Unlocking Bioactive Peptide Potential: How Advanced Fractionation Strategies Enhance Functionality
Abstract
Bioactive peptides derived from food proteins have gained increasing attention due to their diverse health-promoting properties and expanding applications in functional foods, nutraceuticals, cosmetics, and biomedicine. However, crude protein hydrolysates often exhibit inconsistent or diluted bioactivity because they contain complex mixtures of peptides with varying structures and functions. Fractionation has therefore emerged as a critical strategy to enhance peptide performance by separating and enriching peptides with favorable physicochemical characteristics. This blog reviews recent advances in bioactive peptide fractionation, focusing on membrane-based methods, charge-driven techniques such as isoelectric focusing and ion exchange chromatography, and size-exclusion approaches. It further highlights the growing importance of integrated, multi-step fractionation strategies that combine size and charge selectivity to improve bioactivity targeting. By examining current challenges and future development trends, this article underscores the role of fractionation as a key bridge between peptide generation and functional application, enabling the rational development of high-value bioactive peptide ingredients.
Why Fractionation Defines Bioactive Peptide Functionality
Bioactive peptides are highly heterogeneous by nature. Even when generated from the same protein source under identical enzymatic hydrolysis conditions, peptide mixtures contain numerous sequences that differ in molecular weight, amino acid composition, charge, hydrophobicity, and structural conformation. These differences strongly influence how peptides interact with biological targets such as enzymes, receptors, microbial membranes, and reactive oxygen species.
When protein hydrolysates are assessed in their unfractionated form, the observed bioactivity often reflects an averaged effect of many peptides acting simultaneously. Highly active peptides may be masked by inactive or weakly active components, resulting in diluted or inconsistent functional outcomes. In some cases, peptide–peptide interactions can further obscure activity, making it difficult to accurately evaluate biological potential.
Fractionation overcomes this limitation by separating peptides into more homogeneous groups based on key physicochemical properties, including size and charge. Rather than serving solely as a purification step, fractionation acts as a functional enhancement strategy that allows bioactive peptides to be enriched and more clearly expressed. Numerous studies have shown that fractionated peptide samples frequently exhibit superior antioxidant, antihypertensive, antidiabetic, and antimicrobial activities compared with unfractionated hydrolysates.
Mechanistically, fractionation concentrates peptides that are structurally better suited to interact with specific biological targets. Smaller peptides may access enzyme active sites more efficiently, while charged or hydrophobic peptides often display improved binding and scavenging capabilities. As a result, fractionation plays a central role in transforming complex peptide mixtures into functionally optimized ingredients for research and application.
Ultrafiltration and Membrane Technologies: Linking Peptide Size to Bioactivity
Among current peptide fractionation technologies, membrane-based methods—particularly ultrafiltration—are the most widely applied due to their operational simplicity, scalability, and compatibility with food and biomedical systems. Ultrafiltration separates peptides primarily according to molecular weight using semi-permeable membranes with defined molecular weight cut-off (MWCO) values, commonly ranging from 1 to 100 kDa.
In practice, protein hydrolysates are passed through membranes with decreasing MWCOs, yielding peptide fractions of different size ranges. Numerous studies demonstrate that low-molecular-weight fractions, especially those below 1–3 kDa, frequently exhibit enhanced antioxidant, angiotensin-converting enzyme (ACE) inhibitory, and dipeptidyl peptidase-IV (DPP-IV) inhibitory activities. These peptides are often more effective because their small size allows easier access to enzyme active sites and improved interaction with target molecules.
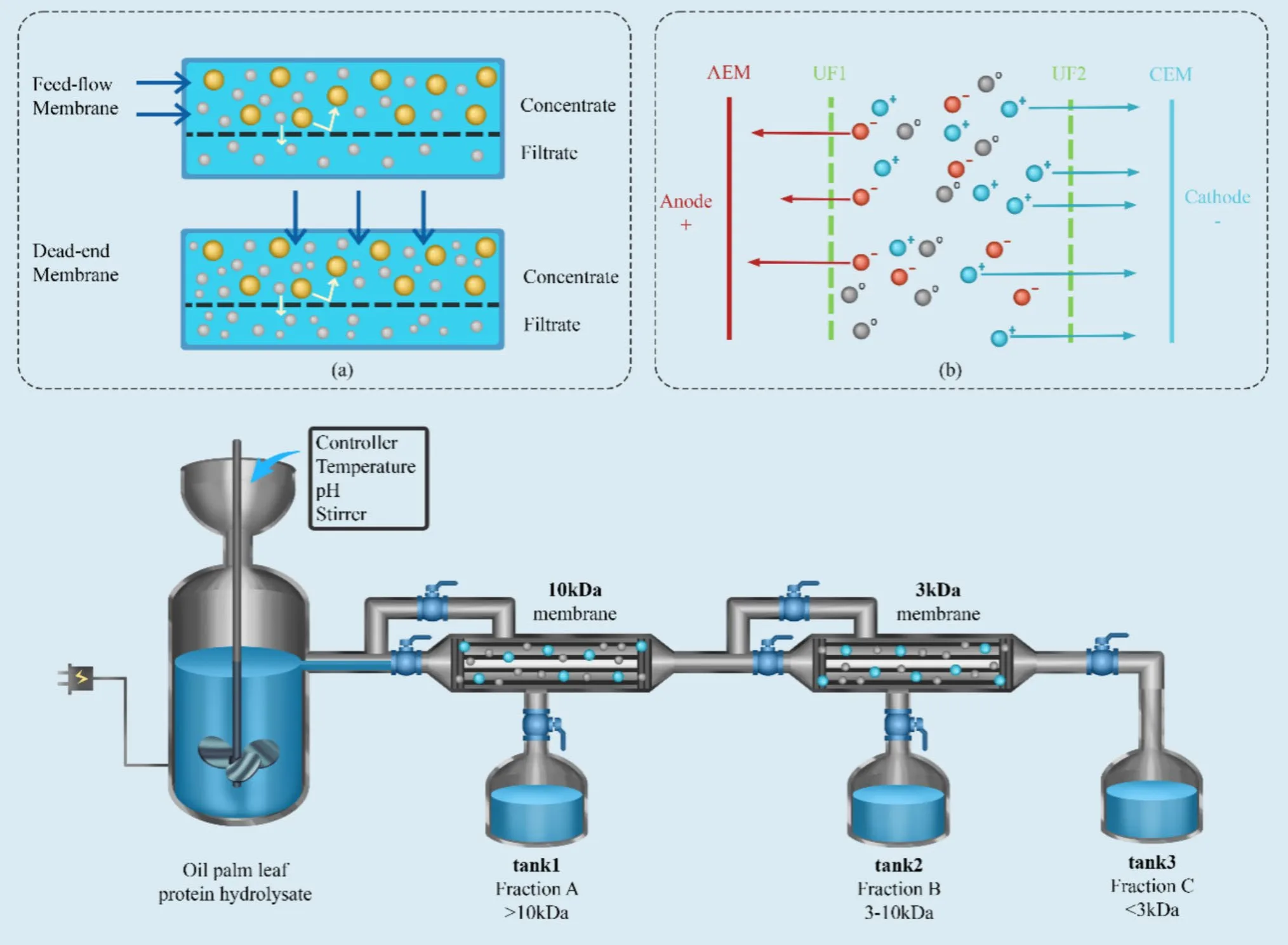
However, the relationship between peptide size and bioactivity is not linear. In several systems, medium-sized fractions (3–10 kDa) outperform smaller peptides, highlighting the importance of amino acid composition and sequence characteristics rather than molecular weight alone. This underscores the need for careful fraction selection rather than assuming that smaller peptides are always superior.
Recent advances have further expanded the potential of membrane-based fractionation. Integrated systems such as enzymatic membrane reactors (EMR) combine hydrolysis and separation in a single process, while electrodialysis with ultrafiltration membranes (EDUF) enables simultaneous separation by size and charge. Despite challenges such as membrane fouling and operational costs, ultrafiltration remains a cornerstone technology for producing bioactive peptide fractions at scale.
Charge-Driven Separation: Isoelectric Focusing and Ion Exchange Strategies
While membrane-based methods focus primarily on peptide size, charge-based fractionation techniques provide an additional layer of selectivity by exploiting differences in peptide net charge and isoelectric point. These approaches are particularly valuable for isolating peptides whose biological activity is closely linked to electrostatic interactions.
Isoelectric focusing (IEF) separates peptides according to their isoelectric point (pI), the pH at which a peptide carries no net charge. Under an applied electric field and a stable pH gradient, peptides migrate until they reach their pI, allowing precise separation of acidic, neutral, and basic fractions. Studies have shown that basic or cationic peptide fractions often display strong antimicrobial, antihypertensive, and anti-inflammatory activities, largely due to their ability to interact with negatively charged biological membranes or enzyme surfaces. In contrast, acidic or neutral fractions may dominate antioxidant activity, depending on amino acid composition and sequence structure.
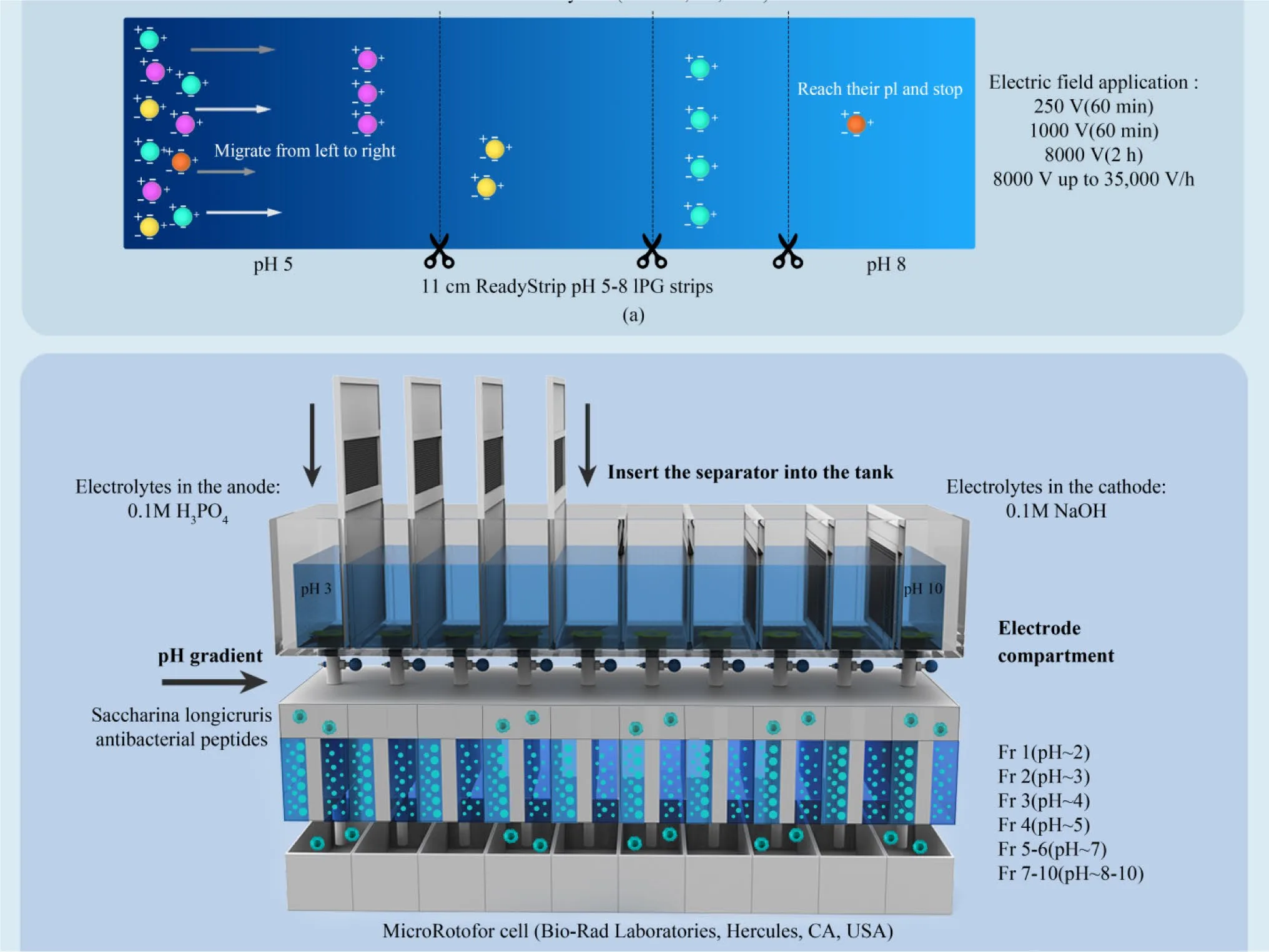
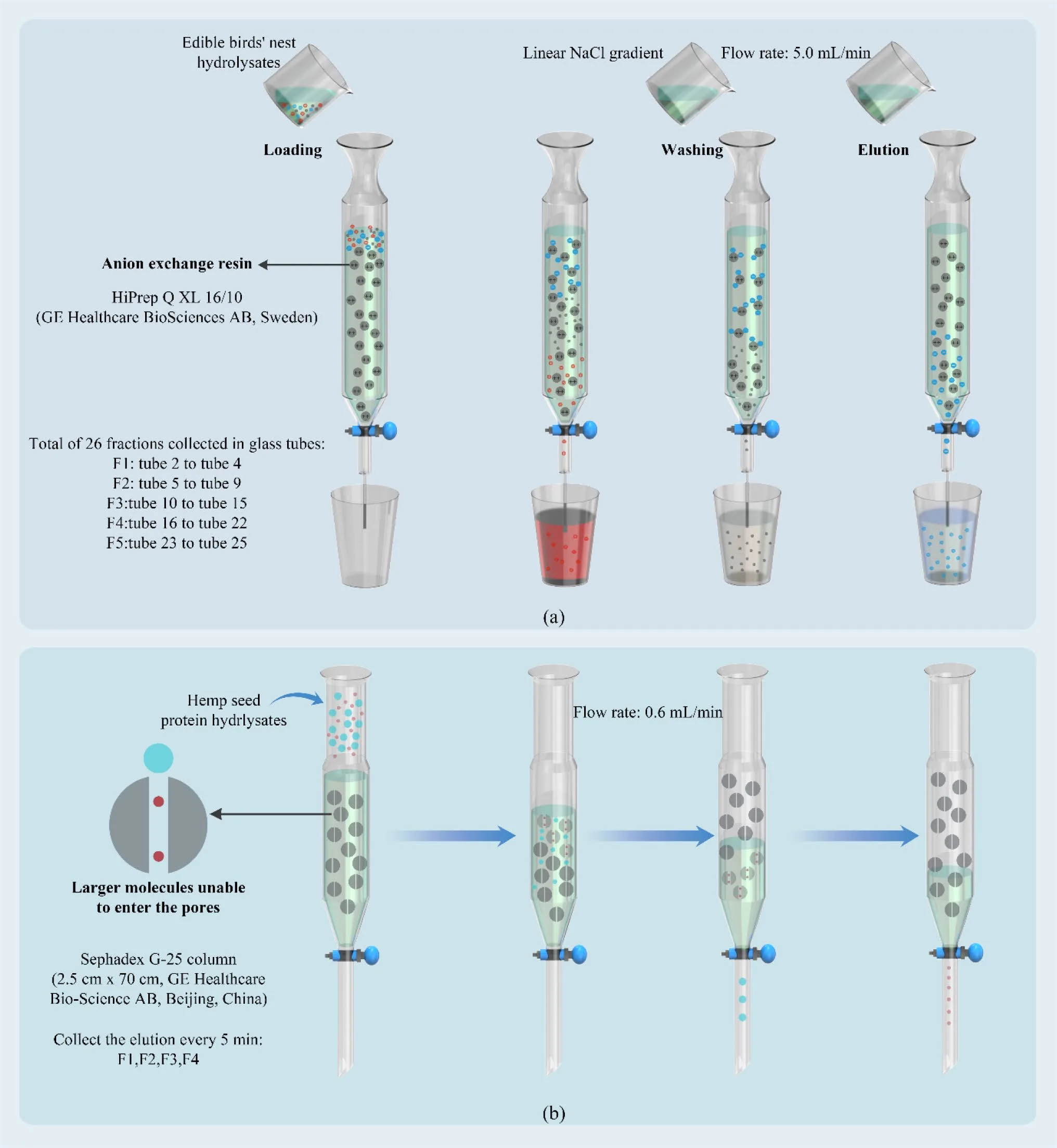
Ion exchange chromatography (IEC) applies similar principles in a column-based format, using charged resins to selectively bind peptides. Cation exchange chromatography enriches positively charged peptides, while anion exchange targets negatively charged species. By adjusting pH and ionic strength, peptides can be eluted in distinct fractions with differing bioactivities. IEC has been widely used to isolate peptides with angiotensin-converting enzyme inhibition, antibacterial activity, and enzyme-modulating properties.
Although charge-based methods offer high selectivity and strong structure–activity insights, their higher cost, operational complexity, and limited scalability currently restrict their use primarily to research, screening, and high-value peptide applications.
Beyond Single Techniques: Size-Exclusion and Integrated Fractionation Approaches
Size-exclusion chromatography (SEC), also known as gel filtration chromatography, provides a complementary size-based separation approach that is often used to further refine peptide fractions obtained by other methods. Unlike ultrafiltration, which relies on membrane cut-offs, SEC separates peptides based on their hydrodynamic volume as they pass through a porous gel matrix. Larger peptides elute first, while smaller peptides penetrate the gel pores and elute later, enabling fine resolution across narrow molecular weight ranges.
Many studies report that later-eluting, lower-molecular-weight fractions obtained by SEC exhibit enhanced bioactivities, including antioxidant, angiotensin-converting enzyme inhibitory, antidiabetic, and antimicrobial effects. However, as observed with ultrafiltration, bioactivity does not always correlate strictly with peptide size. In some cases, fractions with intermediate molecular weights show superior functional performance, reinforcing the importance of peptide sequence and composition.
These findings highlight a key limitation of single-method fractionation: no single technique can fully resolve the complexity of peptide hydrolysates. As a result, combined fractionation strategies have emerged as a best practice in bioactive peptide research. Common workflows include ultrafiltration followed by SEC for size refinement, ion exchange chromatography followed by SEC for charge-to-size separation, or multi-step sequences such as UF–IEC–SEC that integrate molecular weight and charge selectivity.
By applying fractionation in stages, researchers can achieve higher purity, improved activity targeting, and reduced fraction overlap. Such integrated approaches enable more precise identification of bioactive peptides and support the development of functionally optimized peptide ingredients from complex protein sources.
From Laboratory Precision to Industrial Reality: Challenges and Future Perspectives
Despite significant progress in bioactive peptide fractionation, several challenges continue to limit large-scale translation and commercial adoption. One of the most pressing issues is the balance between separation precision and industrial feasibility. Highly selective methods such as ion exchange chromatography and isoelectric focusing offer excellent resolution but are often constrained by high equipment costs, energy consumption, and operational complexity. Conversely, scalable approaches like ultrafiltration may sacrifice selectivity, particularly when peptides share similar molecular weights or physicochemical properties.
Another critical concern is the preservation of bioactivity during fractionation. Extreme pH conditions, high salt concentrations, or prolonged processing can disrupt peptide structure, leading to aggregation or functional loss. Additionally, most bioactivity assessments rely heavily on in vitro assays, which do not always translate directly to in vivo efficacy due to digestion, absorption, and metabolism effects.
Looking ahead, future research is expected to focus on developing mild, low-energy, and cost-effective fractionation technologies that better preserve peptide functionality while remaining scalable. Integrated systems, intelligent membrane design, and AI-assisted process optimization may improve selectivity and efficiency. The incorporation of omics tools such as proteomics and metabolomics is also likely to guide more targeted fractionation strategies by linking peptide structure directly to biological function.
Ultimately, advancing bioactive peptide fractionation will require not only technical innovation but also standardized workflows that bridge laboratory research and industrial production, enabling consistent quality and reliable functional performance.
Reference
Kan, M., Ter, Z. Y., Sofian-Seng, N. S., Chang, L. S., Wang, S., & Lim, S. J. (2025). Recent Advances on Bioactive Peptide Fractionation Methods. Food and Bioprocess Technology, 1-28.
https://doi.org/10.1007/s11947-025-03893-8
Abdelhedi, O., Mora, L., Jridi, M., Toldra, F., & Nasri, M. (2024). Proteolysis coupled with membrane separation for the isolation of bioactive peptides from defatted smooth hound byproduct proteins. Waste and Biomass Valorization, 15(4), 1959-1974.
https://doi.org/10.1007/s12649-023-02303-4
Duša, F., & Šlais, K. (2013). New solution IEF device for micropreparative separation of peptides and proteins. Electrophoresis, 34(11), 1519-1525.
https://doi.org/10.1002/elps.201200485
Hau, E. H., Chew, L. Y., Yeo, S. K., Owatworakit, A., Teh, S. S., & Mah, S. H. (2025). Oil palm leaf protein hydrolysate and its novel peptides as alternative plant-based α-glucosidase inhibitors. International Journal of Biological Macromolecules, 291, 138897.
 LinkPeptide
LinkPeptide