Peptide Cancer Vaccines Revisited: Overcoming Biological Limitations Through Rational Design
Abstract
Peptide-based cancer vaccines were once regarded as a promising yet ultimately unsuccessful immunotherapeutic strategy, largely due to limited clinical efficacy in early trials. However, these outcomes reflected fundamental biological and design limitations rather than intrinsic shortcomings of peptide antigens. Advances in cancer immunology have clarified the essential requirements for effective cytotoxic T lymphocyte (CTL) priming, including dendritic cell cross-presentation, CD4⁺ T-cell help, and appropriate innate immune activation. In parallel, progress in peptide chemistry and formulation has enabled the development of long and multi-epitope peptides capable of overcoming MHC restriction and inducing durable T-cell memory. Coupled with rational adjuvant selection and targeted delivery systems, modern peptide vaccines now function as precision immunotherapy platforms rather than simplistic antigen carriers. This article examines the limitations that constrained early peptide cancer vaccines, explores the mechanistic insights that reshaped their design, and discusses how these advances position peptide-based vaccines as viable components of next-generation cancer immunotherapy.
Why Early Peptide Cancer Vaccines Fell Short—and Why the Field Is Re-Emerging
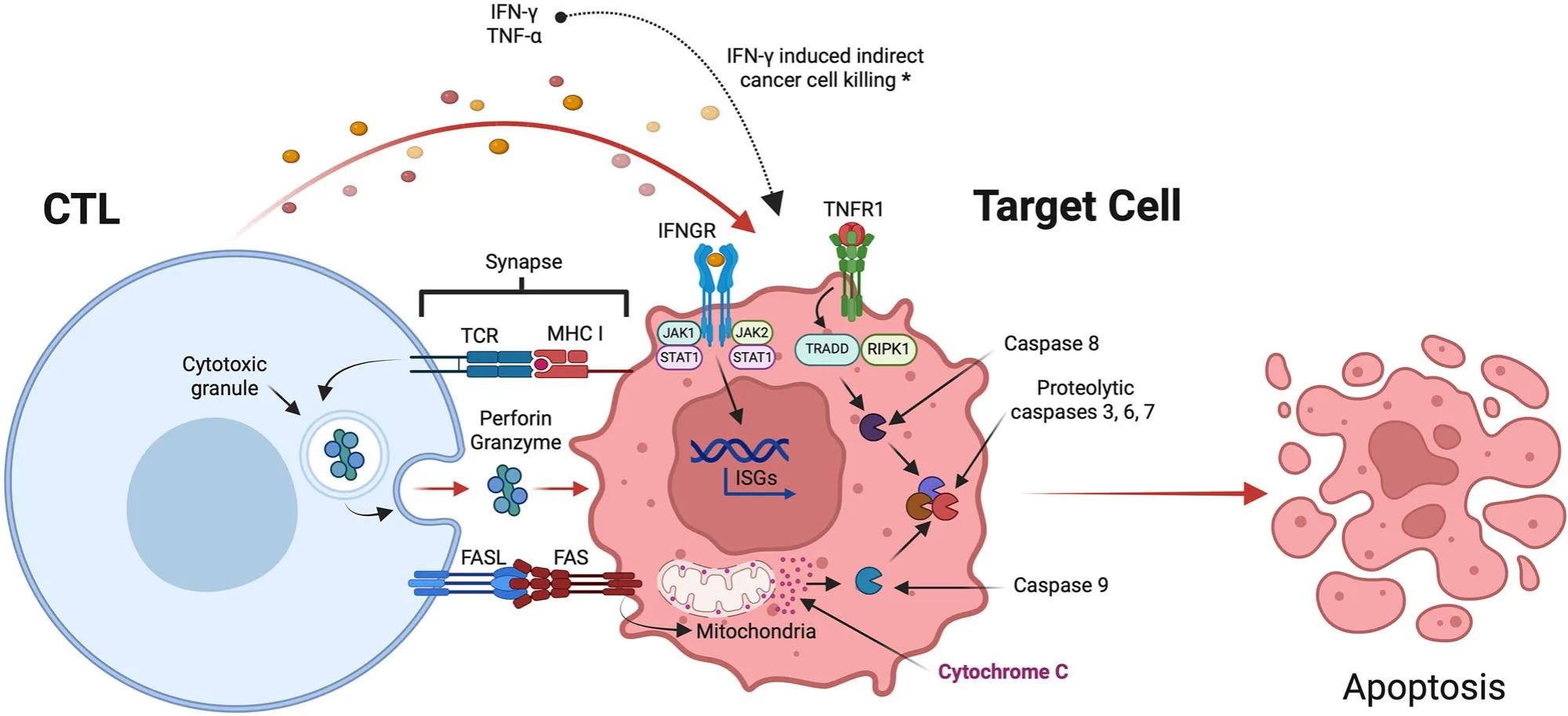
Durable antitumor immunity ultimately depends on the generation of robust, antigen-specific CD8⁺ cytotoxic T lymphocyte (CTL) responses. CTLs play a central role in immune-mediated tumor control by directly eliminating malignant cells through cytolytic mechanisms and by shaping the tumor microenvironment via cytokine secretion. Across multiple cancer types, high intratumoral CTL infiltration has been consistently associated with improved prognosis and long-term survival, underscoring the importance of therapeutic strategies capable of inducing sustained CTL immunity.
Peptide-based cancer vaccines emerged early as a rational and highly attractive approach to achieve this goal. Their conceptual appeal lies in their molecular precision, favorable safety profile, and compatibility with scalable, chemically defined manufacturing. However, despite strong mechanistic rationale and encouraging preclinical studies, early clinical trials largely failed to deliver meaningful therapeutic benefit. These disappointing outcomes led to the widespread perception that peptide vaccines were inherently ineffective in the oncologic setting.
Importantly, this perception stemmed not from intrinsic shortcomings of peptide antigens themselves, but from fundamental limitations in first-generation vaccine design. Chief among these were weak immunogenicity, strict MHC (HLA) restriction that severely limited patient coverage, insufficient engagement of CD4⁺ T-cell help, and an inability to counteract the profoundly immunosuppressive tumor microenvironment (TME). Most early formulations relied on short, minimal epitopes delivered without adequate consideration of antigen processing, dendritic cell licensing, or T-cell exhaustion dynamics.
Over the past decade, advances in cancer immunology, antigen presentation biology, and peptide chemistry have fundamentally altered our understanding of how effective T-cell immunity is generated and maintained. These insights have clarified why earlier peptide vaccines underperformed and, more importantly, how their limitations can now be systematically addressed. Rather than representing a failed modality, peptide cancer vaccines were introduced before the field possessed the necessary biological and technological maturity. Modern strategies now position them not as a revival of outdated concepts, but as a re-engineered immunotherapy platform built on mechanistic precision.
Biological Requirements for Effective CTL Priming: Lessons from Early Limitations
The failure of early peptide cancer vaccines can be traced to an incomplete appreciation of the biological requirements for effective CD8⁺ cytotoxic T lymphocyte (CTL) priming. CTL activation is not a passive consequence of antigen exposure, but a tightly regulated, multi-step process that depends on precise coordination between antigen-presenting cells, costimulatory pathways, and inflammatory signals. A central limitation of first-generation peptide vaccines was their inability to consistently engage this full activation cascade.
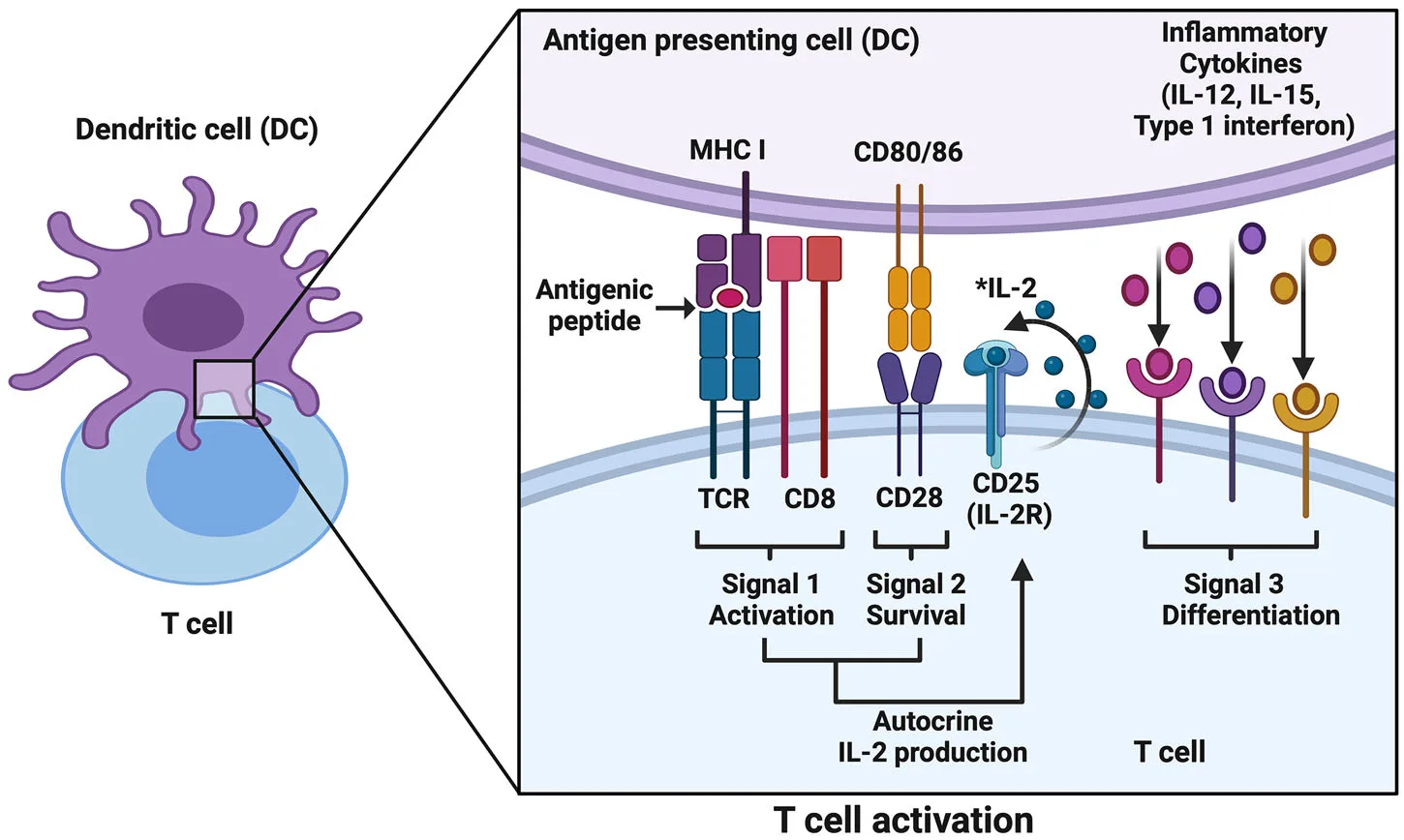
Productive CTL priming requires three distinct signals delivered by professional antigen-presenting cells, particularly dendritic cells (DCs). Signal 1 involves recognition of peptide–MHC class I complexes by the T-cell receptor. However, antigen recognition alone is insufficient. Signal 2, provided through costimulatory interactions such as CD80/86–CD28, is essential for clonal expansion and survival. Signal 3 consists of pro-inflammatory cytokines, including IL-12, IL-15, and type I interferons, which drive CTL differentiation into functional effector and memory populations. Early peptide vaccines often delivered antigen in isolation, failing to reliably provide costimulation or inflammatory context, resulting in weak or tolerogenic T-cell responses.
Another major limitation was inadequate engagement of CD4⁺ T helper cells, which are now recognized as critical regulators of CTL immunity. CD4⁺ T cells license DCs through CD40–CD40L interactions, enhancing antigen presentation, cytokine production, and costimulatory molecule expression. Without this licensing step, CTL responses are typically short-lived, poorly differentiated, and highly susceptible to exhaustion—particularly within the tumor microenvironment. Many early vaccine formulations excluded CD4⁺ helper epitopes altogether, reflecting a reductionist focus on minimal CTL peptides.
Advances in immunology have made clear that effective peptide vaccines must be designed to activate DCs, recruit CD4⁺ T-cell help, and support durable CTL memory. Recognizing and addressing these biological limitations has fundamentally reshaped modern peptide vaccine strategies, shifting the field from empirical antigen delivery toward rational, mechanism-driven immunotherapy design.
Breaking MHC Restriction: Long and Multi-Epitope Peptides as a Design Solution
Among the most significant limitations of early peptide cancer vaccines was their reliance on short, minimal epitopes restricted to a single major histocompatibility complex (MHC) allele. MHC polymorphism is extensive in human populations, meaning that a peptide capable of binding one HLA molecule may be completely invisible to another. As a result, many early vaccine strategies were inherently applicable to only a narrow subset of patients, severely constraining both clinical efficacy and translational scalability.
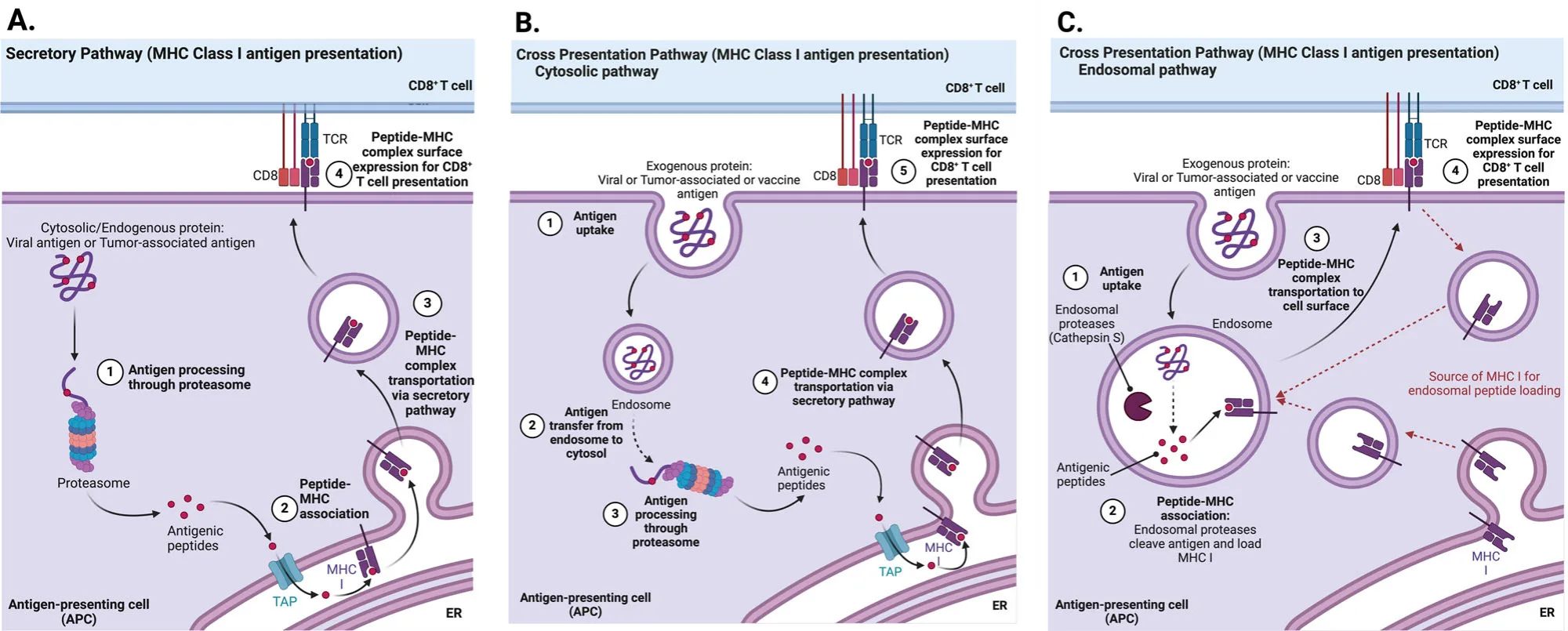
Short peptides, typically 8–10 amino acids in length, were originally favored because they directly bind MHC class I molecules. However, this approach introduced multiple additional limitations. Minimal epitopes can be loaded onto non-professional antigen-presenting cells, bypassing dendritic cell processing and increasing the risk of suboptimal T-cell activation or tolerance. Moreover, repeated exposure to persistent short peptides—particularly in depot-forming adjuvant systems—can drive T-cell dysfunction and exhaustion rather than durable antitumor immunity.
The introduction of synthetic long peptides (SLPs) has addressed many of these challenges. SLPs, generally 25–35 amino acids in length, require intracellular processing by professional antigen-presenting cells, thereby enforcing dendritic cell involvement and promoting cross-presentation through physiologically relevant pathways. This design feature not only enhances CTL priming but also reduces the risk of tolerance associated with direct peptide loading. Importantly, long peptides can simultaneously contain CD8⁺ CTL and CD4⁺ T helper epitopes, directly addressing the earlier limitation of inadequate helper T-cell engagement.
Beyond peptide length, multi-peptide and multi-epitope formulations further mitigate MHC restriction by targeting multiple HLA supertypes within a single vaccine. By broadening epitope coverage, these strategies increase population applicability and promote polyfunctional T-cell responses that are more resilient to immune escape. Modified or heteroclitic peptides have also been explored to enhance immunogenicity, although their use requires careful validation to ensure effective recognition of native tumor antigens.
Collectively, these advances reflect a paradigm shift away from reductionist epitope selection toward integrated, population-aware vaccine design. By systematically addressing MHC restriction and antigen-processing limitations, modern peptide vaccines are engineered to generate broader, more durable, and clinically relevant antitumor T-cell immunity.
Engineering Immunogenicity: Adjuvants and Delivery Systems that Overcome Peptide Limitations
Even with optimized antigen selection and epitope design, peptide vaccines face an inherent limitation: peptides alone are weak immunogens. Unlike live or replicating platforms, synthetic peptides lack intrinsic danger signals and therefore fail to adequately activate the innate immune system. Without appropriate innate stimulation, dendritic cells remain poorly activated, costimulatory signals are insufficient, and antigen presentation may skew toward tolerance rather than effective T-cell priming. Consequently, adjuvants and delivery systems are not ancillary components of peptide vaccines, but central determinants of their immunological outcome.
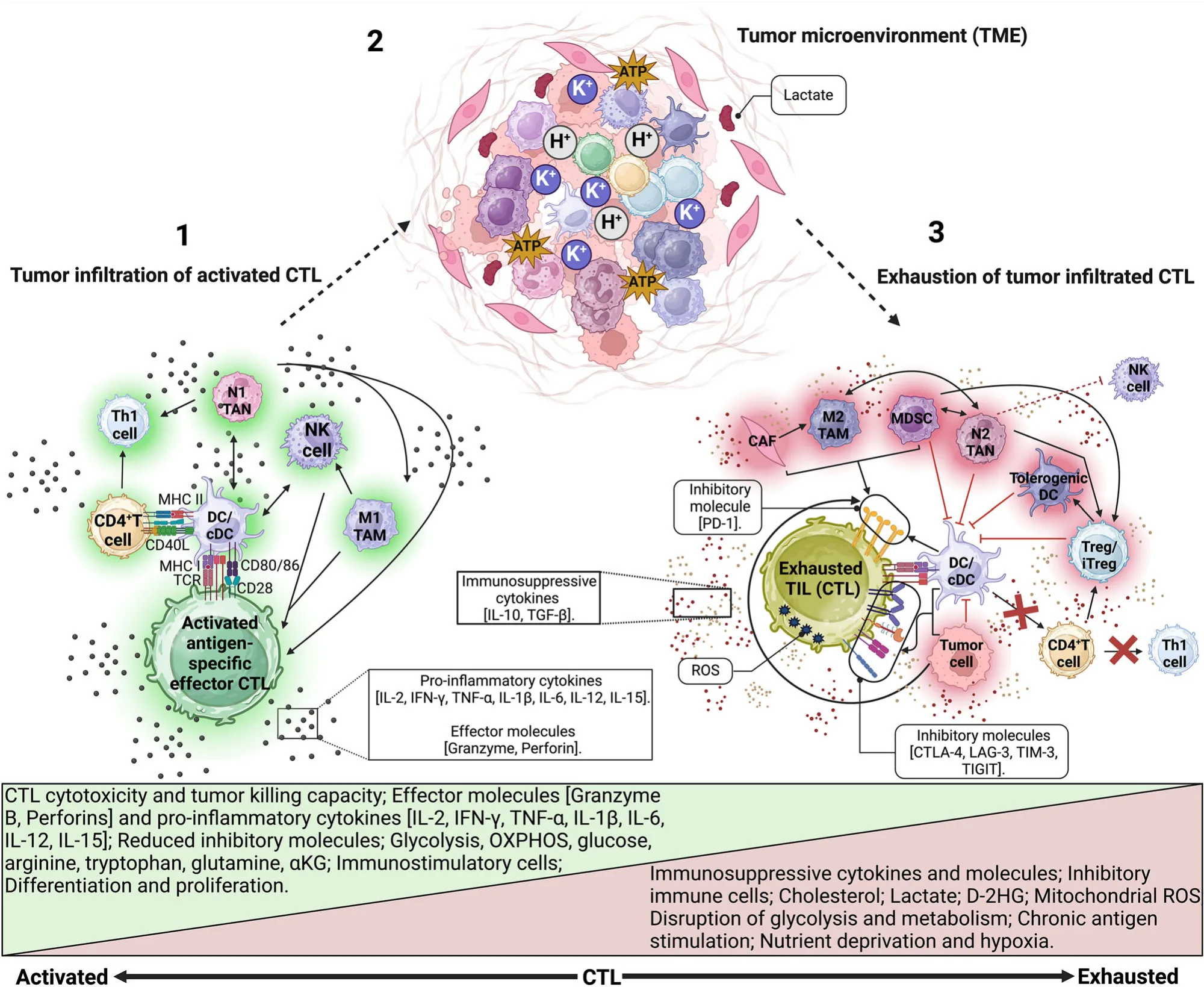
Modern peptide vaccine strategies increasingly rely on pattern-recognition receptor (PRR) agonists, particularly toll-like receptor (TLR) ligands, to overcome this limitation. Agonists of TLR3, TLR4, TLR7/8, and TLR9 promote dendritic cell maturation, upregulation of costimulatory molecules, and secretion of pro-inflammatory cytokines that favor Th1-biased and CTL-dominant responses. Importantly, different TLR pathways engage distinct antigen cross-presentation mechanisms, highlighting the value of rational adjuvant selection or combination to optimize CTL priming and memory formation.
Traditional depot-forming adjuvants, while effective at prolonging antigen exposure, have revealed additional limitations. Persistent antigen presentation can sequester activated T cells at the injection site, drive chronic stimulation, and ultimately promote T-cell exhaustion. These observations have prompted a shift away from prolonged antigen depots toward delivery strategies that favor transient, lymph node–targeted immune activation.
Nanoparticle-based delivery systems have emerged as a promising solution to these challenges. Platforms such as calcium phosphate, chitosan, and biodegradable polymer nanoparticles protect peptides from degradation, enhance uptake by antigen-presenting cells, and improve trafficking to draining lymph nodes. Critically, nanoparticle size and composition can be tuned to preferentially engage cross-presenting dendritic cell subsets, thereby amplifying CTL responses while supporting CD4⁺ T-cell help.
By integrating immunostimulatory adjuvants with rational delivery systems, modern peptide vaccines overcome long-standing immunogenicity limitations and establish the inflammatory context required for durable antitumor T-cell immunity.
From Shared Tumor Antigens to Neoantigens: Toward Precision Peptide Immunotherapy
As peptide vaccine design has matured, attention has increasingly shifted toward antigen selection as a critical determinant of clinical success. Historically, most peptide cancer vaccines have targeted tumor-associated antigens (TAAs)—self-derived proteins that are aberrantly overexpressed or dysregulated in malignant cells. While TAAs remain attractive due to their shared expression across patient populations, immune tolerance represents a persistent limitation, often resulting in low-affinity T-cell responses that struggle to overcome immunosuppressive tumor environments.
The emergence of neoantigen-based peptide vaccines has revitalized the field by directly addressing this limitation. Neoantigens arise from tumor-specific genetic alterations and are absent from normal tissues, rendering them intrinsically non-self and highly immunogenic. Peptide vaccines are uniquely well suited to target these antigens, as they enable precise inclusion of mutation-containing epitopes that would be diluted or ignored in whole-protein or nucleic acid–based platforms. Early clinical studies have demonstrated that long neoantigen peptides can elicit robust, polyfunctional CD4⁺ and CD8⁺ T-cell responses, including durable memory populations.
However, personalized neoantigen vaccines introduce new practical limitations, including manufacturing complexity, cost, and scalability. In response, growing interest has focused on public or shared neoantigens, such as recurrent KRAS mutations, which offer a compromise between immunogenicity and broad applicability. These shared targets may enable off-the-shelf peptide vaccine strategies while retaining tumor specificity.
Looking forward, peptide cancer vaccines are unlikely to function as standalone therapies. Instead, their greatest potential lies in combination regimens, integrating immune checkpoint blockade, optimized adjuvants, and rational delivery systems. By systematically addressing historical biological and translational limitations, peptide vaccines are now positioned as flexible, precision immunotherapy platforms capable of complementing and enhancing existing cancer treatment paradigms.
Peptide Vaccines Reconsidered: From Conceptual Simplicity to Mechanistic Precision
Peptide cancer vaccines have entered a new phase of development defined by mechanistic clarity, chemical precision, and growing clinical maturity. The limitations that constrained early efforts—poor immunogenicity, MHC restriction, lack of CD4⁺ T-cell help, and immune suppression within the tumor microenvironment—are now better understood and increasingly addressable through rational design. Advances in long and multi-epitope peptides, adjuvant engineering, and targeted delivery systems have transformed peptide vaccines from simplistic antigen carriers into sophisticated immunotherapy platforms. Realizing their full potential will require continued interdisciplinary collaboration between immunology, peptide chemistry, and clinical oncology, ensuring that biological insight and chemical innovation translate into durable, patient-relevant antitumor immunity.
Reference
Shah, B. A., Holden, J. A., Lenzo, J. C., Hadjigol, S., & O’Brien-Simpson, N. M. (2025). Multi-disciplinary approaches paving the way for clinically effective peptide vaccines for cancer. npj Vaccines, 10(1), 68.https://doi.org/10.1038/s41541-025-01118-9
Lin, C. L., & Kao, J. H. (2020). Hepatitis B: immunization and impact on natural history and cancer incidence. Gastroenterology Clinics, 49(2), 201-214.https://doi.org/10.1016/j.gtc.2020.01.010
Rojas, L. A., Sethna, Z., Soares, K. C., Olcese, C., Pang, N., Patterson, E., … & Balachandran, V. P. (2023). Personalized RNA neoantigen vaccines stimulate T cells in pancreatic cancer. Nature, 618(7963), 144-150.https://doi.org/10.1038/s41586-023-06063-y
Kondo, S., Shimizu, T., Koyama, T., Sato, J., Iwasa, S., Yonemori, K., … & Yamamoto, N. (2021). First‐in‐human study of the cancer peptide vaccine TAS0313 in patients with advanced solid tumors. Cancer Science, 112(4), 1514-1523.https://doi.org/10.1111/cas.14765
 LinkPeptide
LinkPeptide