Elamipretide: A Mitochondria-Targeted Peptide for Cardiolipin-Focused Research
Abstract
Mitochondrial dysfunction is a central pathological feature of numerous diseases, including cardiovascular disorders, neurodegeneration, metabolic syndromes, and age-related tissue decline. Elamipretide (SS-31, MTP-131) is a mitochondria-targeted tetrapeptide developed to address mitochondrial impairment through selective interaction with cardiolipin, a phospholipid essential for inner mitochondrial membrane integrity and bioenergetic function. Structurally optimized for cellular permeability and mitochondrial accumulation, elamipretide stabilizes cardiolipin, preserves cristae architecture, enhances electron transport chain efficiency, and reduces excessive mitochondrial reactive oxygen species production. Preclinical studies across cardiovascular, renal, neurological, and metabolic disease models demonstrate its ability to restore mitochondrial bioenergetics, limit oxidative stress, and mitigate apoptosis and fibrosis. Clinical investigations further highlight its translational relevance, particularly in conditions driven by cardiolipin dysregulation. Collectively, elamipretide serves as a valuable research peptide for investigating mitochondrial biology and cardiolipin-centered disease mechanisms.
Mitochondrial Dysfunction as a Central Driver of Disease
Mitochondria are indispensable to cellular physiology, serving as the primary source of adenosine triphosphate (ATP) through oxidative phosphorylation. In addition to energy production, they play critical roles in calcium homeostasis, reactive oxygen species (ROS) regulation, and the intrinsic apoptotic pathway. When mitochondrial structure or function is compromised, cells experience energetic failure, oxidative damage, and increased susceptibility to programmed cell death. As a result, mitochondrial dysfunction has emerged as a unifying pathological mechanism across a broad range of diseases.
Extensive research has linked impaired mitochondrial bioenergetics to cardiovascular disorders, neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease, metabolic syndromes, renal disease, and age-related tissue decline. In many of these conditions, oxidative damage to mitochondrial membranes and loss of ATP-generating capacity precede irreversible cellular injury. Despite this central role, conventional therapies—such as systemic antioxidants or metabolic modulators—have often failed to deliver meaningful clinical benefits, largely due to insufficient targeting of mitochondrial membranes.
These limitations have fueled interest in mitochondria-targeted therapeutic strategies, particularly peptides engineered to localize selectively to mitochondrial compartments. Among these, elamipretide—also known as SS-31 or MTP-131—has gained significant attention as a research peptide due to its unique ability to interact directly with cardiolipin, a phospholipid essential for mitochondrial structure and function.
Structural Features That Enable Selective Mitochondrial Targeting
Elamipretide is a synthetic tetrapeptide with the amino acid sequence D-Arg–Dmt–Lys–Phe-NH2, specifically designed to penetrate cells and accumulate within mitochondria. Its molecular architecture combines hydrophilic and hydrophobic elements, giving the peptide amphipathic properties that support both aqueous solubility and membrane interaction.
Two positively charged residues—D-arginine and lysine—facilitate electrostatic attraction to cardiolipin, which carries a strong negative charge and is highly enriched in the inner mitochondrial membrane. This electrostatic interaction is a key determinant of elamipretide’s selective mitochondrial localization. At the same time, the aromatic residues dimethyltyrosine (Dmt) and phenylalanine contribute hydrophobic interactions that enhance membrane association.
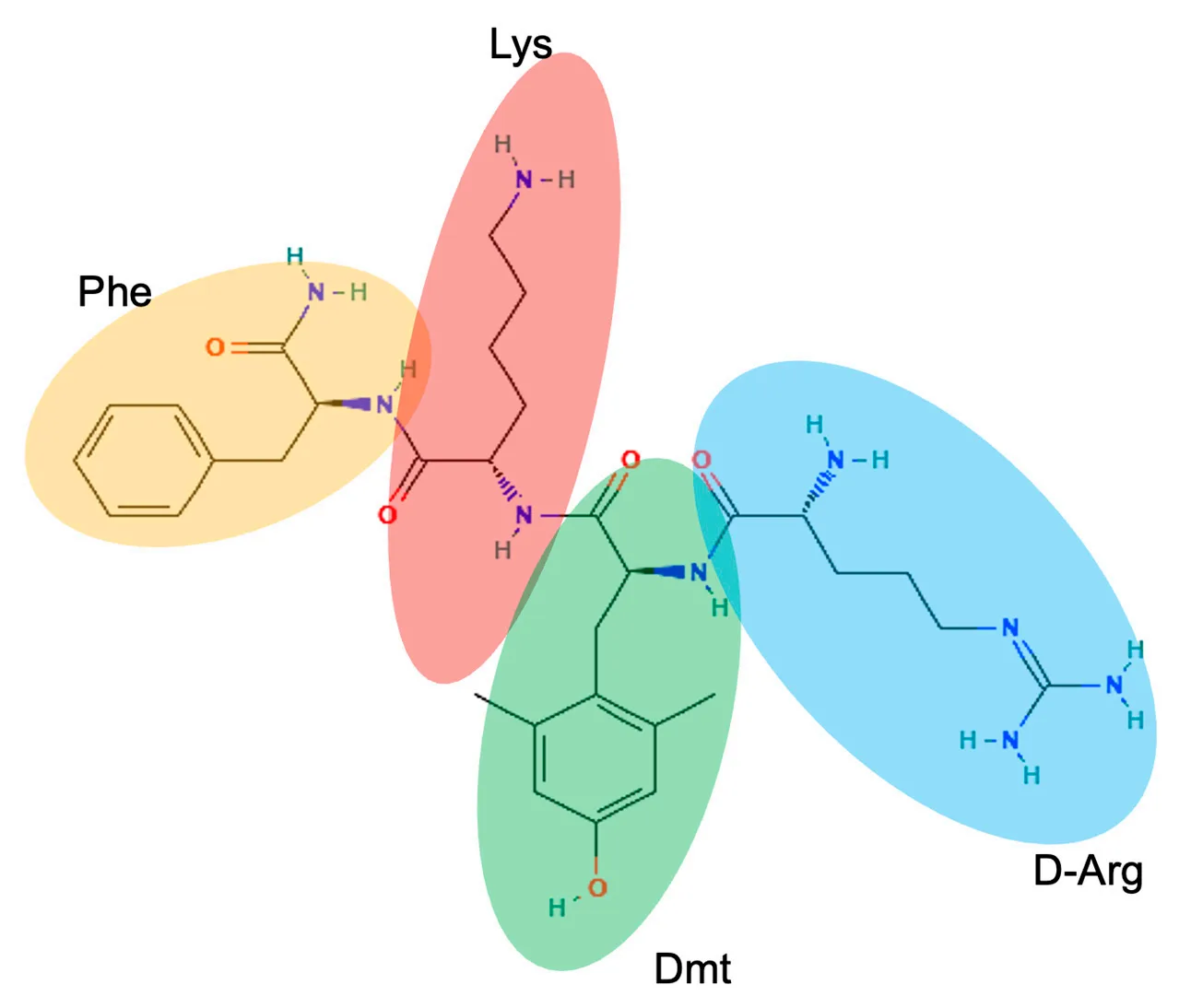
Notably, elamipretide retains high cellular permeability despite its cationic nature. This property is attributed to charge shielding by π-electron clouds in the aromatic rings of Dmt and phenylalanine, which reduces repulsion from the plasma membrane. Dmt also improves chemical stability by limiting oxidative modification, an important feature for peptides intended to function in oxidative mitochondrial environments.
Together, these structural characteristics allow elamipretide to reach mitochondria efficiently without relying on membrane potential–dependent uptake mechanisms. This distinguishes it from many other mitochondria-directed compounds and contributes to its widespread use as a research tool in mitochondrial biology.
Mechanism of Action: Stabilizing Cardiolipin and Restoring Bioenergetics
The biological activity of elamipretide is centered on its selective interaction with cardiolipin, a phospholipid found almost exclusively in the inner mitochondrial membrane. Cardiolipin plays a critical role in maintaining mitochondrial cristae architecture and supporting the assembly and activity of electron transport chain (ETC) complexes. These functions are essential for efficient ATP synthesis.
Under pathological conditions, cardiolipin is particularly susceptible to oxidative damage. Oxidized cardiolipin disrupts cristae structure, destabilizes ETC complexes, and promotes electron leakage, leading to excessive mitochondrial ROS production. In addition, cardiolipin oxidation facilitates mitochondrial membrane permeabilization and the release of pro-apoptotic factors such as cytochrome c.
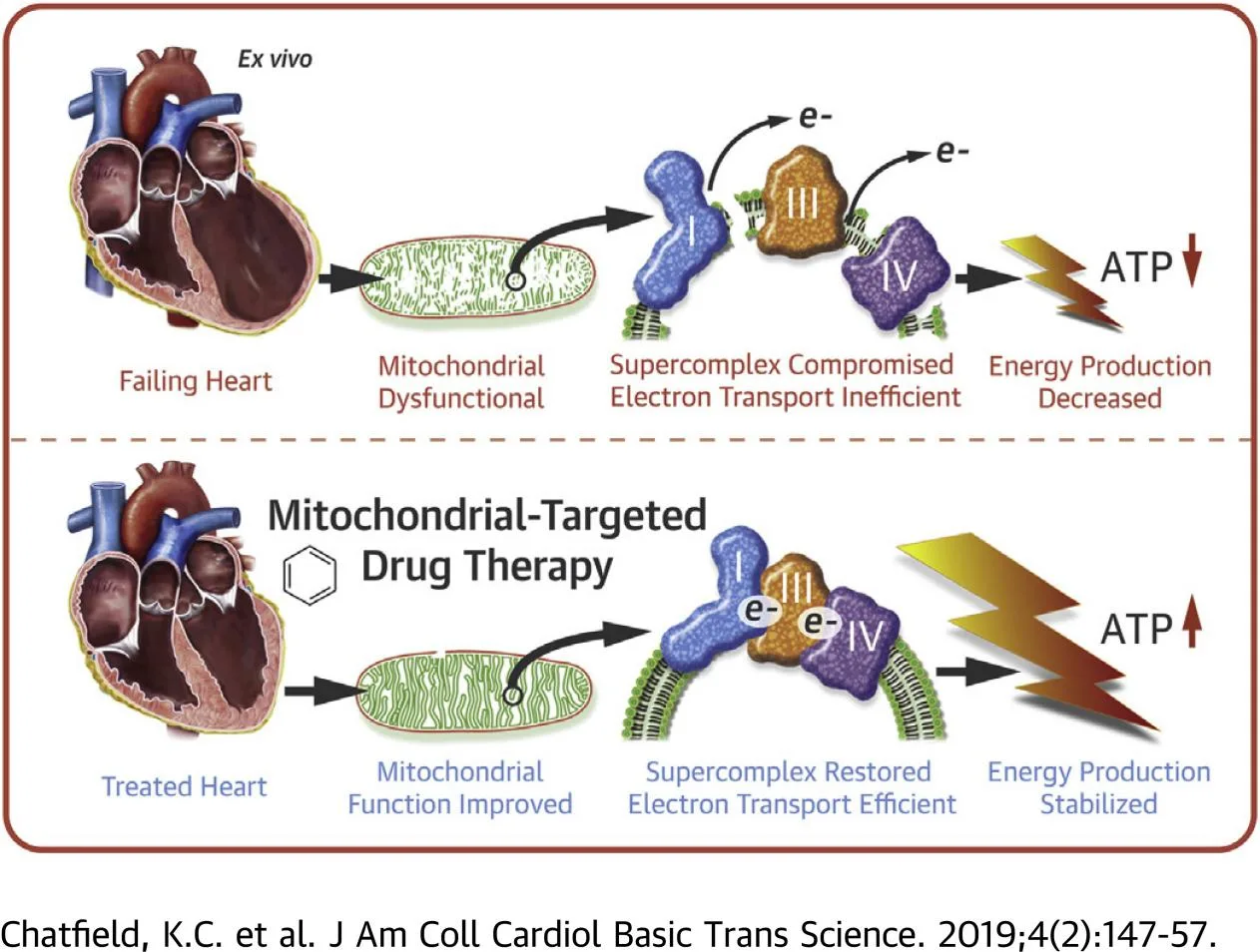
Elamipretide binds cardiolipin through electrostatic interactions, stabilizing the lipid and protecting it from peroxidation. By preserving cardiolipin integrity, the peptide helps maintain cristae structure and supports the proper assembly and function of ETC complexes I, III, and IV. This results in improved electron transfer efficiency and enhanced ATP production.
By reducing electron leakage from the ETC, elamipretide indirectly lowers mitochondrial ROS generation, limiting oxidative damage to mitochondrial DNA, proteins, and lipids. The peptide also helps preserve mitochondrial membrane potential (ΔΨm), a critical determinant of ATP synthesis. Furthermore, cardiolipin stabilization by elamipretide inhibits opening of the mitochondrial permeability transition pore (mPTP), a key event in ischemia–reperfusion injury and apoptosis.
Through these complementary mechanisms, elamipretide supports mitochondrial bioenergetics, reduces oxidative stress, and mitigates cell death signaling at the organelle level.
Preclinical Research Applications Across Disease Models
The cardiolipin-targeting mechanism of elamipretide has been extensively evaluated in preclinical models of diseases characterized by mitochondrial dysfunction. In cardiovascular research, animal models of heart failure have demonstrated that elamipretide improves left ventricular function, enhances ATP synthesis, and reduces myocardial fibrosis. These functional improvements are accompanied by normalization of mitochondrial morphology, reduced ROS formation, and attenuation of pathological cardiac remodeling.
In post-myocardial infarction models, elamipretide preserved mitochondrial integrity and calcium-handling proteins, helping prevent ventricular dilation and progression toward heart failure. Studies have also shown benefits in skeletal muscle mitochondria, highlighting potential relevance to exercise intolerance associated with chronic heart failure.
Renal disease models further illustrate the peptide’s utility in mitochondrial research. In ischemia–reperfusion injury, elamipretide restored mitochondrial structure and accelerated recovery of ATP levels in renal tissue. In models of diabetic nephropathy, the peptide reduced oxidative stress and fibrosis, supporting its use in studies of chronic metabolic kidney disease.
Neurodegenerative disease research has also leveraged elamipretide as a mitochondrial probe. In Alzheimer’s disease models, elamipretide protected neurons from amyloid-β–induced mitochondrial toxicity and supported neurite outgrowth. In Parkinson’s disease models, it preserved dopaminergic neurons by improving mitochondrial respiration and limiting oxidative damage.
Additionally, elamipretide has been studied in models of chemotherapy-induced cardiotoxicity, particularly doxorubicin-associated cardiac injury. By stabilizing cardiolipin and reducing ROS-mediated mitochondrial damage, the peptide attenuated myocardial apoptosis and fibrosis, making it a useful tool for investigating mitochondrial protection strategies in oncology-related cardiac dysfunction.
Translational and Clinical Insights Informing Research Design
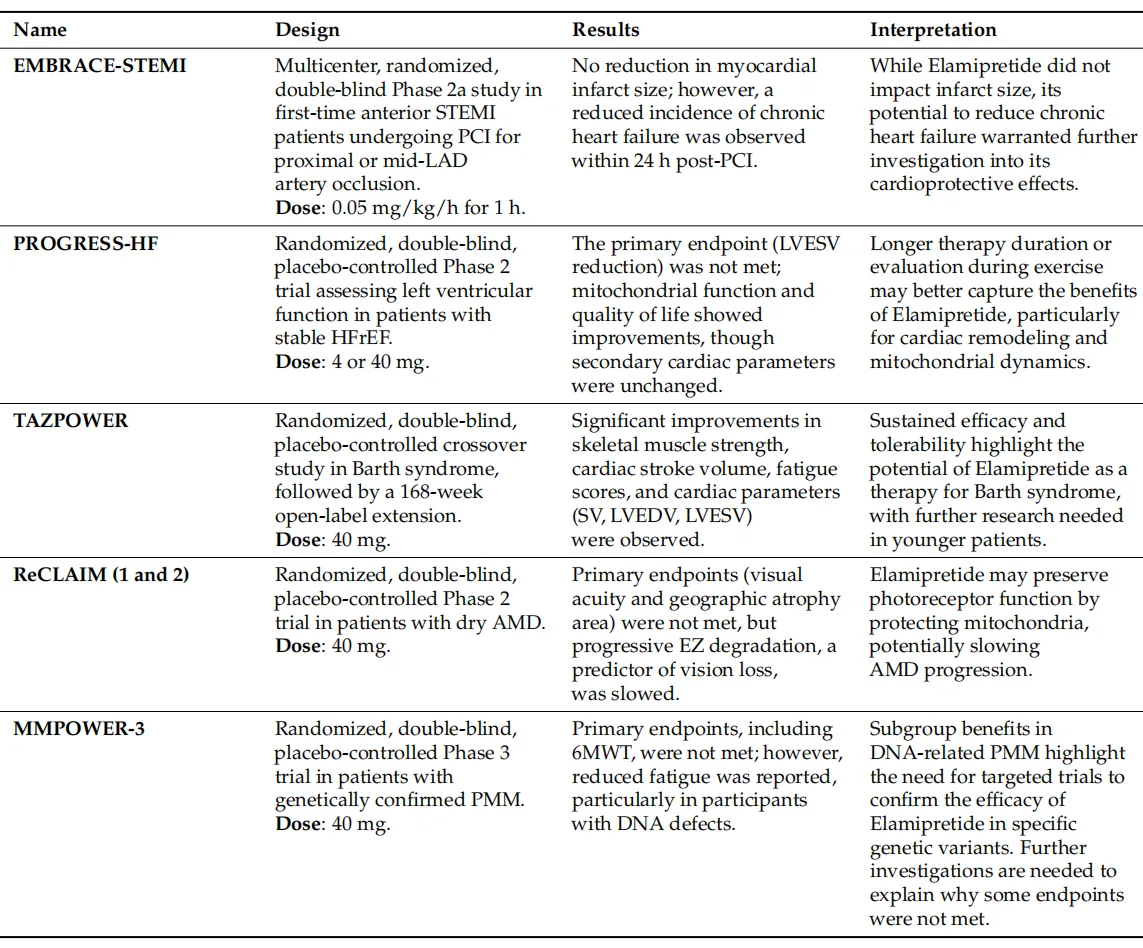
Clinical investigations of elamipretide provide important translational context for researchers studying mitochondrial therapeutics. The peptide has been evaluated in multiple clinical trials involving heart failure with reduced ejection fraction, Barth syndrome, primary mitochondrial myopathy, and dry age-related macular degeneration.
Although not all trials met their primary efficacy endpoints, several demonstrated improvements in mitochondrial function, fatigue, skeletal muscle performance, or patient-reported quality-of-life measures. Notably, studies in Barth syndrome—a genetic disorder driven by defects in cardiolipin remodeling—showed sustained improvements in cardiac and skeletal muscle parameters during long-term treatment. These findings highlight the importance of disease context and molecular etiology when targeting mitochondrial dysfunction.
Across clinical studies, elamipretide has exhibited a favorable safety and tolerability profile, with most adverse events limited to mild injection-site reactions and transient symptoms. Long-term exposure has not revealed major systemic safety concerns, reinforcing its suitability for extended experimental and translational research use.
For researchers, these clinical outcomes emphasize the need for careful endpoint selection, adequate treatment duration, and genetic or mechanistic stratification when evaluating mitochondrial-targeted interventions. Elamipretide’s extensive preclinical and clinical characterization makes it a valuable reference compound for cardiolipin-focused studies and broader investigations into mitochondrial biology.
Conclusion
Elamipretide is a well-characterized mitochondria-targeted peptide with a distinct mechanism centered on cardiolipin stabilization. Its rationally designed structure enables efficient mitochondrial localization, while its interaction with cardiolipin preserves cristae architecture, enhances bioenergetic efficiency, and reduces oxidative stress. Extensive preclinical studies across cardiovascular, neurological, renal, and metabolic disease models demonstrate its value as a research tool for investigating mitochondrial dysfunction. Clinical findings further inform experimental design and translational relevance. Collectively, these attributes position elamipretide as a powerful peptide for advancing research in mitochondrial biology and disease mechanisms.
Reference
Tung, C., Varzideh, F., Farroni, E., Mone, P., Kansakar, U., Jankauskas, S. S., & Santulli, G. (2025). Elamipretide: a review of its structure, mechanism of action, and therapeutic potential. International Journal of Molecular Sciences, 26(3), 944.https://doi.org/10.3390/ijms26030944
Sabbah, H. N., Alder, N. N., Sparagna, G. C., Bruce, J. E., Stauffer, B. L., Chao, L. H., … & Marcinek, D. J. (2025). Contemporary insights into elamipretide’s mitochondrial mechanism of action and therapeutic effects. Biomedicine & Pharmacotherapy, 187, 118056.https://doi.org/10.1016/j.biopha.2025.118056
Chatfield, K. C., Sparagna, G. C., Chau, S., Phillips, E. K., Ambardekar, A. V., Aftab, M., … & Stauffer, B. L. (2019). Elamipretide improves mitochondrial function in the failing human heart. JACC: Basic to Translational Science, 4(2), 147-157.https://doi.org/10.1016/j.jacbts.2018.12.005
Sabbah, H. N. (2022). Elamipretide for Barth syndrome cardiomyopathy: gradual rebuilding of a failed power grid. Heart failure reviews, 27(5), 1911-1923.https://doi.org/10.1007/s10741-021-10177-8
 LinkPeptide
LinkPeptide