Peptide-Functionalized Hydrogels Offer New Hope for Critical Limb Ischemia
Abstract
Critical limb ischemia (CLI) remains one of the most challenging complications of peripheral artery disease, often leading to tissue death and amputation. While endothelial cell therapy holds great promise for restoring blood flow, its effectiveness has been limited by poor cell survival in hostile ischemic environments. Recent research highlights a novel solution: thermoresponsive peptide-functionalized hydrogels made from poly(polyethylene glycol citrate-co-N-isopropylacrylamide) (PPCN). These hydrogels not only protect cells during injection but also provide bioactive cues that enhance survival and function. Among the peptides tested, laminin-derived A5G81 improved cell adhesion and proliferation, while VEGF-mimicking QK promoted angiogenesis. In a mouse model of CLI, these biomaterials significantly improved cell retention, blood perfusion, skeletal muscle regeneration, and functional recovery, with QK showing particularly strong benefits. This approach represents a promising step toward more effective, minimally invasive regenerative therapies for CLI and other ischemic diseases.
Introduction: A Silent Threat to Limbs and Lives
Critical limb ischemia (CLI) is one of the most severe outcomes of peripheral artery disease, where drastically reduced blood flow to the legs and feet triggers chronic pain, non-healing wounds, tissue death, and, in many cases, amputation. For patients, the diagnosis is life-changing — the risk of long-term disability or limb loss is high, and treatment options remain limited.
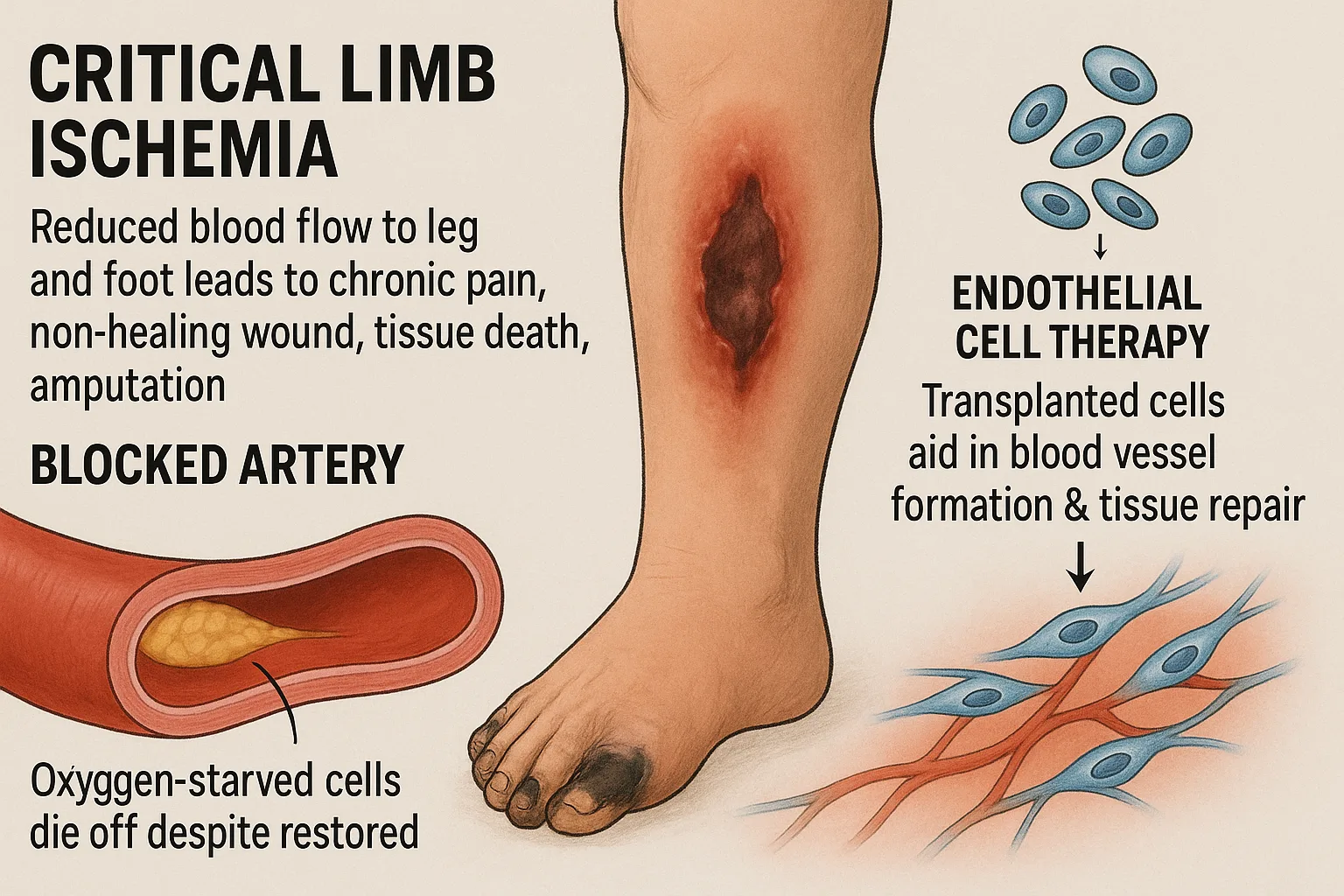
Standard approaches such as surgical revascularization or drug therapies can restore some circulation, but they rarely succeed in reversing the profound tissue damage caused by oxygen deprivation. These interventions address the “plumbing problem” of blocked arteries but do little to rescue the fragile cells that quickly deteriorate in an ischemic environment. Consequently, even with aggressive treatment, many patients continue to suffer progressive tissue loss.
This gap has pushed researchers toward regenerative medicine. By transplanting endothelial cells, which are central to forming new blood vessels, scientists hope to spark tissue repair and revascularization. Yet one critical barrier persists: ensuring these cells survive long enough to restore function in oxygen-starved tissue.
Endothelial Cells: Nature’s Architects of Blood Vessels
At the heart of tissue repair lies the body’s ability to rebuild its vascular network. Endothelial cells (ECs) — the cells that line blood vessels — are essential for this process. They guide angiogenesis, the formation of new vessels, which restores circulation and delivers oxygen and nutrients to damaged tissue. Without them, regeneration stalls, and tissue death accelerates.
This makes EC transplantation a compelling approach for conditions like CLI, where revascularization is vital. By introducing healthy ECs into ischemic tissue, researchers aim to jumpstart vessel growth and support the body’s natural healing mechanisms. However, the hostile environment of oxygen-starved and inflamed tissue presents a formidable challenge. Transplanted cells often die or fail to engraft, limiting their therapeutic potential. Overcoming this survival bottleneck is therefore the key to unlocking the full promise of endothelial cell–based therapies.
PPCN Hydrogels: A Smarter Scaffold for Healing
To address the survival challenge, researchers turned to biomaterials — specifically, hydrogels — as protective carriers for cell delivery. Hydrogels act as soft, water-rich scaffolds that cushion transplanted cells, shield them from mechanical stress, and help them remain in place at the site of injury. Yet not all hydrogels are created equal, and many fail to provide the biochemical cues needed for long-term cell survival.
This is where poly(polyethylene glycol citrate-co-N-isopropylacrylamide) (PPCN) comes in. PPCN is a thermoresponsive hydrogel, meaning it flows like a liquid at room temperature but solidifies into a gel at body temperature. This property allows clinicians to inject cells in a minimally invasive way, after which the material quickly stabilizes into a supportive matrix inside the tissue.
Beyond its mechanical function, PPCN also carries antioxidant and anti-inflammatory properties, giving transplanted cells additional protection against the harsh ischemic environment. This dual capability — mechanical support and biochemical defense — makes PPCN a uniquely promising platform for regenerative medicine.
Peptide Power: Guiding Cells with A5G81 and QK
While PPCN provides a strong foundation for cell delivery, its real breakthrough comes from peptide functionalization. By embedding short bioactive peptide sequences into the hydrogel, researchers created a microenvironment that not only protects transplanted cells but actively supports their survival and function.
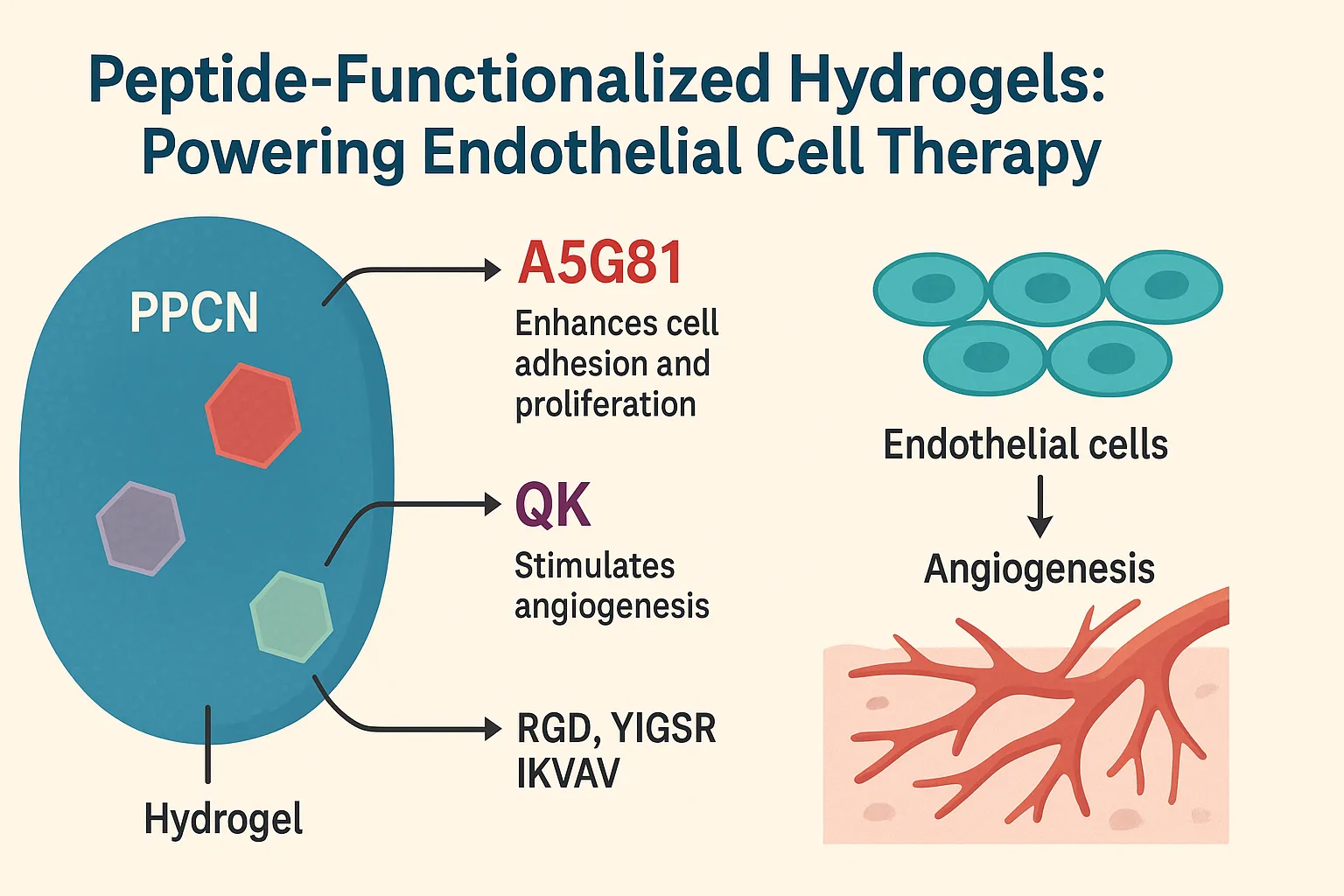
Among the peptides tested, two stood out: A5G81 and QK.
- A5G81, derived from the protein laminin, enhances cell adhesion and proliferation. In simple terms, it helps endothelial cells anchor securely to the hydrogel matrix and replicate, improving their chances of forming stable vascular structures.
- QK, modeled on the sequence of vascular endothelial growth factor (VEGF), mimics the effects of this powerful growth factor by stimulating angiogenesis. Essentially, QK tells endothelial cells to start building new blood vessels — a critical step for restoring circulation in ischemic tissue.
Other peptides, such as RGD, YIGSR, and IKVAV, were also tested but showed less impact on cell survival in this study. The combination of mechanical protection from PPCN and biochemical guidance from A5G81 and QK provided the transplanted cells with exactly what they needed: safety during injection, stability after placement, and signals to grow and function.
This synergy marks a significant step forward in designing biomaterials that do more than carry cells — they actively participate in regeneration.
From Lab Bench to Living Tissue: Proof in Action
The true test of PPCN hydrogels came in the lab. Researchers first examined their effects in vitro with human umbilical vein endothelial cells (HUVECs). On standard PPCN hydrogels, cells struggled to spread, remaining rounded and inactive. But when modified with A5G81 or QK, endothelial cells rapidly adopted a well-spread shape, proliferated robustly, and survived for longer periods in three-dimensional culture.
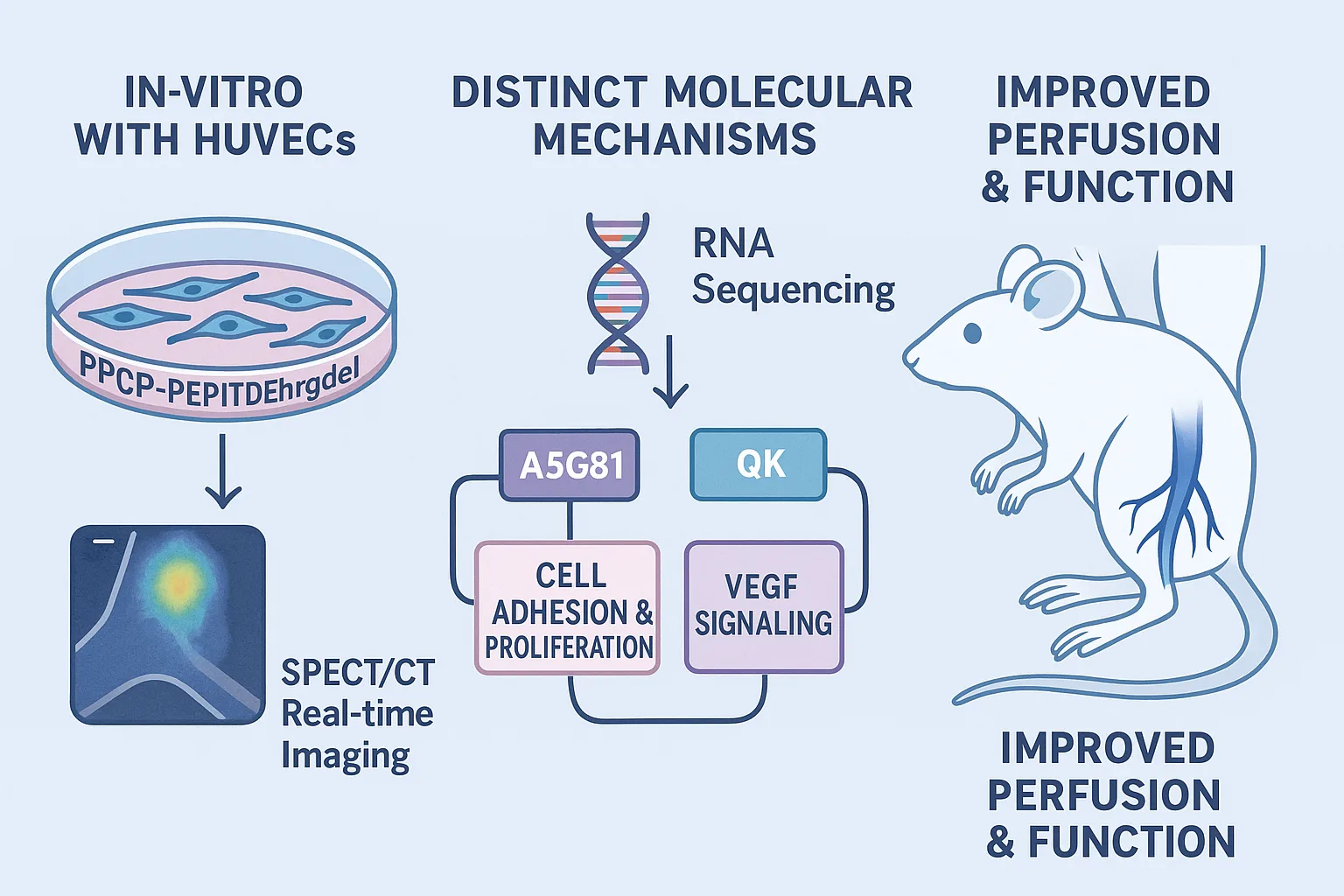
Interestingly, each peptide worked through different molecular mechanisms. RNA sequencing revealed that A5G81 triggered pathways linked to cell adhesion and DNA replication, supporting early growth and stability. In contrast, QK activated the VEGF signaling cascade, a direct driver of angiogenesis. Together, these insights showed that PPCN-peptide hydrogels could be tuned to influence cell behavior in complementary ways.
The real challenge, however, was testing these materials in living organisms. Using a murine hindlimb ischemia model — a well-established mimic of human CLI — the team injected endothelial cells with different hydrogel formulations. To track their fate, cells were genetically modified to carry a sodium iodide symporter, allowing real-time imaging via SPECT/CT.
The results were striking. Cells delivered with PBS or unmodified PPCN quickly disappeared, with little signal detected after a few days. In contrast, cells encapsulated in PPCN-A5G81 or PPCN-QK survived up to four weeks, maintaining strong signals long after controls had failed. Even more importantly, this translated into tangible benefits: mice treated with peptide-functionalized hydrogels showed improved blood perfusion, reduced tissue necrosis, and better limb function compared to all other groups.
These findings underscored a critical point — PPCN hydrogels didn’t just carry cells; they created a protective, instructive environment that enabled them to thrive and heal.
Beyond Blood Flow: Healing the Muscles Too
Critical limb ischemia is often described as a vascular disease, but its damage extends beyond blood vessels. When oxygen and nutrients fail to reach skeletal muscle, the tissue undergoes fibrosis, fat infiltration, and atrophy. For patients, this translates into reduced mobility, weakness, and long-term disability — problems that revascularization alone cannot solve.
In this study, the researchers took a closer look at how peptide-functionalized PPCN hydrogels affected skeletal muscle recovery. In both the tibialis anterior and gastrocnemius muscles of treated mice, those receiving PPCN-QK or PPCN-A5G81 with endothelial cells displayed markedly healthier tissue. Muscle fibers were larger and more mature, fibrosis was reduced, and signs of fat infiltration were far less pronounced compared to control groups.
Among the two, QK-modified hydrogels had the strongest effect, consistent with their ability to stimulate VEGF-driven angiogenesis and improve blood supply. Importantly, this dual impact — rebuilding vasculature while preserving muscle — highlights the holistic potential of PPCN hydrogels to restore not just circulation but also function in ischemic limbs.
Toward the Clinic: What This Means for Future Therapies
The promise of peptide-functionalized PPCN hydrogels extends well beyond the lab bench. By combining mechanical protection, antioxidant support, and biochemical signaling, this platform addresses the major barriers that have historically limited cell-based therapies in ischemic disease. For patients with CLI, this could mean a minimally invasive treatment that not only restores blood flow but also preserves muscle function, reducing the risk of amputation and long-term disability.
Another significant aspect of this study is the recognition of sex-specific differences. Female mice exhibited slower recovery and more severe muscle damage, mirroring patterns observed in clinical cases. This suggests that regenerative strategies may need to be tailored for men and women, paving the way for sex-specific biomaterials that optimize outcomes across patient populations.
Looking ahead, the versatility of PPCN hydrogels makes them adaptable to other conditions characterized by ischemia, such as heart attacks, strokes, and chronic wounds. Their tunable design means additional peptides or growth factors could be incorporated, opening the door to a new generation of customizable regenerative therapies.
Conclusion: A New Chapter in Regenerative Medicine
This research highlights a powerful innovation in regenerative medicine: thermoresponsive, peptide-functionalized PPCN hydrogels that dramatically improve the survival and function of transplanted endothelial cells. By pairing mechanical protection with bioactive signals, these materials foster both vascular regeneration and muscle repair, tackling the dual challenges of CLI.
The findings not only point to a promising therapy for limb salvage but also showcase the broader potential of bioactive hydrogels in treating a range of ischemic conditions. With further refinement, such materials could redefine the future of cell-based medicine.
Reference
Wang, H., Dang, C., Rivera‐Bolanos, N., Tong, O., Wang, X., Chen, G., … & Jiang, B. (2025). Prolonging Endothelial Cell Survival with Thermoresponsive Peptide‐Functionalized Hydrogels Improves Ischemic Limb Tissue Regeneration and Function. Advanced Functional Materials, 35(10), 2416553.
https://doi.org/10.1002/adfm.202416553
Criqui, M. H., Matsushita, K., Aboyans, V., Hess, C. N., Hicks, C. W., Kwan, T. W., … & Ujueta, F. (2021). Lower extremity peripheral artery disease: contemporary epidemiology, management gaps, and future directions: a scientific statement from the American Heart Association. Circulation, 144(9), e171-e191.
https://doi.org/10.1161/CIR.0000000000001005
Andukuri, A., Minor, W. P., Kushwaha, M., Anderson, J. M., & Jun, H. W. (2010). Effect of endothelium mimicking self-assembled nanomatrices on cell adhesion and spreading of human endothelial cells and smooth muscle cells. Nanomedicine: Nanotechnology, Biology and Medicine, 6(2), 289-297.
https://doi.org/10.1016/j.nano.2009.09.004
Jiang, B., Wang, X., Rivera-Bolanos, N., & Ameer, G. A. (2024). Generation of autologous vascular endothelial cells for patients with peripheral artery disease. Journal of Cardiovascular Translational Research, 17(3), 558-569.
 LinkPeptide
LinkPeptide