GW501516 and the PPARδ Pathway: Exploring the Science Behind Cardarine
Abstract
Cardarine (GW501516) is a potent and selective PPARδ agonist originally developed to treat metabolic disorders such as dyslipidemia, type 2 diabetes, and obesity. Its ability to enhance fatty acid oxidation, improve insulin sensitivity, and increase endurance capacity quickly positioned it as a promising therapeutic agent and a target of interest among athletes. However, preclinical studies revealed a concerning carcinogenic potential, leading to the suspension of clinical trials and its inclusion on the WADA prohibited list. Despite these concerns, Cardarine remains a valuable research tool for understanding PPARδ signaling, mitochondrial function, and systemic metabolic regulation. This article reviews the compound’s pharmacological actions, therapeutic benefits, and oncogenic risks, offering insights into the complex interplay between efficacy and safety in nuclear receptor modulation. Future efforts will depend on developing more refined PPARδ-targeting agents that preserve metabolic advantages while minimizing oncogenic risk.
From Promise to Prohibition: The Rise and Fall of Cardarine
Cardarine, also known by its research designation GW501516, emerged in the 1990s as a promising PPARδ (Peroxisome Proliferator-Activated Receptor delta) agonist, developed collaboratively by GlaxoSmithKline and Ligand Pharmaceuticals. Initially intended for the treatment of metabolic disorders such as dyslipidemia, obesity, and type 2 diabetes mellitus, Cardarine quickly garnered attention for its potent effects on lipid metabolism, energy regulation, and endurance enhancement.
As a selective activator of PPARδ—a nuclear receptor involved in the transcriptional regulation of genes linked to fatty acid oxidation, mitochondrial biogenesis, and inflammation—GW501516 offered a novel approach to modulating systemic energy balance. Preclinical studies revealed compelling benefits, including enhanced fatty acid utilization, preservation of glucose stores, improved insulin sensitivity, and increased exercise capacity. These features positioned Cardarine as a potential therapeutic tool for metabolic syndrome and related cardiovascular pathologies.
However, the clinical trajectory of GW501516 was abruptly interrupted. Long-term toxicology studies in rodents indicated a significant increase in cancer incidence across multiple organs, raising serious safety concerns. Although these findings were species-specific and not definitively translatable to humans, they were sufficient to suspend further clinical development. Consequently, Cardarine was never approved for therapeutic use and was classified as a prohibited substance by the World Anti-Doping Agency (WADA) due to its performance-enhancing potential.
Despite these setbacks, Cardarine continues to attract interest—both in the athletic underground, where it is often sold as a “research chemical,” and within academic research circles, where it serves as a molecular tool to explore PPARδ signaling. As such, GW501516 remains a compound of dual identity: a molecular switch with intriguing therapeutic promise, and a cautionary tale in drug development, reminding researchers of the delicate interplay between efficacy and safety in translational science.
Inside the Pathways: How GW501516 Reprograms Cellular Metabolism
At the core of Cardarine’s pharmacological action lies its ability to selectively activate PPARδ, a member of the nuclear hormone receptor superfamily that orchestrates key metabolic pathways. Unlike PPARα and PPARγ, which are primarily associated with hepatic lipid metabolism and adipocyte differentiation respectively, PPARδ is broadly expressed in metabolically active tissues—notably skeletal muscle, cardiac muscle, liver, adipose tissue, and the central nervous system.
Upon ligand binding, activated PPARδ forms a heterodimer with the retinoid X receptor (RXR) and binds to peroxisome proliferator response elements (PPREs) in the promoter regions of target genes. This interaction initiates a transcriptional cascade that upregulates genes involved in fatty acid transport, β-oxidation, mitochondrial respiration, and anti-inflammatory signaling.
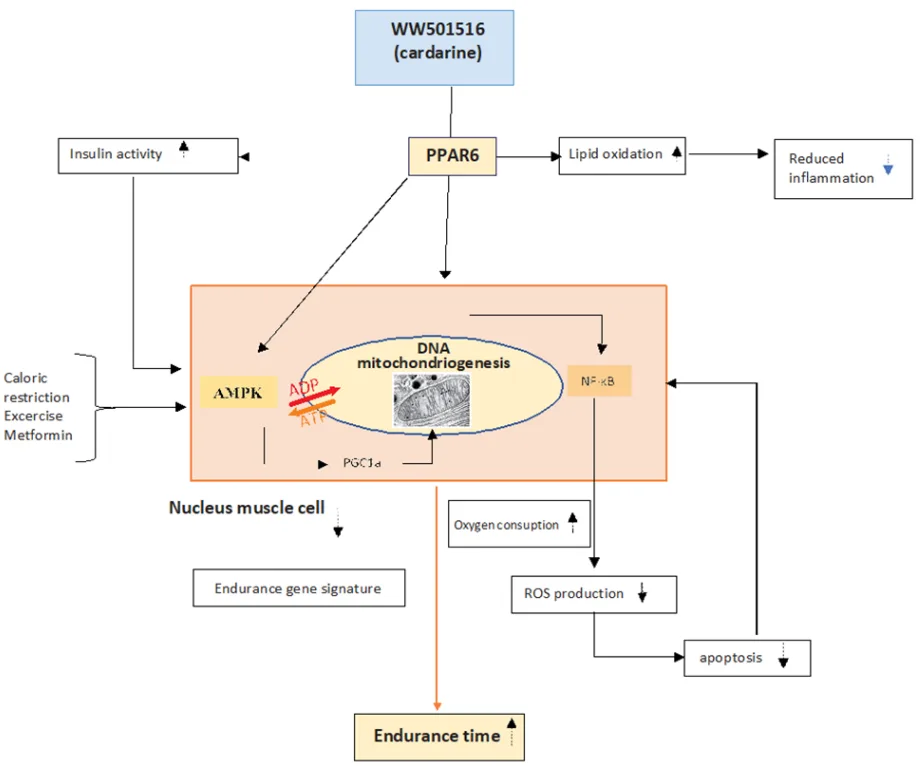
In skeletal muscle, PPARδ activation leads to a pronounced shift toward oxidative fiber type specification, favoring type I slow-twitch fibers that are rich in mitochondria and optimized for endurance. This is achieved through the upregulation of PGC-1α, a master regulator of mitochondrial biogenesis. Moreover, Cardarine stimulates AMP-activated protein kinase (AMPK), an energy sensor that enhances glucose uptake, inhibits lipogenesis, and synergizes with PPARδ to amplify fatty acid oxidation. Notably, AMPK activation also contributes to improved insulin sensitivity and mimics key aspects of caloric restriction and physical exercise at the molecular level.
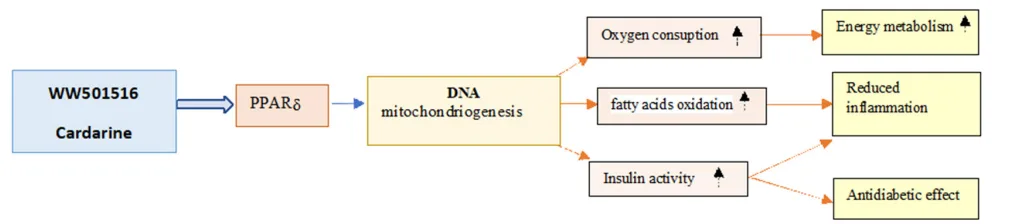
Cardarine’s influence extends to the pancreatic β-cells, where it supports insulin secretion and viability through modulation of lipid signaling pathways and oxidative stress responses. In adipose tissue and the liver, PPARδ activation downregulates pro-inflammatory cytokines (e.g., TNF-α, IL-6), enhances lipid clearance, and mitigates insulin resistance. This anti-inflammatory profile is particularly relevant to the pathophysiology of non-alcoholic fatty liver disease (NAFLD) and type 2 diabetes mellitus (T2DM).
Interestingly, Cardarine also stimulates glucagon-like peptide-1 (GLP-1) secretion from intestinal L-cells, offering an additional mechanism for improving glycemic control. By acting at multiple metabolic checkpoints, GW501516 functions as a systemic modulator of energy homeostasis, effectively rewiring cellular metabolism in a tissue-specific and pathway-selective manner.
These multifaceted effects explain why Cardarine has attracted such intense scientific interest—not merely as a performance enhancer, but as a powerful research probe into PPARδ biology and metabolic regulation.
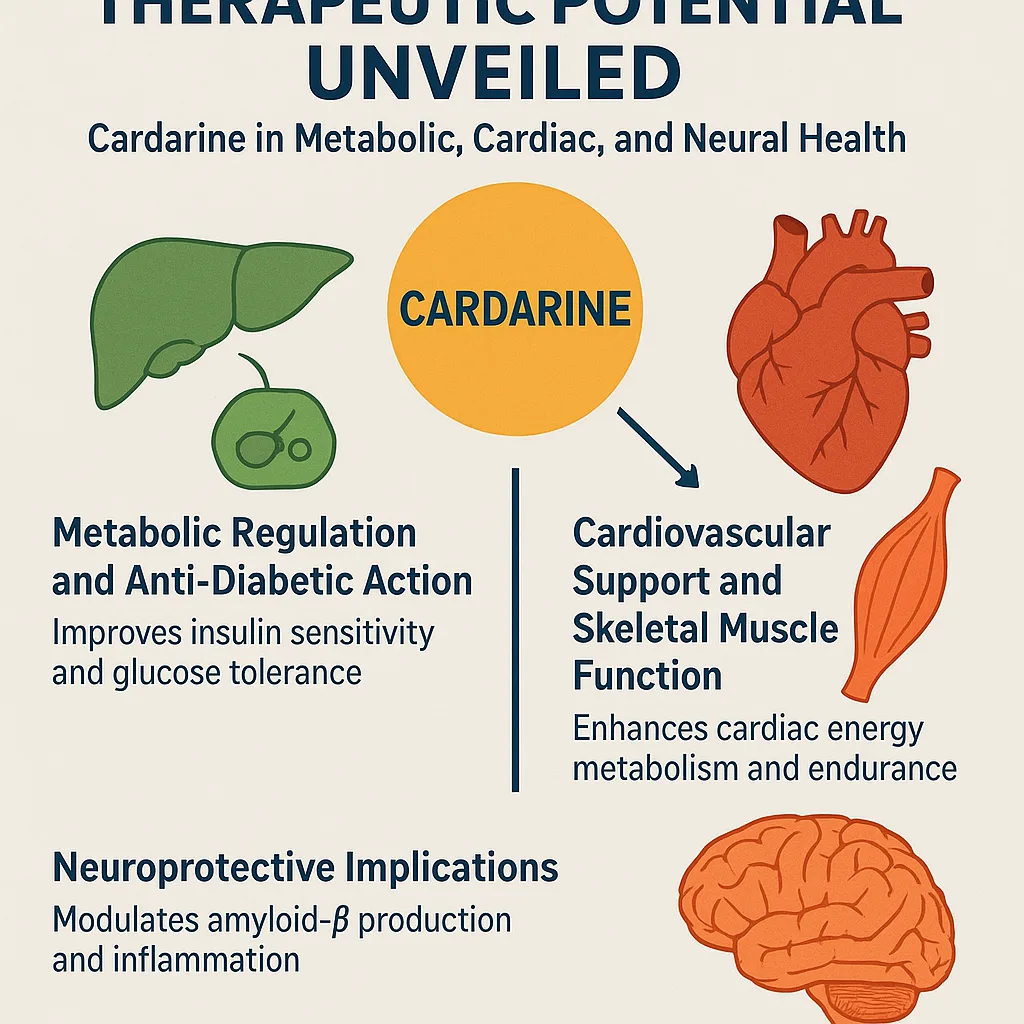
Therapeutic Potential Unveiled: Cardarine in Metabolic, Cardiac, and Neural Health
The broad metabolic activity of Cardarine translates into a diverse therapeutic profile, with experimental evidence supporting its potential utility in a range of chronic metabolic and degenerative disorders. Although clinical translation remains elusive due to safety concerns, preclinical studies have demonstrated robust benefits across multiple organ systems.
Metabolic Regulation and Anti-Diabetic Action
One of Cardarine’s most thoroughly investigated effects is its capacity to improve insulin sensitivity and glucose tolerance, both in genetic and diet-induced models of type 2 diabetes mellitus (T2DM). Through activation of PPARδ and downstream effectors such as AMPK and PGC-1α, Cardarine enhances fatty acid oxidation, reduces lipotoxicity, and preserves β-cell integrity. In rat models, it has been shown to modulate key survival pathways (e.g., Akt/Bcl-2) and increase the expression of GPR40, a G-protein-coupled receptor involved in insulinotropic signaling.
Additionally, Cardarine has been shown to stimulate GLP-1 secretion from enteroendocrine cells, contributing to improved postprandial insulin responses. This dual mechanism—direct action on pancreatic β-cells and indirect incretin stimulation—underscores its relevance for multifactorial metabolic diseases.
Cardiovascular Support and Skeletal Muscle Function
In the cardiovascular system, PPARδ activation by GW501516 facilitates enhanced cardiac energy metabolism, particularly under stress conditions such as ischemia or diabetic cardiomyopathy. By stimulating fatty acid utilization and suppressing inflammatory mediators like NF-κB and IL-6, Cardarine protects cardiomyocytes from oxidative and lipotoxic injury.
It also exerts anti-fibrotic effects: studies in cardiac fibroblasts show a reduction in collagen synthesis and inhibition of angiotensin II-induced matrix remodeling, highlighting its potential to attenuate myocardial fibrosis. Notably, in animal models with left ventricular dysfunction, Cardarine significantly improved physical endurance and skeletal muscle oxidative capacity, mimicking the benefits of exercise in pathophysiological states characterized by reduced functional reserve.
Neuroprotective Implications
Emerging data suggest a neuroprotective role for PPARδ agonists, including Cardarine, in various models of neurodegenerative disease. GW501516 has been shown to suppress BACE1 expression, a key enzyme in the production of neurotoxic amyloid-β, through modulation of SOCS1-JAK/STAT signaling. This effect positions Cardarine as a candidate for Alzheimer’s disease (AD) intervention, where amyloid deposition and neuroinflammation drive disease progression.
Beyond AD, PPARδ activation has demonstrated protective effects in experimental models of Parkinson’s disease, amyotrophic lateral sclerosis (ALS), and multiple sclerosis (MS). These effects are believed to stem from its ability to mitigate oxidative stress, inhibit neuroinflammatory pathways, and promote neuronal survival.
A Molecular Double-Edged Sword: Cancer Risk and Clinical Collapse
Despite its wide-ranging therapeutic potential, Cardarine’s clinical trajectory was ultimately derailed by oncogenic safety concerns. Long-term toxicology studies in rodents revealed that GW501516 significantly accelerated tumor formation across multiple tissues, including the liver, bladder, stomach, and mammary glands. These findings were severe enough to halt its clinical development and reinforce its designation as a banned substance by WADA.
The cancer controversy surrounding Cardarine stems from the complex and sometimes contradictory roles of PPARδ in cell proliferation and differentiation. While certain in vitro studies have shown anti-proliferative and pro-apoptotic effects in cancers such as bladder carcinoma and nasopharyngeal carcinoma, others report enhanced angiogenesis and tumor progression—notably in colorectal and breast cancer models. These inconsistencies suggest that PPARδ may act as a context-dependent regulator, with tumorigenic or tumor-suppressive properties depending on tissue type, co-regulators, and microenvironmental signals.
Additionally, much of the carcinogenic evidence originates from rodent studies, raising questions about their translatability to humans. Human-specific receptor dynamics, metabolic rates, and tumor biology complicate direct extrapolation. Nevertheless, in the absence of long-term human safety data, regulatory bodies have taken a precautionary stance.
Cardarine’s story is emblematic of the challenges in developing nuclear receptor modulators: potent and multi-targeted by nature, but difficult to constrain within safe therapeutic windows. Until these mechanistic ambiguities are resolved, the risk-benefit profile of GW501516 will remain a subject of both caution and curiosity within the biomedical community.
Beyond Cardarine: Lessons for the Future of Metabolic Modulators
Cardarine (GW501516) exemplifies the paradox often faced in translational medicine: a compound with exceptional therapeutic breadth—from improving metabolic efficiency and enhancing physical endurance to offering neuro- and cardioprotection—yet barred from clinical use due to unresolved safety liabilities. Its capacity to reprogram energy metabolism via PPARδ activation has transformed it into both a tool for scientific discovery and a symbol of regulatory caution.
The challenge ahead lies not in discarding PPARδ agonism as a therapeutic strategy, but in refining its precision and safety. Selective modulators that disentangle beneficial metabolic effects from pro-oncogenic pathways represent an active area of pharmaceutical interest. The ongoing development of newer agents, including dual and pan-PPAR agonists, may eventually succeed where GW501516 fell short—offering metabolic modulation without carcinogenic compromise.
Moreover, as the field of exercise mimetics evolves, the mechanistic insights gleaned from Cardarine research continue to inform our understanding of endurance biology, AMPK signaling, and mitochondrial dynamics. Even in the absence of approved use, Cardarine remains a cornerstone molecule in the study of nuclear receptor pharmacology.
In conclusion, while GW501516 may never reach the clinic in its original form, its legacy will likely endure as a stepping stone toward next-generation metabolic therapies—provided the scientific community maintains both ambition and rigor in equal measure.
Reference
Oliver Jr, W. R., Shenk, J. L., Snaith, M. R., Russell, C. S., Plunket, K. D., Bodkin, N. L., … & Willson, T. M. (2001). A selective peroxisome proliferator-activated receptor δ agonist promotes reverse cholesterol transport. Proceedings of the national academy of sciences, 98(9), 5306-5311.
https://doi.org/10.1073/pnas.091021198
Forman, B. M., Chen, J., & Evans, R. M. (1997). Hypolipidemic drugs, polyunsaturated fatty acids, and eicosanoids are ligands for peroxisome proliferator-activated receptors α and δ. Proceedings of the National Academy of Sciences, 94(9), 4312-4317.
https://doi.org/10.1073/pnas.94.9.4312
Bianchi, V. E., & Locatelli, V. (2023). GW501516 (Cardarine): Pharmacological and Clinical Effects. Genet Mol Med, 5(1), 1-12.
 LinkPeptide
LinkPeptide