Oxyntomodulin: The Dual-Action Gut Hormone Shaping the Future of Metabolic Therapy
Abstract
Oxyntomodulin (OXM), a 37-amino-acid peptide derived from the proglucagon precursor, has long existed in the shadows of better-known incretin hormones like GLP-1. However, its ability to activate both the GLP-1 and glucagon receptors—albeit with modest affinity—has recently propelled it to the forefront of metabolic research. Originally identified for its role in inhibiting gastric acid secretion, OXM is now recognized for its potential to suppress appetite, increase energy expenditure, and influence hepatic lipid metabolism. These properties have inspired the development of a new class of dual and triple agonists aimed at treating obesity, type 2 diabetes, and metabolic dysfunction-associated steatotic liver disease (MASLD). Though still under clinical investigation, OXM’s legacy as a pharmaceutical prototype underscores its value—not merely as a hormone, but as a roadmap for designing next-generation metabolic therapies that balance efficacy with tolerability.
Old Molecule, New Promise: Why Oxyntomodulin Matters in Modern Metabolic Medicine
In the vast landscape of gut-derived hormones, few have received as much attention as glucagon-like peptide-1 (GLP-1)—a key player in modern treatments for type 2 diabetes and obesity. But nestled within the same proglucagon lineage lies a lesser-known molecule with remarkable potential: oxyntomodulin (OXM).
Once considered a physiological footnote, OXM is now stepping into the spotlight. This 37-amino-acid peptide, secreted by intestinal L-cells, is structurally unique in that it contains the full sequence of glucagon along with an extension that partially mimics GLP-1. As a result, OXM can bind to both the glucagon receptor and the GLP-1 receptor—a rare dual-receptor activity that hints at powerful, multifaceted effects.
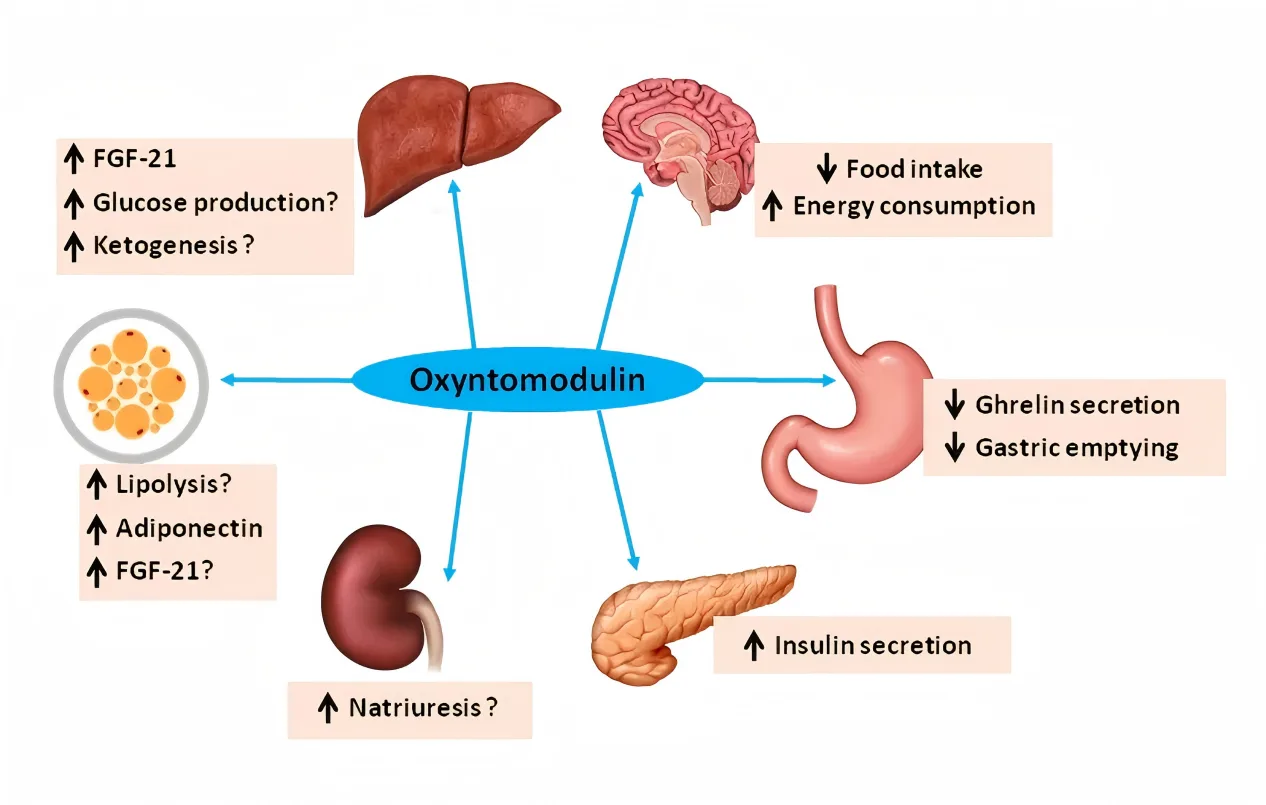
Recent studies have shown that oxyntomodulin can reduce appetite, enhance energy expenditure, and influence lipid metabolism. These properties make it more than a biological curiosity—they make it a promising blueprint for the development of advanced metabolic therapies. In fact, its dual-action profile has inspired a new class of investigational drugs known as glucagon–GLP-1 co-agonists, designed to harness the synergistic benefits of both hormones.
In this post, we’ll explore how oxyntomodulin evolved from an obscure gut peptide to a catalyst for pharmaceutical innovation. We’ll examine its discovery, physiological significance, and its growing influence in the design of next-generation therapies for obesity, type 2 diabetes, and metabolic liver disease. As the field of metabolic medicine advances, oxyntomodulin may well prove to be the hormone we didn’t know we needed.
From Gut Glucagon to Dual Agonist: The Fascinating Discovery of Oxyntomodulin
The story of oxyntomodulin begins in the early days of endocrinology, when researchers first noted that glucagon-like immunoreactivity (GLI) wasn’t exclusive to the pancreas. In fact, this activity appeared prominently in the L-cells of the gastrointestinal tract, sparking a scientific mystery: were these cells producing glucagon, or something entirely different?
In the 1960s and ’70s, advances in radioimmunoassays and peptide chemistry gradually unraveled the truth. Researchers discovered that the glucagon-like material in the gut was not authentic pancreatic glucagon, but larger and structurally distinct molecules. Two such peptides stood out: glicentin, a 69-amino-acid peptide containing the full glucagon sequence, and a shorter fragment—later named oxyntomodulin—comprising 37 amino acids. This smaller peptide retained the entire glucagon structure and part of the glicentin sequence, positioning it as a unique bioactive fragment rather than a simple degradation product.
The name “oxyntomodulin” was proposed by French scientist Dominique Bataille, who observed that the peptide inhibited acid secretion in oxyntic glands of the stomach—glands responsible for producing hydrochloric acid. Though the name suggested a gastric role, further research revealed that its biological significance might extend far beyond the stomach.
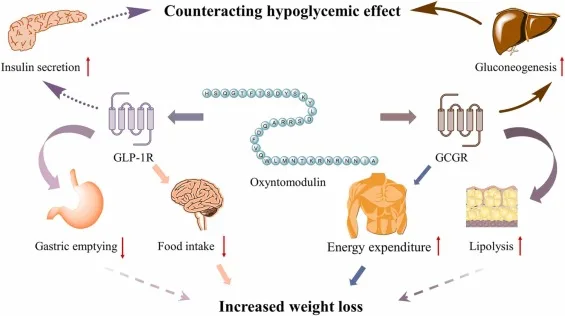
By the 1980s, oxyntomodulin had been structurally characterized and shown to be secreted from intestinal L-cells in response to food intake, particularly carbohydrates. Yet, what truly set it apart was its dual receptor activity: OXM could bind both the glucagon receptor (GCGR) and the GLP-1 receptor (GLP-1R), though with much lower affinity than their native ligands.
This duality raised an intriguing possibility—could one molecule mimic the effects of both glucagon and GLP-1? At the time, the idea was novel. But as the understanding of incretins and energy regulation deepened, oxyntomodulin’s potential as a natural co-agonist began to attract serious interest.
What started as a curious gut peptide—often overshadowed by its more famous relatives—was slowly gaining recognition. Scientists began to see oxyntomodulin not just as a physiological relic, but as a template for future therapeutics. Its unique structure and modest activity hinted at a larger story, one that was just beginning to unfold.
Small but Mighty: How Oxyntomodulin Regulates Appetite, Energy, and Digestion
While its structure hinted at potential, the real intrigue around oxyntomodulin began with studies of its biological activity. Early experiments confirmed that OXM wasn’t just structurally unique—it also played a meaningful role in human physiology, especially in regulating digestion, glucose metabolism, and appetite.
A Modest but Multifaceted Hormone
In both animal and human models, oxyntomodulin was shown to inhibit gastric acid secretion. In one clinical study, intravenous infusion of synthetic OXM at physiological doses significantly reduced pentagastrin-stimulated acid output by up to 76%, mirroring the effects of GLP-1. It also modestly stimulated insulin and C-peptide release, leading some researchers to propose that OXM functions as both an incretin and enterogastrone—a hormone that inhibits gastrointestinal secretions.
Yet, the most striking effects of oxyntomodulin appeared in the realm of appetite regulation and energy balance.
Appetite Suppression and Weight Loss
Building on findings from GLP-1 research, investigators tested the effect of OXM on the central nervous system. In rodents, intracerebroventricular injections of oxyntomodulin into the hypothalamus powerfully suppressed food intake. The effects were comparable to GLP-1 and could be blocked by GLP-1 receptor antagonists, suggesting that OXM acts—at least in part—through the same neuronal circuits.
Human studies soon followed. In a landmark 2003 trial, infusions of oxyntomodulin reduced calorie intake by 19%, without causing discomfort or nausea. A follow-up study in 2005 found that subcutaneous injections administered three times daily over four weeks led to a significant 2.3 kg weight loss in overweight individuals. Intriguingly, a separate study showed that OXM not only decreased energy intake but also increased energy expenditure, largely through enhanced physical activity.
Circulating Levels and Measurement Challenges
Despite these promising effects, oxyntomodulin’s physiological relevance remains a topic of debate. Its plasma concentrations are typically low—about 1–20 pmol/L even after meals—and measuring it accurately is difficult. Most assays struggle to distinguish OXM from glicentin or glucagon, requiring complex chromatography to isolate true levels.
Furthermore, OXM binds to the glucagon receptor with only ~2% of glucagon’s affinity and to the GLP-1 receptor with 50–100 times lower affinity than GLP-1. No unique receptor for oxyntomodulin has ever been identified, raising questions about its evolutionary role. Is it a true hormone, or merely a byproduct of proglucagon processing?
What’s clear, however, is that when administered exogenously, oxyntomodulin has potent effects on appetite, energy use, and body weight. And these effects, though modest in their natural form, have sparked a revolution in drug design.
Blueprint for Breakthroughs: How Oxyntomodulin Inspired Glucagon–GLP-1 Co-Agonists
Oxyntomodulin’s modest natural potency might have relegated it to biological obscurity—until pharmaceutical scientists recognized its hidden superpower: dual receptor activity. Though OXM binds weakly to both glucagon and GLP-1 receptors, its ability to simultaneously engage two critical metabolic pathways became the blueprint for a new class of therapeutics: glucagon–GLP-1 co-agonists.
The rationale behind co-agonism is simple but powerful. GLP-1 agonists reduce appetite, enhance insulin secretion, and slow gastric emptying. Glucagon, while classically viewed as a hyperglycemic hormone, also suppresses appetite, stimulates energy expenditure, and profoundly modulates hepatic lipid metabolism. When used together—or mimicked by a single co-agonist—they produce synergistic effects: greater weight loss, improved lipid profiles, and enhanced metabolic flexibility.
This concept took off in the late 2000s. Researchers like Day et al. and Pocai et al. developed long-acting dual agonists using structural modifications (e.g., pegylation or lipidation) to prolong activity. In animal models, these compounds led to dramatic weight loss, driven by both reduced food intake and elevated energy use. Moreover, they significantly improved hepatic steatosis, making them ideal candidates for treating metabolic dysfunction-associated steatotic liver disease (MASLD).
Several co-agonists are now in advanced clinical development:
- Survodutide (Zealand/Boehringer-Ingelheim): Now in Phase 3 trials for type 2 diabetes, obesity, and MASH.
- Mazdutide (Innovent/Lilly): Achieved up to 14% weight loss and 80% liver fat reduction over 48 weeks.
- Efinopegdutide and Pemvidutide: Demonstrated promising outcomes in fat reduction and glucose control.
- Retatrutide, a triple agonist (GLP-1, glucagon, GIP), delivered over 24% weight loss in a Phase 2 trial but with notable side effects.
Yet, these therapies are not without risk. Common adverse effects include nausea, elevated heart rate, and potential loss of lean body mass, likely due to glucagon’s impact on amino acid metabolism. Balancing efficacy with tolerability remains the core challenge for co-agonist therapies moving forward.
Still, the message is clear: oxyntomodulin’s dual-action profile has reshaped the metabolic drug landscape, transforming a once-overlooked peptide into a pharmaceutical muse for the 21st century.
What’s Next for Oxyntomodulin? Promise, Caution, and the Path Forward
Oxyntomodulin’s story is a testament to how scientific curiosity and persistence can elevate a forgotten molecule into a model for innovation. Although no glucagon–GLP-1 co-agonist has yet received regulatory approval (as of early 2025), the clinical momentum is undeniable. With multiple candidates in late-stage trials, including Survodutide, Mazdutide, and Retatrutide, approval now seems a question of “when”, not “if.”
Yet, the path forward demands caution. As these therapies grow more potent, managing side effects—particularly gastrointestinal distress, cardiovascular effects, and lean mass loss—will be critical. Dosing strategies, patient-specific tailoring, and nutritional support (e.g., protein supplementation) may help mitigate these risks.
More broadly, oxyntomodulin challenges our understanding of what defines a “useful” hormone. Despite its low natural abundance and lack of a unique receptor, its dual receptor activity has catalyzed some of the most promising developments in obesity and liver disease treatment.
In the end, OXM’s greatest contribution may not be its direct clinical use, but rather the inspiration it provides: that biological nuance can unlock therapeutic breakthroughs, even decades after a molecule’s discovery.
 LinkPeptide
LinkPeptide