Targeting Collagen Quality: Cheminformatics-Designed Peptides Unlock a New Era in Anti-Aging Science
Abstract
Advances in cosmetic science are increasingly driven by a deeper understanding of skin biology and the development of targeted, mechanism-based active ingredients. This study highlights a novel cheminformatics-guided approach to discovering peptides that support collagen quality rather than simply boosting its synthesis. By screening a virtual library of 67 million peptides, researchers identified a lead dipeptide, ICP-1225, capable of enhancing both Hsp47—a key collagen-specific chaperone—and collagen gene expression in dermal fibroblasts. Further optimization produced ICP-1236, an acetylated derivative that demonstrated strong biological activity and effective topical performance. In ex vivo human skin, ICP-1236 restored Hsp47 levels and improved dermal collagen density following UVB damage. These findings underscore the potential of computational design and chaperone-targeted strategies to advance next-generation cosmetic peptides. The research illustrates a promising path toward more precise, biologically informed anti-aging solutions.
Introduction: Rethinking Anti-Aging Through Collagen Quality
Aging and photo-damage gradually erode the skin’s structural framework, with collagen loss as one of the most visible hallmarks. As fibroblasts slow down and UV exposure accumulates, collagen fibers become fragmented, disorganized, and increasingly difficult for the skin to rebuild. For decades, cosmetic science has focused on stimulating collagen synthesis using peptides, retinoids, and growth-factor–inspired actives. These approaches can certainly boost production, but they often overlook an equally important dimension of skin biology: the quality control systems that determine whether newly made collagen is actually usable.
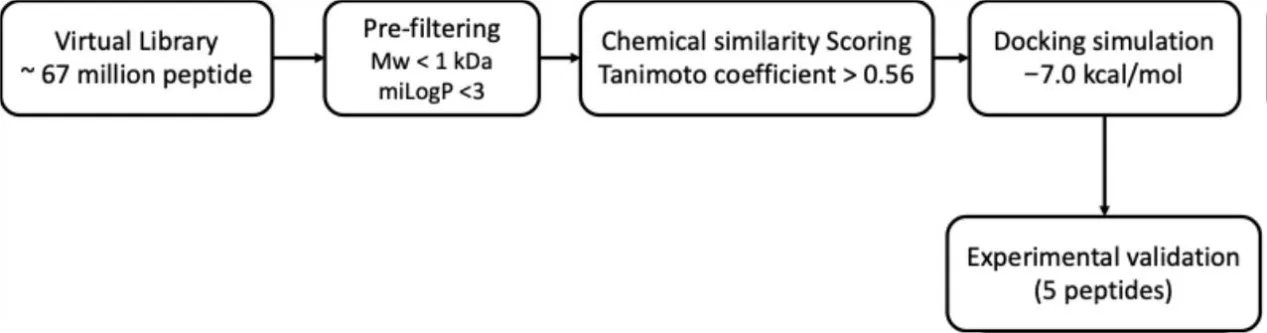
This gap has led researchers to revisit intracellular pathways that govern collagen homeostasis. Among them, Heat Shock Protein 47 (Hsp47) has emerged as a particularly compelling target. Hsp47 acts as a collagen-specific chaperone, ensuring that procollagen molecules fold correctly and are properly prepared for secretion into the extracellular matrix. Notably, Hsp47 levels decline with both chronological aging and UV exposure, suggesting that impaired collagen maturation—not just reduced synthesis—contributes to visible signs of skin aging.
Against this backdrop, the study discussed in this blog takes on special significance. By combining cheminformatics with peptide design, the researchers set out to discover novel cosmetic actives capable of supporting collagen quality from within the cell. Their work reflects a broader shift in the industry: using computational tools to develop more precise, mechanism-driven anti-aging strategies.
Hsp47: The Collagen Chaperone at the Heart of Skin Aging
Anti-aging research often concentrates on turning up collagen production, yet the journey from gene expression to robust, well-organized collagen fibers is far more complex. One of the most critical—and frequently overlooked—players in this journey is Heat Shock Protein 47 (Hsp47), a collagen-specific molecular chaperone located in the endoplasmic reticulum. Hsp47 binds to procollagen triple helices, stabilizing their structure and helping them fold correctly before they are transported out of the cell. Without this chaperoning step, newly synthesized collagen is more likely to be misfolded, degraded, or poorly assembled in the dermal matrix.
The study’s data help explain why Hsp47 merits serious attention from cosmetic researchers. Immunostaining of human skin samples from different age groups reveals a clear decline in Hsp47 expression, especially in individuals in their 50s and 60s. This pattern parallels the decline in collagen quality and integrity typically observed in aged skin. In addition, UVB irradiation—one of the most potent drivers of extrinsic aging—was shown to markedly suppress Hsp47 levels in ex vivo human skin models. Environmental stressors, in other words, may damage the skin’s collagen-folding machinery long before surface changes become visible.
For formulators and R&D teams, these findings broaden the anti-aging toolbox. Rather than focusing solely on collagen synthesis, targeting Hsp47 opens a new pathway: supporting the maturation of high-quality collagen fibers. This chaperone-centered approach offers a more comprehensive strategy for maintaining dermal resilience and aligns well with the industry’s move toward targeted, mechanism-based cosmetic actives.
Digital Peptide Discovery: A Cheminformatics Pipeline for Innovation
Discovering new cosmetic peptides has traditionally relied on trial-and-error synthesis, biological screening, and long development timelines. In contrast, the study introduces a modern, data-driven pipeline that applies cheminformatics to dramatically narrow down potential candidates before entering the lab. The researchers began by constructing a vast virtual peptide library of approximately 67 million short peptides, ranging from dipeptides to hexapeptides. To ensure that only cosmetically relevant structures were considered, they applied filters based on molecular weight, predicted lipophilicity, and charge—criteria essential for skin compatibility and topical formulation.
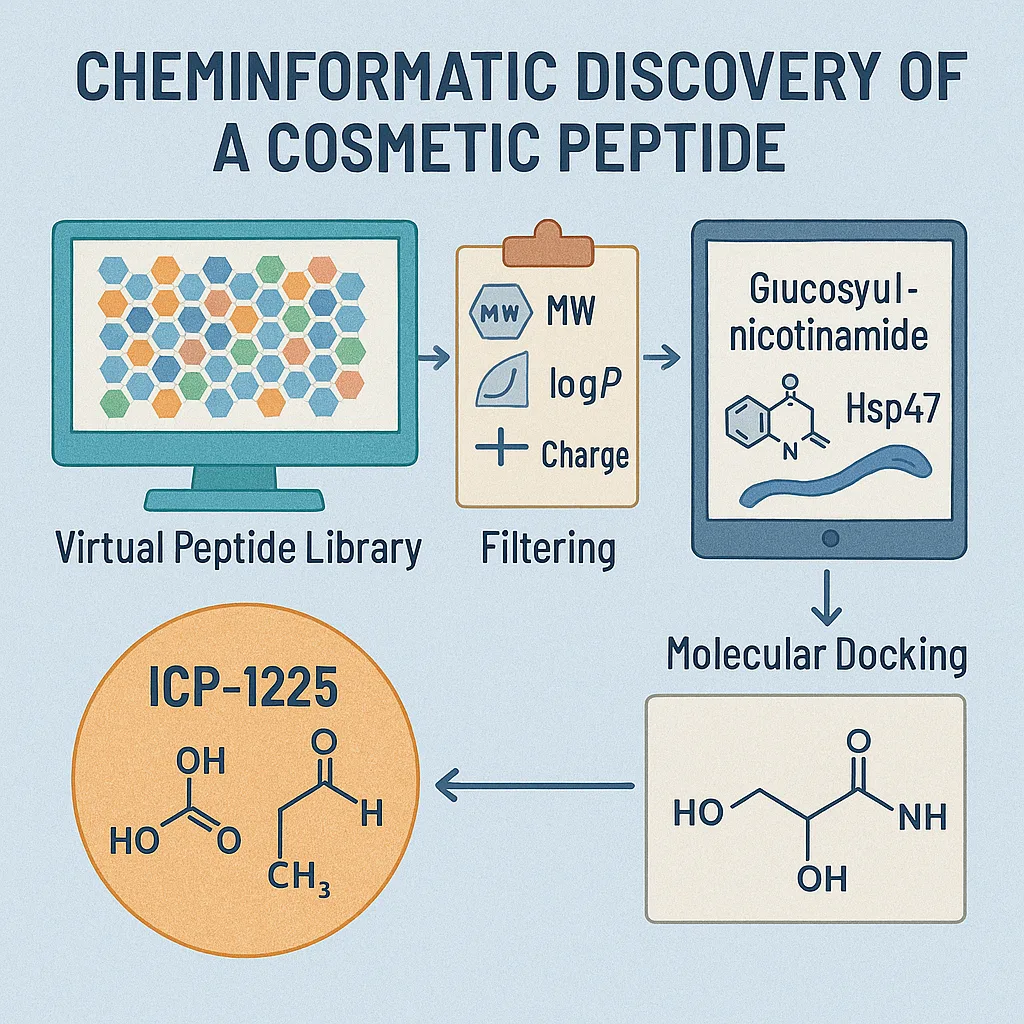
The next step involved similarity screening using glucosyl-nicotinamide as a reference structure, chosen for its known ability to modulate heat shock proteins. By calculating molecular fingerprints and Tanimoto similarity scores, the team identified a small subset of peptides with chemical features likely to influence Hsp47 activity. These candidates were then evaluated through molecular docking against the Hsp47 crystal structure to predict binding affinity and stability.
From this computational triage, five top-ranking peptides were synthesized and tested in human dermal fibroblasts. Among them, ICP-1225, a simple dipeptide composed of threonine and tyrosine, emerged as the standout performer. It displayed no cytotoxicity and significantly increased both Hsp47 expression and key collagen markers such as COL1A1 and COL3A1. Importantly, this dual effect—enhancing collagen synthesis while also supporting its proper folding—sets ICP-1225 apart from many existing cosmetic peptides.
This digital-to-biological workflow highlights how computational tools can streamline early discovery, reduce experimental workload, and guide peptide innovation toward more targeted, mechanism-driven solutions.
From Lab to Skin: Optimizing Peptides for Topical Performance
While identifying a bioactive peptide is an important milestone, transforming it into a viable cosmetic ingredient requires addressing challenges such as stability, skin penetration, and formulation compatibility. To enhance these properties, the researchers modified the lead dipeptide ICP-1225 through derivatization, generating three new variants: an acetylated form (ICP-1236), a hexanoylated form (ICP-1237), and a palmitoylated form (ICP-1238). Each modification was designed to tune the peptide’s lipophilicity and improve its behavior in topical applications.
Interestingly, the biological outcomes of these derivatives differed significantly. Among the three, ICP-1236—the acetylated version—demonstrated the clearest, dose-dependent activity, increasing both Hsp47 and collagen I expression in fibroblasts at concentrations of 10–100 µM. In contrast, the medium- and long-chain acyl derivatives showed limited or inconsistent performance, despite exhibiting no cytotoxicity. This finding underscores an important formulation insight: enhancing lipophilicity does not always translate to better biological outcomes, and subtle structural changes can dramatically shape a peptide’s efficacy.
The research further validated ICP-1236 using ex vivo NativeSkin® human skin models, a system highly valued in cosmetic R&D for bridging in vitro data with real-world topical performance. When UVB-damaged skin was treated with ICP-1236, the peptide successfully restored Hsp47 expression in the epidermis and increased collagen density in the dermis, as confirmed by immunofluorescence and Masson’s trichrome staining. These results provide compelling evidence that ICP-1236 can exert meaningful effects when applied topically—an essential benchmark for any cosmetic active seeking market relevance.
Future Directions: Toward Smarter, Mechanism-Driven Cosmetic Peptides
The findings from this study offer valuable insights for the next generation of peptide-based cosmetic actives. First, the successful identification of ICP-1225 and its optimized derivative ICP-1236 demonstrates how cheminformatics can drastically accelerate peptide discovery, reducing reliance on large-scale synthesis and traditional screening. As computational tools become more sophisticated and accessible, they are poised to reshape anti-aging ingredient development by enabling more targeted, mechanism-driven research.
Second, the focus on Hsp47 introduces a new dimension to collagen care. Rather than merely stimulating collagen production, supporting its correct folding and maturation may produce higher-quality fibers with improved structural integrity. For formulators, this offers a compelling way to differentiate products in a market crowded with “collagen boosters.” Hsp47-targeting peptides like ICP-1236 could complement existing actives by enhancing the efficiency and stability of collagen assembly, creating a more holistic approach to dermal rejuvenation.
The study also highlights important considerations for future R&D. Although the results are promising, the mechanism of action still requires deeper validation, including direct binding assays and long-term studies on fibroblast behavior. Clinical testing will also be necessary to determine real-world efficacy, stability in finished formulations, and consumer-relevant outcomes. Additionally, expanding the virtual peptide library to include D-amino acids or non-natural residues could open pathways to even more stable and potent actives.
Overall, this research reflects a broader industry movement: integrating computational chemistry, cellular biology, and formulation science to develop smarter, more effective cosmetic ingredients. As the field evolves, peptides like ICP-1236 may represent the future of precision anti-aging solutions.
Reference
Baek, S., Jeong, S., Yoon, S., Kim, Y., Kim, S., Chung, H. J., … & Nam, G. (2025). Development of New Anti-Wrinkle Peptide Using Cheminformatics-Assisted Peptidomimetic Design. Cosmetics, 12(6), 260.https://doi.org/10.3390/cosmetics12060260
Pintea, A., Manea, A., Pintea, C., Vlad, R. A., Bîrsan, M., Antonoaea, P., … & Ciurba, A. (2025). Peptides: Emerging candidates for the prevention and treatment of skin senescence: A review. Biomolecules, 15(1), 88.https://doi.org/10.3390/biom15010088
Xiao, B., Zhou, Y., Zhao, L., Huang, H., Fei, X., & Zhang, Y. B. (2025). A comprehensive dataset of therapeutic peptides on multi-function property and structure information. Scientific Data, 12(1), 1213.https://doi.org/10.1038/s41597-025-05528-1
Kim, Y., Yoon, S., Kim, S., Kim, Y., Jeong, S., & Kim, H. J. (2025). Clinical Efficacy of Adiponectin-Stimulating Peptide on UV-Induced Skin Damage. Cosmetics, 12(2), 54.https://doi.org/10.3390/cosmetics12020054
 LinkPeptide
LinkPeptide