Labeled Peptides: Choosing the Right Label for Peptide Studies
Introduction: Why Peptide Labeling Matters in Modern Research
Peptides are widely used in biomedical research, drug discovery, and diagnostic development due to their high specificity and versatility. However, studying peptide behavior—such as binding, localization, and biological activity—often requires visualization or tracking, which is not possible with unlabeled molecules.
This is where labeled peptides become essential.
By attaching functional labels such as fluorophores, isotopes, or affinity tags, researchers can monitor peptide interactions in real time, quantify biological processes, and improve experimental accuracy. Choosing the right label is therefore a critical decision that directly impacts data quality and experimental outcomes.
What Are Labeled Peptides?
Labeled peptides are peptides that have been chemically modified with a detectable or functional group. These labels enable researchers to:
- Track peptide localization in cells or tissues
- Measure binding interactions
- Quantify biological activity
- Visualize molecular processes in real time
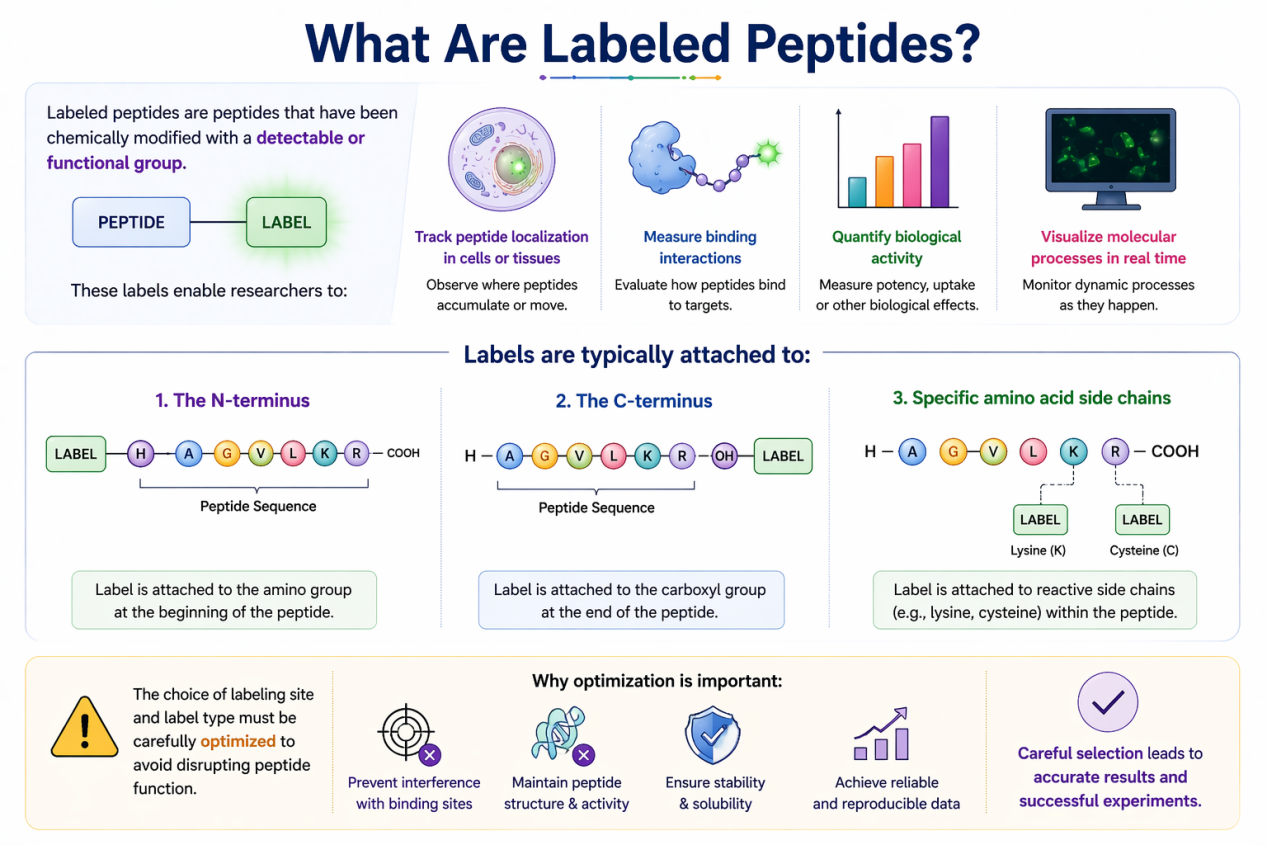
Labels are typically attached to:
- The N-terminus
- The C-terminus
- Specific amino acid side chains (e.g., lysine, cysteine)
The choice of labeling site and label type must be carefully optimized to avoid disrupting peptide function.
Major Types of Peptide Labels
Selecting the appropriate label depends on your experimental goal. Below are the most commonly used categories.
Fluorescent Labels: For Imaging and Visualization
Fluorescent labeling is one of the most widely used approaches in peptide research.
Common Fluorophores:
- FITC (Fluorescein isothiocyanate)
- Rhodamine
- Cy3 / Cy5
- Alexa Fluor dyes
Applications:
- Cell imaging
- Receptor binding studies
- Intracellular tracking
Advantages:
- Real-time visualization
- High sensitivity
- Compatible with microscopy and flow cytometry
Considerations:
- Fluorophore size may affect peptide activity
- Photobleaching can limit long-term studies
- Signal overlap in multiplex experiments
- Best for: Cellular imaging and localization studies
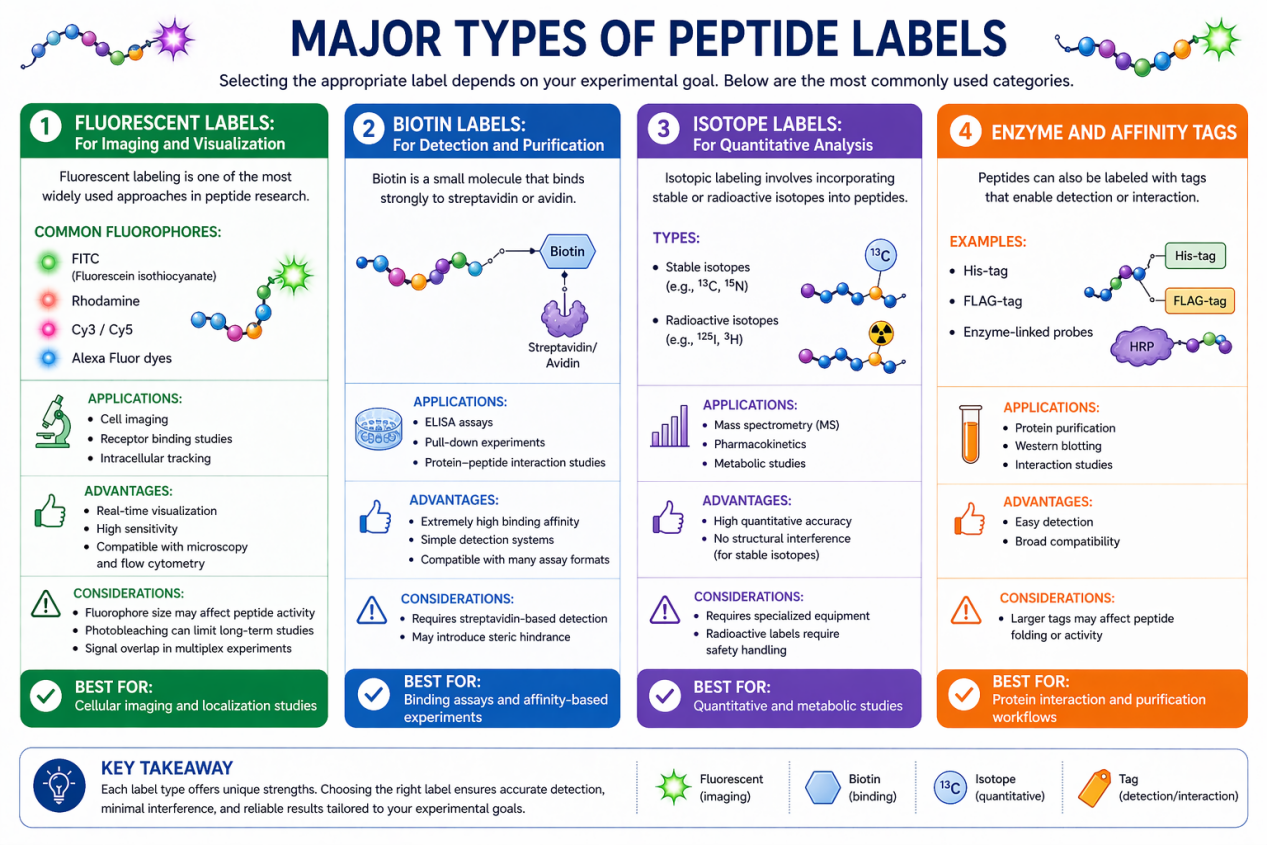
Biotin Labels: For Detection and Purification
Biotin is a small molecule that binds strongly to streptavidin or avidin.
Applications:
- ELISA assays
- Pull-down experiments
- Protein–peptide interaction studies
Advantages:
- Extremely high binding affinity
- Simple detection systems
- Compatible with many assay formats
Considerations:
- Requires streptavidin-based detection
- May introduce steric hindrance
- Best for: Binding assays and affinity-based experiments
Isotope Labels: For Quantitative Analysis
Isotopic labeling involves incorporating stable or radioactive isotopes into peptides.
Types:
- Stable isotopes (e.g., 13C, 15N)
- Radioactive isotopes (e.g., 125I, 3H)
Applications:
- Mass spectrometry (MS)
- Pharmacokinetics
- Metabolic studies
Advantages:
- High quantitative accuracy
- No structural interference (for stable isotopes)
Considerations:
- Requires specialized equipment
- Radioactive labels require safety handling
- Best for: Quantitative and metabolic studies
Enzyme and Affinity Tags
Peptides can also be labeled with tags that enable detection or interaction.
Examples:
- His-tag
- FLAG-tag
- Enzyme-linked probes
Applications:
- Protein purification
- Western blotting
- Interaction studies
Advantages:
- Easy detection
- Broad compatibility
- Considerations:
- Larger tags may affect peptide folding or activity
- Best for: Protein interaction and purification workflows
Key Factors When Choosing a Peptide Label
Selecting the right label requires balancing multiple experimental considerations.
1. Experimental Objective
Start by defining your goal:
- Imaging → fluorescent label
- Quantification → isotope label
- Binding assay → biotin
- The label must match the biological question
2. Peptide Function and Structure
Labels can interfere with:
- Binding sites
- Conformation
- Biological activity
Best practice:
- Avoid labeling near active or binding regions
- Use linkers (e.g., PEG spacers) when necessary
3. Label Size and Steric Effects
Larger labels may:
- Reduce binding affinity
- Alter peptide folding
Smaller labels are generally preferred when:
- Structural integrity is critical
4. Detection Sensitivity
Consider:
- Required signal strength
- Background noise
Fluorescent dyes vary in:
- Brightness
- Stability
- Wavelength range
5. Stability and Compatibility
Ensure the label is stable under:
- Experimental conditions (pH, temperature)
- Biological environments (enzymes, serum)
6. Multiplexing Capability
If using multiple labels:
- Avoid spectral overlap
- Choose compatible detection systems
Labeling Strategies: Where and How to Attach Label
The position of labeling significantly affects peptide performance.
N-Terminal Labeling
- Most common approach
- Minimal interference in many cases
C-Terminal Labeling
- Useful when N-terminus is functionally important
Side-Chain Labeling
- Targets residues like Lys or Cys
- Enables site-specific modification
Use of Linkers
Linkers such as PEG can:
- Reduce steric hindrance
- Improve solubility
- Preserve biological activity
Common Challenges in Peptide Labeling
Despite its advantages, peptide labeling presents several challenges:
Signal Interference
Labels may affect:
- Binding affinity
- Biological function
Photobleaching (Fluorescent Labels)
- Signal loss over time
- Requires careful experimental design
Stability Issues
Some labels degrade under:
- Light exposure
- Enzymatic conditions
Cost and Complexity
- Custom labeling increases synthesis complexity
- Some labels require specialized handling
Practical Tips for Researchers
To achieve reliable results:
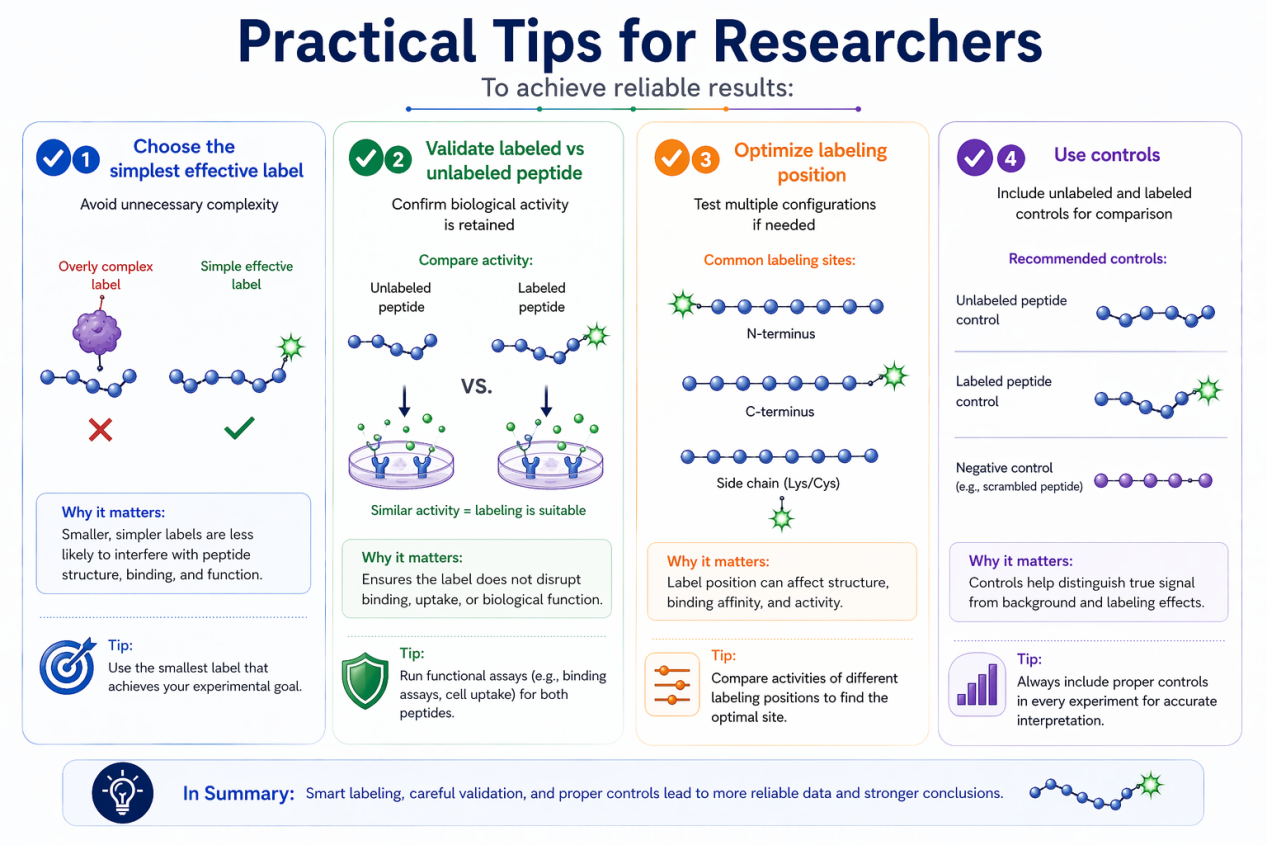
✔ Choose the simplest effective label
Avoid unnecessary complexity
✔ Validate labeled vs unlabeled peptide
Confirm biological activity is retained
✔ Optimize labeling position
Test multiple configurations if needed
✔ Use controls
Include unlabeled and labeled controls for comparison
Applications of Labeled Peptides in Research
Labeled peptides are used across multiple disciplines:
- Drug discovery → receptor binding studies
- Cancer research → tumor imaging
- Neuroscience → signaling pathway tracking
- Immunology → antigen–antibody interactions
- Cosmetics research → skin penetration and activity studies
How LinkPeptide Supports Labeled Peptide Development
At LinkPeptide, we provide comprehensive solutions for labeled peptide synthesis, including:
- Custom fluorescent peptide labeling
- Biotinylated peptides
- Isotope-labeled peptides
- Site-specific modification strategies
- High-purity synthesis with analytical validation (HPLC, MS)
We help researchers design functionally optimized labeled peptides tailored to their experimental needs.
Conclusion
Labeled peptides are indispensable tools for modern biological research, enabling precise tracking, visualization, and quantification of molecular processes.
However, selecting the right label is not a trivial step—it requires careful consideration of:
- Experimental goals
- Peptide structure
- Detection methods
- Biological context
By making informed choices and optimizing labeling strategies, researchers can unlock the full potential of peptide-based studies and generate more reliable, meaningful data.
Reference
Richter, S., & Wuest, F. (2014). 18F-labeled peptides: the future is bright. Molecules, 19(12), 20536-20556.
https://doi.org/10.3390/molecules191220536
Zhang, R., Sioma, C. S., Wang, S., & Regnier, F. E. (2001). Fractionation of isotopically labeled peptides in quantitative proteomics. Analytical chemistry, 73(21), 5142-5149.
https://doi.org/10.1021/ac010583a
Kuil, J., Velders, A. H., & van Leeuwen, F. W. (2010). Multimodal tumor-targeting peptides functionalized with both a radio-and a fluorescent label. Bioconjugate chemistry, 21(10), 1709-1719.
https://doi.org/10.1021/bc100276j
Gonçalves, M. S. T. (2009). Fluorescent labeling of biomolecules with organic probes. Chemical reviews, 109(1), 190-212.
 LinkPeptide
LinkPeptide