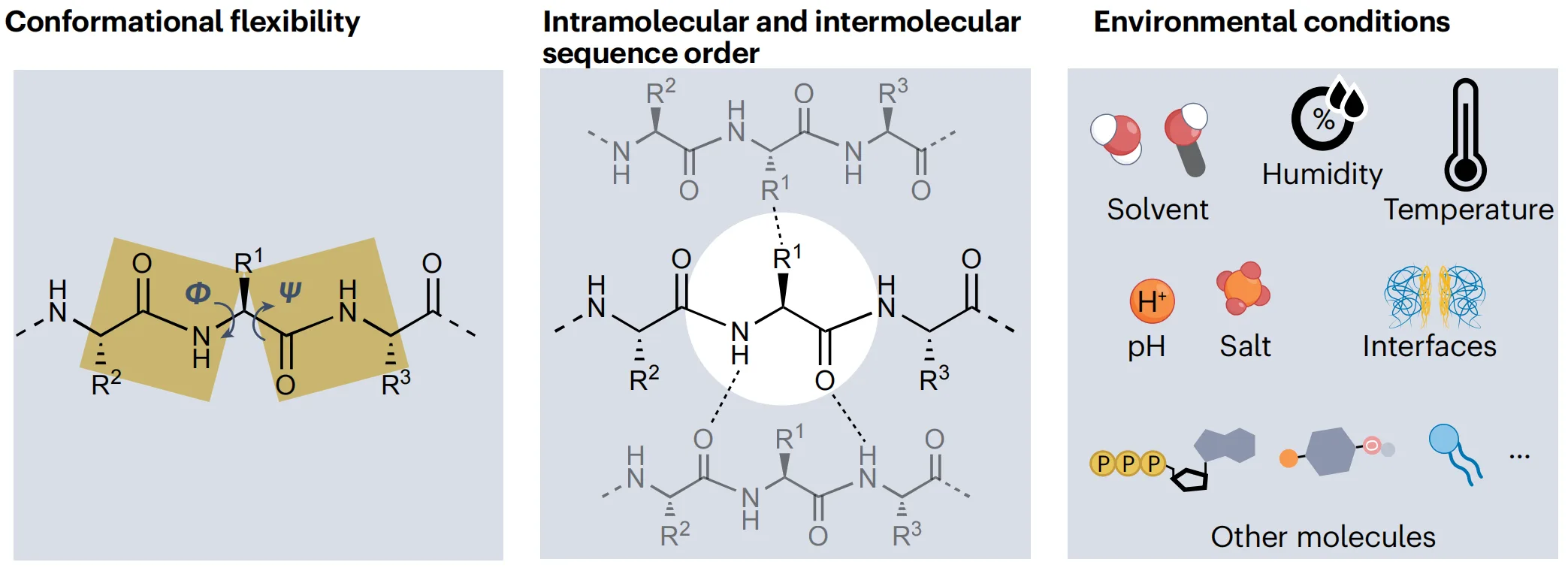
Despite this promise, the rational design of peptide materials remains a persistent challenge. Conventional design rules, which often rely on broad classifications such as hydrophobic versus hydrophilic residues or mimicry of protein secondary structures, frequently fall short. In practice, seemingly minor changes—a single amino-acid substitution, a rearrangement in sequence order, or a small shift in pH or ionic strength—can lead to dramatically different assembly outcomes, ranging from soluble states to liquid condensates, fibrous networks, or crystalline solids.
Recent research points to a unifying explanation: peptide self-assembly is governed not by a fixed set of rules, but by a context-dependent assembly code. In this framework, sequence, local interactions, and environmental conditions act together to determine material structure and behavior.
Peptides as Programmable Molecular Materials
Peptides occupy a distinctive position among molecular building blocks for materials design. Unlike synthetic polymers, which often rely on statistical chain compositions, peptides are defined by an exact amino-acid sequence. At the same time, they are far simpler and more modular than full-length proteins, making them easier to synthesize, modify, and study. This combination of precision and simplicity gives peptides an unusual level of design freedom.
What truly sets peptide materials apart, however, is the diversity of interactions they can access simultaneously. Peptide backbones engage in highly directional hydrogen bonding, while their side chains participate in a rich array of non-covalent interactions, including hydrophobic effects, electrostatic attractions, dipole–dipole interactions, and aromatic π-based interactions. These interaction modes do not act in isolation. Instead, they cooperate, compete, and reorganize dynamically as peptides assemble.
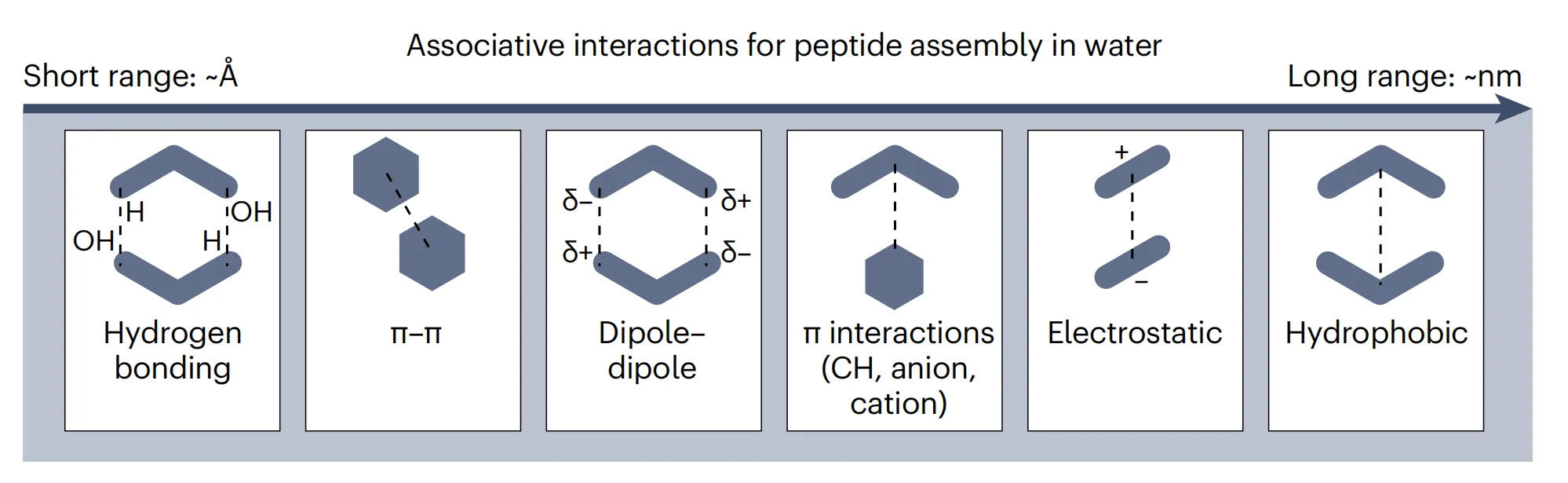
This interaction richness distinguishes peptides from other programmable materials such as DNA. DNA self-assembly relies on a relatively simple and predictable base-pairing code, enabling highly ordered and rationally designed structures. Peptides, by contrast, operate within a far larger interaction space, where outcomes are not dictated by a single dominant force. As a result, peptide assemblies can access a wide spectrum of material states, from highly ordered crystals to dynamic, disordered liquids.
This versatility is both a strength and a challenge. Peptides are powerful because they can form adaptive, responsive materials, but they are difficult to predict precisely because their behavior emerges from multiple, context-sensitive interactions acting at once. Understanding peptide materials therefore requires moving beyond fixed rules toward a more holistic view of assembly.
Sequence Context as the Governing Principle of Peptide Assembly
A central message emerging from recent peptide materials research is that amino acids do not possess fixed, context-independent roles in self-assembly. Instead, peptide behavior is governed by sequence context—the combined influence of amino-acid identity, sequence order, neighboring residues, and environmental conditions. This perspective fundamentally reshapes how peptide materials should be understood and designed.
Traditional approaches often assign amino acids simple functions, labeling them as hydrophobic, charged, or disorder-promoting. While useful as a starting point, such classifications fail to capture how the same residue can produce opposite outcomes in different sequences. An aromatic amino acid may stabilize a rigid crystal in one context, promote dynamic liquid-like behavior in another, or even enhance solubility when its interactions with water dominate. What matters is not only which amino acids are present, but where they are placed and what surrounds them.
Sequence context determines how side-chain interactions combine with backbone hydrogen bonding to shape the free-energy landscape of assembly. Subtle changes—such as rearranging the order of three amino acids or replacing a single residue—can alter the balance between directional and multidirectional interactions. These shifts may drive transitions between ordered fibres, liquid condensates, supramolecular dispersions, or fully soluble states, even when overall composition remains unchanged.
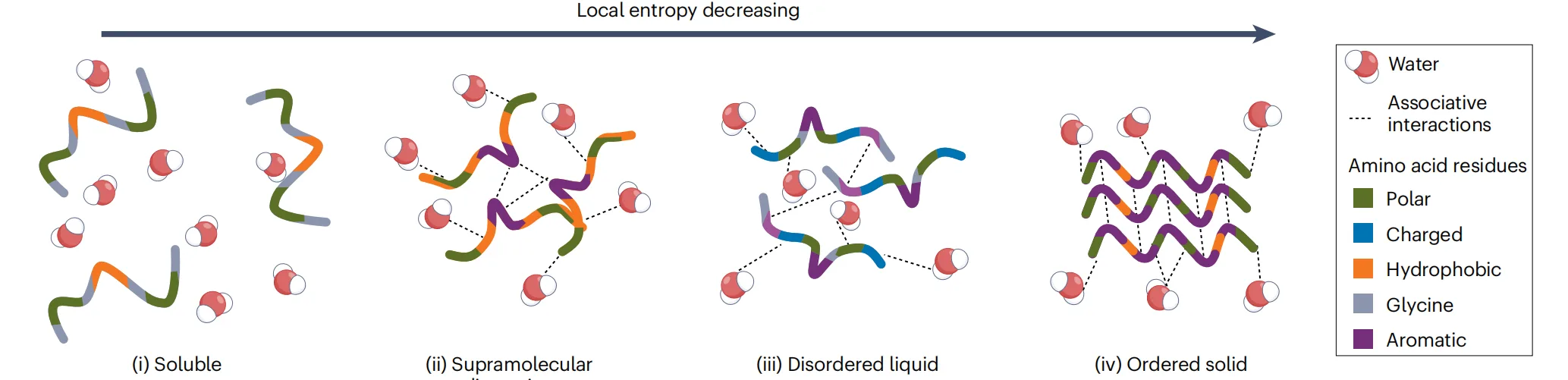
Importantly, no amino acid can be universally labeled as “order-promoting” or “disorder-promoting.” Residues such as glycine and proline regulate backbone flexibility, while aromatic residues like tyrosine and tryptophan act as versatile interaction hubs capable of engaging in hydrogen bonding, π–π interactions, and dipole-driven effects. Their impact depends entirely on sequence context.
Recognizing sequence context as the true assembly code moves peptide materials beyond rigid design rules and toward a more realistic, system-level understanding of structure formation.
From Crystals to Condensates: The Full Spectrum of Peptide Materials
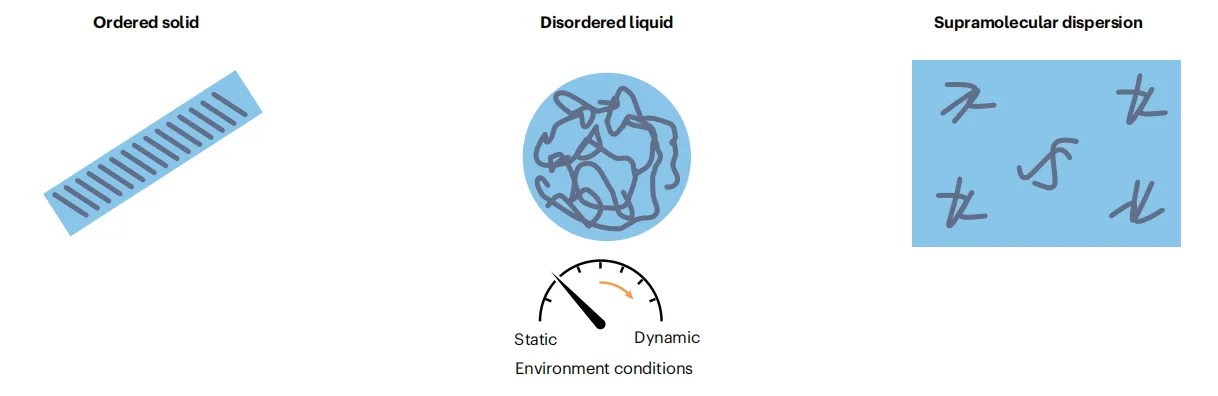
Peptide self-assembly does not produce a single, well-defined structural outcome. Instead, peptides populate a continuum of material states that spans from fully soluble molecules to highly ordered solids. Between these extremes lie supramolecular dispersions, liquid-like condensates formed through liquid–liquid phase separation, fibrous gels, and semi-crystalline networks. Understanding peptide materials therefore requires abandoning binary notions of “assembled” versus “non-assembled” in favor of a spectrum defined by order, dynamics, and hydration.
At one end of this continuum are ordered structures, such as fibres and crystals, stabilized by directional backbone hydrogen bonding and well-aligned side-chain interactions. These assemblies exhibit long-range molecular order and high mechanical stability. At the opposite end are disordered and dynamic systems, including liquid condensates and supramolecular dispersions, where peptides interact through transient, multidirectional contacts and remain highly mobile. Far from being structural defects, these disordered states often resemble biological assemblies and enable adaptability, reversibility, and responsiveness.
Crucially, small changes in sequence context can shift a peptide along this continuum without altering its overall composition. Rearranging aromatic residues, modifying backbone flexibility, or subtly changing side-chain orientation can suppress long-range order and favor dynamic interactions with water. In many cases, water itself becomes an integral part of the supramolecular network, mediating interactions and stabilizing disordered states.
Equally important is the role of assembly pathways. Peptide materials are sensitive to kinetic history: the route by which assembly occurs—such as temperature changes, solvent exchange, or mechanical agitation—can determine whether a system reaches a thermodynamic minimum or becomes trapped in a metastable state. This pathway dependence adds another layer of complexity but also expands the design space.
Viewing peptide assembly as a continuum highlights why these materials are exceptionally versatile and why controlling disorder can be just as important as achieving structural order.
How Environment Rewrites the Rules of Peptide Assembly
In peptide materials, environmental conditions are not passive background parameters—they actively participate in defining how peptides assemble. Factors such as pH, ionic strength, temperature, solvent composition, and humidity can reorganize the balance of non-covalent interactions that govern self-assembly. As a result, the same peptide sequence can access entirely different material states simply by changing its surroundings.
pH directly alters the protonation state of charged and polar side chains, reshaping electrostatic interactions and hydrogen-bonding networks. Salt concentration further modulates these effects through charge screening, while specific ions can strengthen or weaken hydrophobic interactions by reorganizing surrounding water molecules. Temperature introduces an additional layer of control by shifting the relative importance of enthalpic versus entropic contributions, often enhancing hydrophobic effects at elevated temperatures.
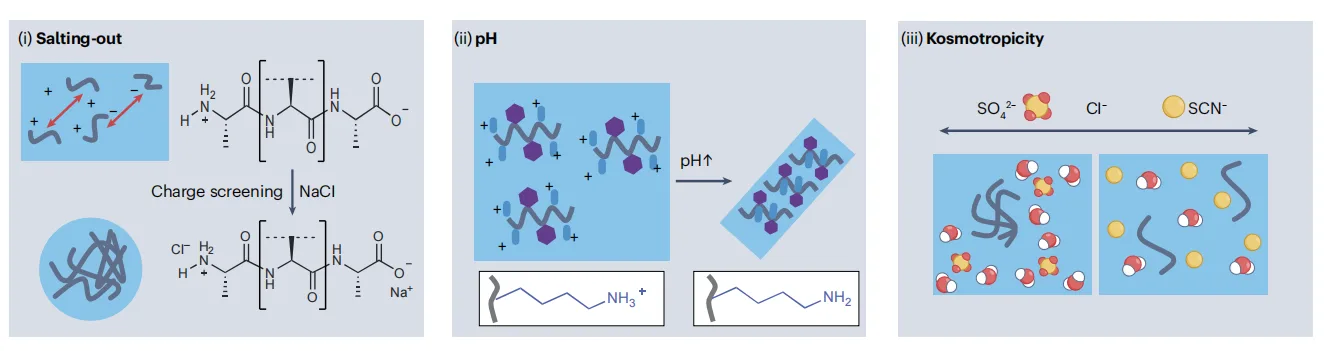
Water plays a particularly central role. Rather than acting solely as a solvent, water competes with peptide–peptide interactions and can stabilize either ordered or disordered assemblies depending on sequence context. In hydrated, flexible environments, peptides tend to favor dynamic, liquid-like structures. In contrast, dehydrated or hydrophobic environments promote tighter packing and long-range order.
This sensitivity enables peptides to function as stimuli-responsive materials. Transitions between soluble states, liquid condensates, gels, and solids can be triggered reversibly by modest environmental changes, without altering molecular composition. From a design perspective, this means that environmental conditions must be treated as an integral part of the assembly code.
Harnessing this principle allows researchers to engineer peptide materials that respond rapidly and predictably to external cues—an ability that is difficult to achieve with conventional synthetic polymers.
Designing the Next Generation of Peptide Materials
The growing recognition of context-dependent assembly marks a turning point in how peptide materials are designed and understood. Rather than searching for universal sequence rules, future progress lies in embracing peptides as complex, adaptive systems, whose properties emerge from the interplay between sequence, interactions, and environment. This shift challenges traditional trial-and-error approaches but also opens unprecedented opportunities for innovation.
Because peptide assembly depends on multiple competing interactions, predictive design requires systematic exploration of sequence space combined with detailed characterization of environmental effects. In this landscape, computational tools—particularly atomistic simulations and machine learning—are becoming indispensable. By analyzing large libraries of sequences and conditions, data-driven methods can uncover non-intuitive relationships that would be difficult to identify experimentally or through human intuition alone.
Equally important is the role of automation and standardized experimental protocols. Given the sensitivity of peptide assemblies to preparation history and pathway dependence, reproducibility and controlled exploration are essential for translating fundamental insights into practical materials.
Ultimately, peptide materials offer a bridge between the sophistication of biological systems and the scalability of synthetic materials. By mastering the principles of sequence context and environmental control, researchers can design peptide-based systems that are not only structurally diverse but also responsive, adaptable, and functional. In this sense, biology’s assembly code may evolve into a universal framework for next-generation material design.
Reference
Kaygisiz, K., Sementa, D., Athiyarath, V., Chen, X., & Ulijn, R. V. (2025). Context dependence in assembly code for supramolecular peptide materials and systems. Nature Reviews Materials, 1-24.https://doi.org/10.1038/s41578-025-00782-6
Ibusuki, R., Morishita, T., Furuta, A., Nakayama, S., Yoshio, M., Kojima, H., … & Furuta, K. Y. (2022). Programmable molecular transport achieved by engineering protein motors to move on DNA nanotubes. Science, 375(6585), 1159-1164.https://doi.org/10.1126/science.abj5170
Alshareedah, I., Moosa, M. M., Pham, M., Potoyan, D. A., & Banerjee, P. R. (2021). Programmable viscoelasticity in protein-RNA condensates with disordered sticker-spacer polypeptides. Nature communications, 12(1), 6620.https://doi.org/10.1038/s41467-021-26733-7
Frederix, P. W., Scott, G. G., Abul-Haija, Y. M., Kalafatovic, D., Pappas, C. G., Javid, N., … & Tuttle, T. (2015). Exploring the sequence space for (tri-) peptide self-assembly to design and discover new hydrogels. Nature chemistry, 7(1), 30-37.https://doi.org/10.1038/nchem.2122
 LinkPeptide
LinkPeptide