NPRA, NPRB, and NPRC: How Natriuretic Peptide Receptors Are Shaping the Future of Cancer Treatment
Abstract
Natriuretic peptides, long known for their role in cardiovascular regulation, are gaining attention as key players in cancer biology. Their receptors—NPRA, NPRB, and NPRC—show strikingly different effects on tumor development, progression, and treatment response. NPRA emerges as a strong oncogenic driver, promoting angiogenesis, metastasis, and resistance to chemotherapy. NPRB plays a dual role, but its activation by engineered ligands such as dCNP can normalize tumor vasculature, enhance immune infiltration, and improve outcomes when combined with existing therapies. In contrast, NPRC functions primarily as a tumor suppressor, linked to improved prognosis and potential use in molecular imaging and targeted therapy. Together, these findings position the natriuretic peptide receptor family as both promising biomarkers and therapeutic targets. By understanding their context-dependent roles, researchers are uncovering new opportunities for precision oncology and next-generation cancer treatment strategies.
Introduction: From Heart Hormones to Cancer Signals
When most people think of natriuretic peptides (NPs), the heart immediately comes to mind. These small signaling molecules—atrial natriuretic peptide (ANP), brain natriuretic peptide (BNP), and C-type natriuretic peptide (CNP)—were first discovered for their crucial roles in controlling blood pressure, fluid balance, and overall cardiovascular stability. For decades, they have been valuable tools in diagnosing and monitoring heart failure and related diseases.
Yet, science has revealed a far more complex story. Beyond the cardiovascular system, NPs and their receptors are emerging as important players in cancer biology. The three primary receptors—NPRA, NPRB, and NPRC—collectively known as the natriuretic peptide receptor family, were once regarded mainly as gatekeepers of vascular function. Today, researchers recognize them as key regulators of tumor growth, invasion, metastasis, and even treatment resistance.
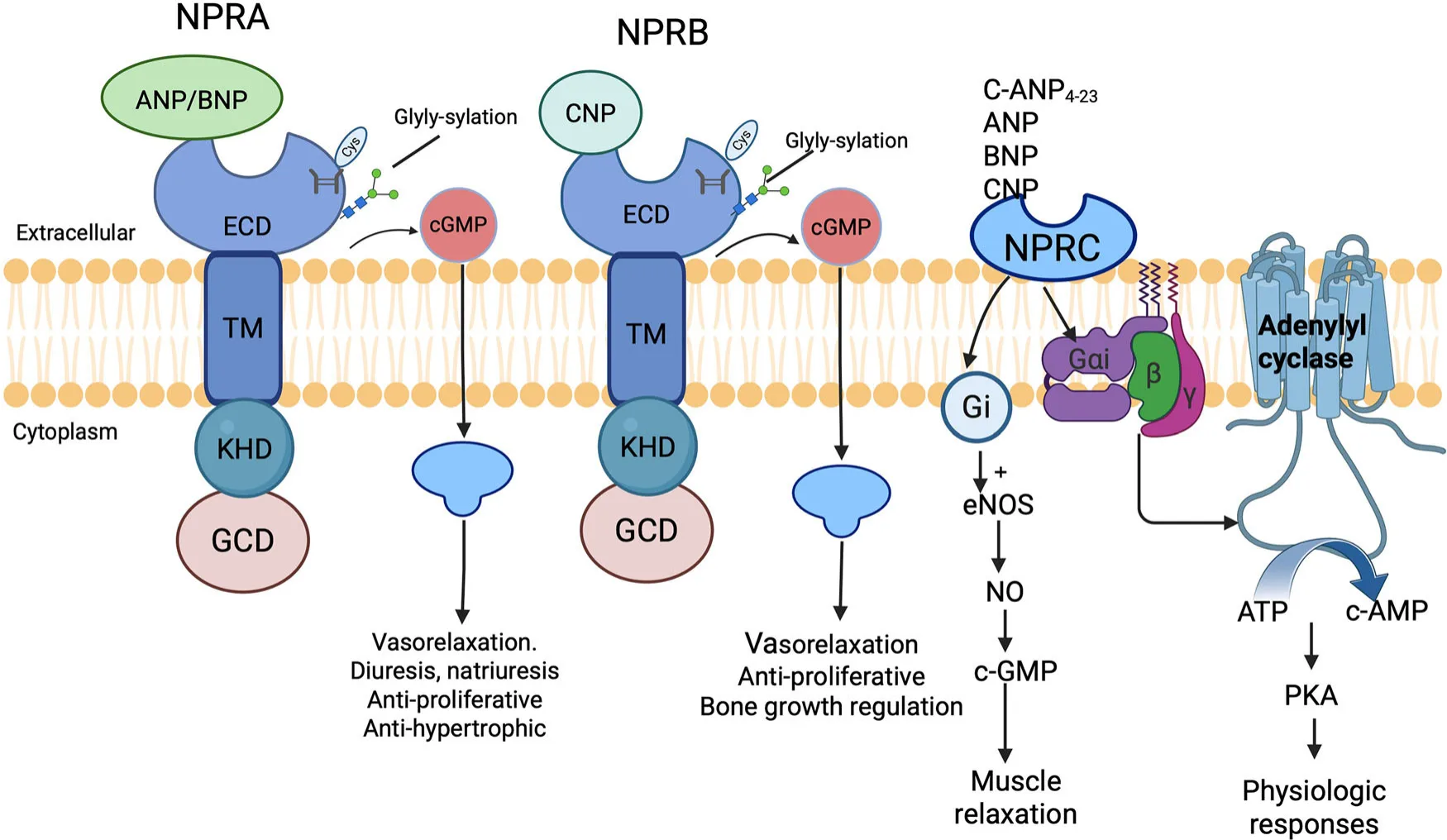
What makes these receptors especially intriguing is their context-dependent duality. In some cancers, they act as drivers of malignancy, promoting angiogenesis, metastasis, and therapy resistance. In others, they function as tumor suppressors, triggering apoptosis and slowing disease progression. This paradox has drawn increasing attention to their potential as both biomarkers for prognosis and targets for innovative cancer therapies.
In this blog, we will explore the distinct roles of NPRA, NPRB, and NPRC in cancer development, and highlight why they may represent the next frontier in precision oncology.
NPRA – Fueling Tumor Growth and Resistance
Among the three natriuretic peptide receptors, NPRA (natriuretic peptide receptor A) stands out as the most firmly linked to cancer progression. In multiple studies, NPRA is consistently found to be overexpressed in a range of tumors—including breast, gastric, prostate, and squamous cell carcinomas—where its presence correlates strongly with aggressive disease and poor patient outcomes.
Table 1 Characteristics and functions of natriuretic peptide receptor family members.
| Receptor type | Gene location | Primary ligands | Signaling
pathway |
Physiological function | Primary role in tumors |
|---|---|---|---|---|---|
| NPRA (NPR1/GC-A) | 1q21.3 | ANP > BNP > CNP | cGMP/PKG | Cardiovascular homeostasis, BP regulation | Pro-tumor: Promotes angiogenesis, inhibits apoptosis |
| NPRB (NPR2/GC-B) | 9p13.3 | CNP > ANP ≥ BNP | cGMP/PKG | Cartilage development, bone formation, heart rate regulation | Dual-role: Promotes vascular normalization, inhibits metastasis |
| NPRC (NPR3) | 5p13.3 | ANP > CNP > BNP | Clearance/Gi-protein | Peptide clearance, vascular tone regulation | Antitumor: Promotes apoptosis, inhibits proliferation |
Mechanistically, NPRA fuels tumor growth by orchestrating several hallmark cancer processes. One of its key actions is the promotion of angiogenesis, the formation of new blood vessels that supply tumors with oxygen and nutrients. By stabilizing hypoxia-inducible factor 1-alpha (HIF-1α), NPRA increases the production of vascular endothelial growth factor (VEGF), a powerful signal that drives the sprouting of blood vessels into tumors. This vascular support not only accelerates growth but also provides channels for metastasis.
NPRA also contributes to tumor invasion and spread through the activation of STAT3, a transcription factor that upregulates matrix metalloproteinase-9 (MMP9). MMP9 breaks down the extracellular matrix, effectively clearing a path for cancer cells to migrate and establish secondary tumors. In breast cancer, this signaling axis has been directly linked to reduced survival, making NPRA a potential prognostic marker.
Even more concerning is NPRA’s role in chemoresistance and cancer stemness. In gastric cancer, NPRA engages in a feedback loop with mesenchymal stem cells (MSCs), enhancing fatty acid oxidation and maintaining cancer stem cell traits. This loop not only sustains tumor growth but also shields cancer cells from chemotherapy-induced apoptosis, leading to treatment failure and relapse.
Preclinical research suggests that silencing NPRA or blocking its signaling can halt tumor growth, impair metastasis, and restore chemosensitivity. One particularly interesting study showed that combining the diabetes drug glipizide with ANP (NPRA’s natural ligand) effectively suppressed breast cancer progression in mice by reducing tumor-induced angiogenesis. This highlights the exciting possibility of drug repurposing and combination therapies targeting NPRA.
Taken together, NPRA emerges as a critical oncogenic driver—a receptor that not only accelerates cancer progression but also undermines treatment. This makes it a highly attractive target for the next generation of anticancer therapies.
NPRB – Remodeling the Tumor Environment
If NPRA is largely a tumor promoter, NPRB (natriuretic peptide receptor B) plays a more complicated, context-dependent role. NPRB is the primary receptor for C-type natriuretic peptide (CNP), which is mainly produced by vascular endothelial cells. While NPRB shares structural similarities with NPRA, its effects in cancer are far less straightforward—sometimes protective, sometimes harmful.
One of the most promising findings is NPRB’s role in vascular normalization. Unlike the chaotic, leaky blood vessels typically seen in tumors, NPRB activation by engineered CNP derivatives—most notably dCNP—has been shown to stabilize tumor vasculature. This leads to tighter endothelial junctions, better pericyte coverage, and reduced vessel leakage. As a result, oxygen delivery improves, hypoxia decreases, and chemotherapy drugs penetrate more effectively. In preclinical models, dCNP treatment doubled intratumoral drug concentration and reduced tumor hypoxia by nearly 40%.
Beyond vascular effects, NPRB also shows potential in immunomodulation. By normalizing vessels, dCNP enhances immune cell trafficking into tumors, boosting the infiltration of cytotoxic T cells and natural killer (NK) cells. In mouse models of renal cell carcinoma, combining dCNP with anti-PD-1 immunotherapy increased response rates from 20% to 70%. dCNP has also been shown to augment radiotherapy by sensitizing hypoxic tumors and even enhance CAR-T cell efficacy by improving infiltration into solid tumors.
Yet NPRB’s story is not entirely positive. In some cancers, its expression is reduced as disease advances, and its protective role becomes blunted. This duality makes NPRB a double-edged sword—not universally oncogenic or suppressive, but highly dependent on cancer type and tumor microenvironment.
The take-home message is that NPRB, especially through dCNP-based therapies, represents a powerful adjunct strategy: not a direct killer of cancer cells, but a remodeler of the tumor environment that makes other treatments far more effective.
NPRC – The Protective Receptor With Diagnostic Power
In contrast to NPRA’s tumor-promoting role, NPRC (natriuretic peptide receptor C) often acts as a brake on cancer progression. Unlike NPRA and NPRB, which are guanylyl cyclase receptors, NPRC primarily functions as a “clearance receptor,” removing excess natriuretic peptides from circulation. But in the context of cancer, NPRC takes on a broader role as a tumor suppressor.
Studies have shown that overexpression of NPRC can slow tumor cell proliferation, induce apoptosis, and block pathways that normally drive malignancy. In osteosarcoma, for example, NPRC activation was found to halt cell cycle progression at the G1 phase and promote programmed cell death. Mechanistically, this effect was linked to the downregulation of the PI3K/AKT pathway, one of the most well-known survival and growth signaling cascades in cancer.
Clinical data further highlight NPRC’s protective role. In clear cell renal cell carcinoma (ccRCC), patients with higher NPRC expression had significantly better survival outcomes. Similarly, NPRC has been identified as part of favorable molecular signatures that predict less aggressive tumor subtypes. This positions NPRC not just as a tumor suppressor but also as a valuable prognostic biomarker.
NPRC’s utility extends into diagnostics as well. Researchers have developed molecular imaging probes that specifically target NPRC, allowing for highly sensitive PET imaging of prostate cancer. Such innovations could make NPRC an important player in precision oncology imaging.
Importantly, NPRC agonists such as C-ANP4-23 have already shown promising antitumor effects in preclinical studies, suggesting a therapeutic future where NPRC activation could be harnessed to slow tumor progression. While exceptions exist—such as certain colorectal cancers where NPRC upregulation may support growth—the overall trend points to NPRC as the protective member of the NPR family, with clear clinical relevance.
Looking Ahead: NPRs as Biomarkers and Therapies
The contrasting roles of NPRA, NPRB, and NPRC highlight both the complexity and opportunity within the natriuretic peptide receptor family. What began as a set of heart-related signaling pathways has now expanded into a promising field of cancer research and therapy.
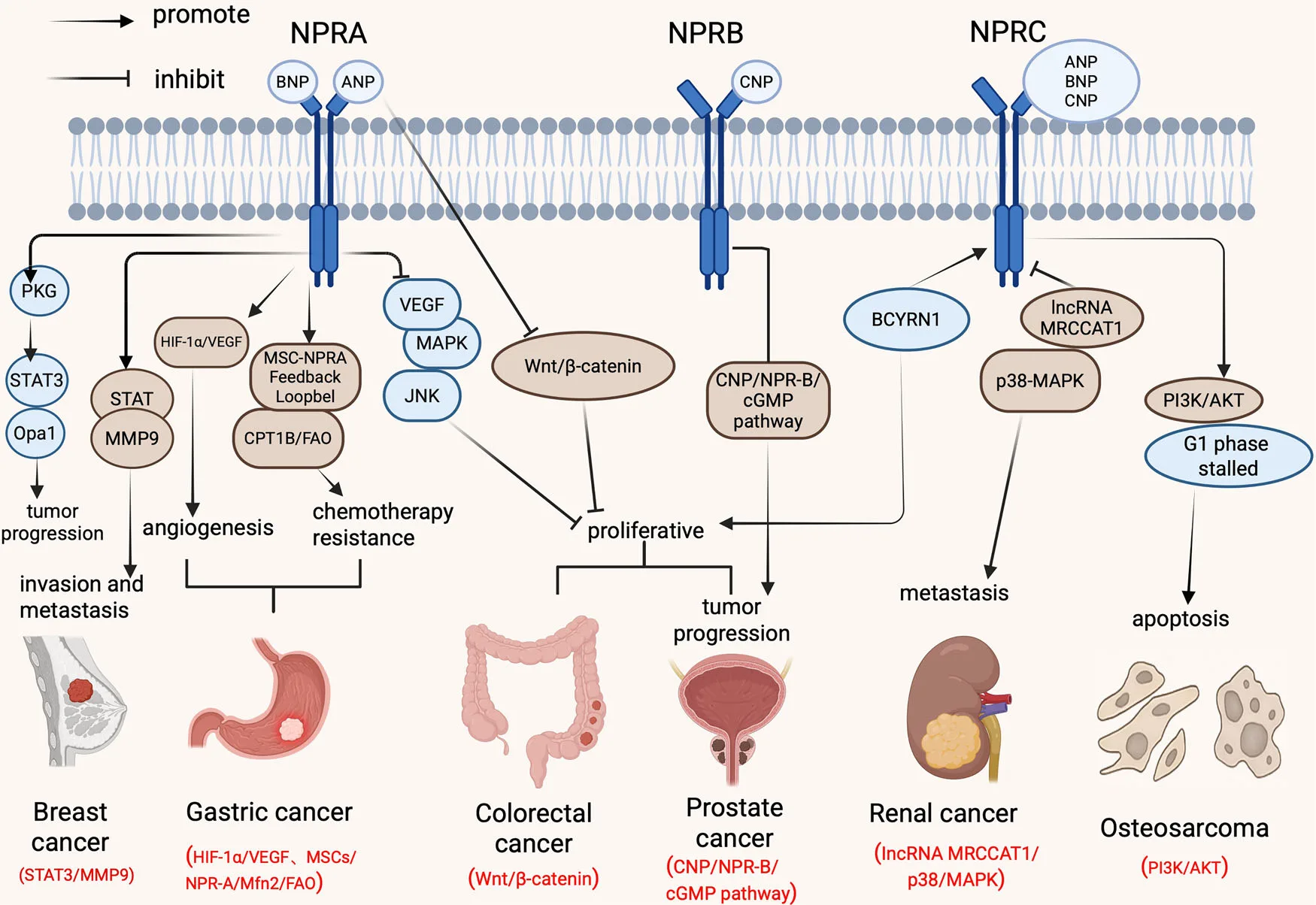
From a clinical standpoint, these receptors are emerging as biomarkers with real diagnostic and prognostic value. High NPRA expression correlates with advanced stage and poor prognosis in gastric and esophageal cancers, making it a potential marker for patient stratification. NPRC, on the other hand, is associated with improved survival in renal cancers and is being developed as a molecular imaging target in prostate cancer. Circulating fragments such as NT-proCNP may even pave the way for liquid biopsy applications, enabling non-invasive early detection and monitoring.
Therapeutically, the outlook is equally promising. NPRA inhibitors could suppress angiogenesis, invasion, and chemoresistance. NPRC agonists like C-ANP4-23 are already demonstrating anti-tumor activity in preclinical models. Meanwhile, engineered CNP derivatives such as dCNP show great potential as adjuvant therapies, remodeling the tumor microenvironment to enhance chemotherapy, immunotherapy, and radiotherapy.
Looking ahead, future research must focus on tumor-specific targeting strategies. Since receptor functions vary widely depending on cancer type, a one-size-fits-all approach is unlikely. Instead, precision oncology approaches—integrating multi-omics data, receptor profiling, and nanodelivery systems—could unlock their full therapeutic potential.
Ultimately, natriuretic peptide receptors are more than cardiovascular regulators; they represent a new frontier in oncology, bridging molecular signaling with the next generation of personalized cancer care.
Reference
Quan, C., Shao, W., Yang, Y., Yao, Q., Yang, Z., & Yao, Z. (2025). The role and clinical value of natriuretic peptide receptor family in malignant tumor. Cell Death Discovery, 11(1), 412.
https://doi.org/10.1038/s41420-025-02656-w
Wang, X., Raulji, P., Mohapatra, S. S., Patel, R., Hellermann, G., Kong, X., … & Mohapatra, S. (2011). Natriuretic peptide receptor a as a novel target for prostate cancer. Molecular cancer, 10(1), 56.
https://doi.org/10.1186/1476-4598-10-56
De Bold, A. J., Borenstein, H. B., Veress, A. T., & Sonnenberg, H. (1981). A rapid and potent natriuretic response to intravenous injection of atrial myocardial extract in rats. Life sciences, 28(1), 89-94.
https://doi.org/10.1016/0024-3205(81)90370-2
Chinkers, M., Garbers, D. L., Chang, M. S., Lowe, D. G., Chin, H., Goeddel, D. V., & Schulz, S. (1989). A membrane form of guanylate cyclase is an atrial natriuretic peptide receptor. Nature, 338(6210), 78-83.
 LinkPeptide
LinkPeptide