Redefining Bone Health: A New Era with Selective Androgen Receptor Modulators
Osteoporosis, a widespread and debilitating condition, remains underdiagnosed and undertreated, especially in aging populations. Traditional therapies—while effective for some—carry significant risks, including hormonal imbalances, cardiovascular complications, and low adherence. Selective Androgen Receptor Modulators (SARMs) offer a promising alternative. Engineered for tissue-specific action, SARMs selectively activate androgen receptors in bone and muscle without affecting reproductive tissues, minimizing side effects commonly associated with testosterone-based therapies. This blog explores the molecular mechanisms behind SARMs, their clinical development in treating osteoporosis, and the medicinal chemistry innovations driving their selectivity and safety. Though not yet FDA-approved, SARMs like Enobosarm and LGD-4033 have shown promising results in early trials. With continued research, SARMs may revolutionize osteoporosis care by providing safer, more targeted, and orally available treatments for patients in need. The era of precision bone health is fast approaching.
The Hidden Burden: Why Osteoporosis Needs a Smarter Solution
Osteoporosis is often called a “silent disease” for a reason—it develops gradually and painlessly, usually going unnoticed until a sudden fracture reveals its devastating impact. Characterized by the progressive loss of bone density and structural integrity, osteoporosis significantly increases the risk of fractures, particularly in the spine, hips, and wrists. Globally, this condition affects an estimated 200 million individuals, predominantly postmenopausal women and older adults. Each year, it contributes to over 8.9 million fractures, yet awareness and proactive treatment remain startlingly low.
Current therapeutic options include bisphosphonates (such as alendronate and zoledronic acid), monoclonal antibodies like denosumab, and parathyroid hormone analogs. While these drugs can be effective, they are far from perfect. Many require strict dosing schedules, can cause gastrointestinal issues, and in rare cases, are linked to severe adverse effects such as osteonecrosis of the jaw or atypical femoral fractures. Hormonal therapies using testosterone or estrogen offer benefits but carry their own risks, including cardiovascular complications, liver toxicity, and increased cancer susceptibility.
The search for better alternatives has led researchers to explore Selective Androgen Receptor Modulators (SARMs)—a novel class of compounds that could reshape the treatment landscape. SARMs are designed to mimic the bone- and muscle-strengthening effects of traditional androgens, but with tissue-selective activity that minimizes side effects in non-target organs. With their favorable oral bioavailability and reduced androgenic risks, SARMs represent a compelling step forward in osteoporosis therapy.
As the global population continues to age, the demand for safer, more effective treatments will only intensify. SARMs may be the answer—offering a way to rebuild bone strength without compromising overall health.
Smarter Molecules: How SARMs Redefine Hormonal Therapy
To understand the appeal of Selective Androgen Receptor Modulators (SARMs), it helps to begin with their biological target: the androgen receptor (AR). This receptor is a type of nuclear hormone receptor that, when activated, influences the expression of genes involved in muscle and bone growth. Androgens like testosterone are natural AR activators, but their broad activity across multiple tissues often leads to side effects when used therapeutically.
SARMs were developed to address this problem. Unlike traditional androgens, SARMs are tissue-selective modulators, engineered to activate the AR only in anabolic tissues—namely bone and muscle—while sparing androgenic tissues such as the prostate, skin, and reproductive organs. This selectivity is what sets SARMs apart and gives them immense therapeutic potential.
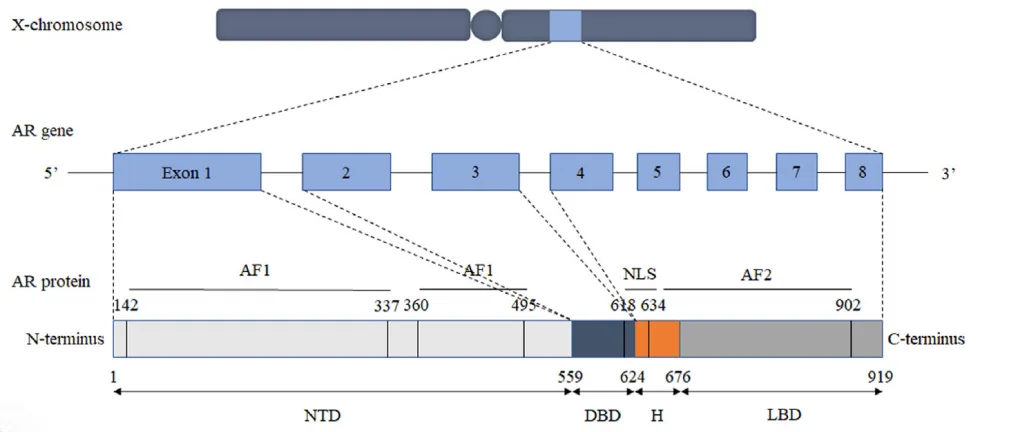
Here’s how they work: SARMs enter cells and bind to the ligand-binding domain (LBD) of the androgen receptor. This interaction initiates a cascade of molecular events. The AR, now activated by the SARM, dissociates from heat shock proteins and translocates into the cell nucleus. Once there, it binds to androgen response elements (AREs) in DNA, recruiting coactivators that turn on genes involved in bone formation, muscle maintenance, and tissue repair.
Importantly, SARMs show excellent oral bioavailability, avoiding the need for injections or patches like traditional testosterone therapies. They also exhibit partial agonism in non-target tissues, which helps reduce risks like prostate enlargement or virilization in women.
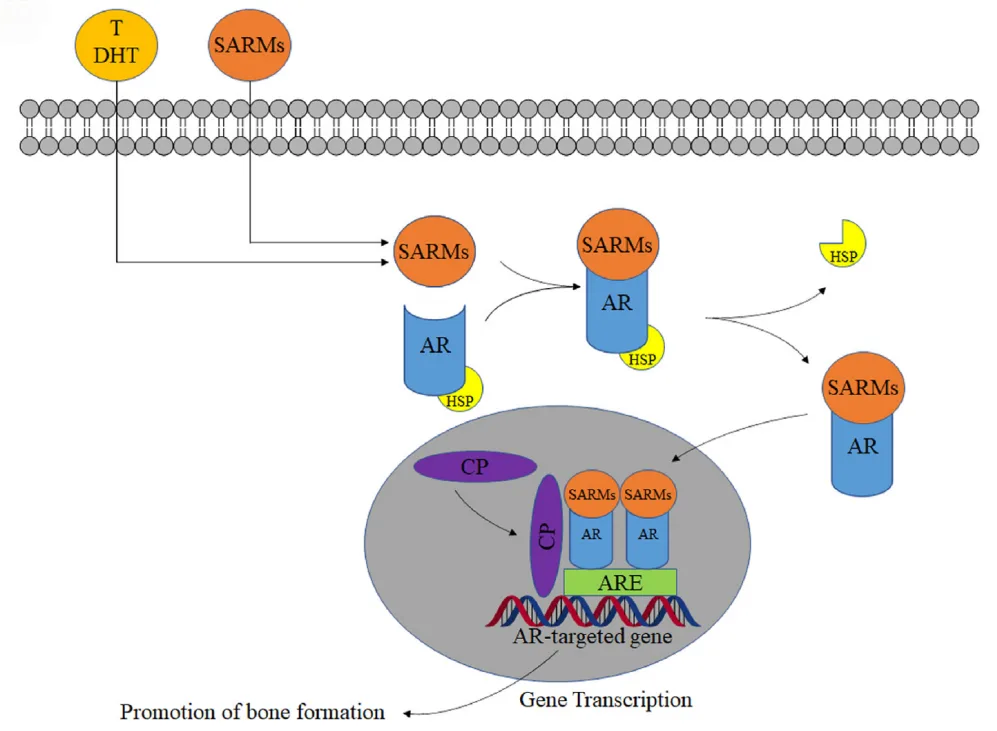
In the context of osteoporosis, SARMs offer a strategic advantage: they stimulate osteoblast activity and enhance bone mineral density without the collateral damage of conventional hormonal treatments. This makes them especially promising for vulnerable populations such as elderly men with low testosterone and postmenopausal women, who are often underserved by current treatment modalities.
By combining molecular precision with therapeutic power, SARMs represent a new generation of agents poised to redefine how we treat skeletal disorders.
From Lab Bench to Clinic: The Journey of SARMs in Osteoporosis Trials
Over the past two decades, researchers have made remarkable strides in developing SARMs with the potential to treat osteoporosis and other muscle-wasting conditions. While hundreds of SARM candidates have been synthesized and tested, only a select few have advanced into clinical trials—a critical step toward regulatory approval.
Among the most promising candidates is Enobosarm (also known as Ostarine or GTx-024), a non-steroidal SARM currently in Phase III clinical trials. Initially developed for muscle wasting in cancer patients, Enobosarm has shown favorable effects on bone mineral density and lean muscle mass, making it a strong contender for osteoporosis treatment. In animal models and early human trials, it demonstrated a high anabolic-to-androgenic ratio, preserving muscle and bone while avoiding typical androgenic effects like prostate enlargement.
MK-0773, developed by Merck, is another notable compound. This molecule completed Phase II trials, showing significant improvement in muscle function and a moderate increase in bone density in elderly women with sarcopenia. Although its development has slowed, MK-0773 remains a proof of concept that selective AR activation can yield clinically meaningful outcomes.
LGD-4033 (Ligandrol) and RAD-140 (Testolone) are two additional SARMs that have garnered attention. LGD-4033 is in Phase II trials and has demonstrated excellent oral bioavailability and tissue-selective activity. RAD-140, currently in Phase I, has shown potent anabolic effects in preclinical models, particularly in restoring muscle mass and improving bone architecture.
However, not all candidates have made it to the finish line. GSK-2881078 and PF-06260414, for instance, were discontinued due to side effects or insufficient efficacy. Some adverse effects, such as elevated liver enzymes, cardiovascular risks, or altered lipid profiles, have raised concerns during human testing. In rare cases, SARMs have been linked to drug-induced liver injury, reinforcing the importance of long-term safety evaluation.
Despite these hurdles, the selective nature of SARMs continues to fuel interest. They are widely recognized for preserving lean mass, improving bone strength, and enhancing functional performance—all without the full range of side effects seen in traditional hormone therapies. For osteoporosis patients, especially those unable or unwilling to undergo estrogen or testosterone-based treatments, SARMs may offer a more balanced and tolerable solution.
With several candidates still in the pipeline and new scaffolds emerging through ongoing research, the clinical journey of SARMs is far from over. Their progress so far, though uneven, signals a potential breakthrough in musculoskeletal medicine.
Designing Precision: The Chemistry Behind Selectivity and Safety
At the heart of every promising SARM lies a carefully crafted molecular design. Unlike traditional androgens, which often trigger unwanted effects due to their broad activity profile, SARMs are engineered for precision—activating the androgen receptor (AR) only where it matters most: muscle and bone. This selectivity doesn’t happen by chance; it’s the result of structure-activity relationship (SAR) studies, sophisticated modeling, and iterative chemical refinement.
SARMs can be broadly classified into steroidal and non-steroidal types. Early SARMs were steroidal derivatives of testosterone or dihydrotestosterone (DHT), offering improved oral bioavailability but still causing androgenic side effects. The real breakthroughs came with non-steroidal SARMs, which introduced a rich diversity of chemical scaffolds, allowing for fine-tuned biological responses.
Some of the most studied non-steroidal scaffolds include:
- N-arylpropanamides (e.g., Enobosarm)
- Diarylhydantoins
- Bicyclic hydantoins
- 2-Quinolinones
- Tetrahydroquinolines (THQ)
Each class brings unique features to the table. For example, the N-arylpropanamide scaffold was born from modifying the AR antagonist bicalutamide, flipping its activity from antagonist to selective agonist. Substituents like cyano (–CN) or nitro (–NO₂) groups enhance receptor binding through hydrogen bonding with key residues such as Arg752 and Gln711 within the ligand-binding pocket (LBP) of AR.
These interactions have been visualized through X-ray crystallography, which has played a pivotal role in guiding SARM development. Studies reveal that subtle changes—such as shifting a hydroxyl group, adding a trifluoromethyl moiety, or introducing a rigid bicyclic core—can significantly influence binding affinity, agonist potency, and tissue selectivity.
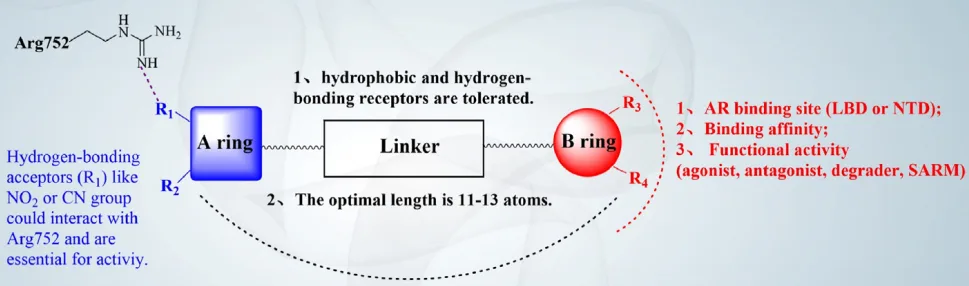
Design strategies often aim to:
- Maximize binding efficiency in anabolic tissues.
- Minimize activation in androgenic tissues like the prostate.
- Improve oral absorption and metabolic stability.
- Avoid hepatic enzyme induction or toxic metabolites.
Medicinal chemists also pay close attention to lipophilic ligand efficiency (LipE), aiming to balance hydrophobic interactions with aqueous solubility—an essential factor for drug-like behavior. For example, certain 2-quinolinone derivatives were found to enhance both bone-forming activity and pharmacokinetic properties by optimizing substituent size and polarity.
This deep dive into molecular architecture has allowed researchers to fine-tune SARMs down to the atomic level, ensuring they act more like a scalpel than a sledgehammer. In the pursuit of safer osteoporosis therapies, this chemical finesse is not just important—it’s absolutely essential.
A Future in Balance: Where SARMs Could Take Bone Health Next
Despite over two decades of research and hundreds of compounds tested, no SARM has yet gained FDA approval for the treatment of osteoporosis. But this is not a sign of failure—it’s a reflection of the complexity involved in developing highly selective, long-term safe therapeutics for chronic diseases. The science is sound. The clinical data, though still evolving, is promising. What lies ahead is the refinement of safety profiles, the completion of large-scale trials, and greater regulatory clarity.
A significant concern today is the unauthorized use of SARMs, especially among athletes and bodybuilders who obtain them through unregulated online sources. These black-market compounds often lack quality control and are marketed at dangerously high doses. While SARMs have fewer side effects than anabolic steroids, the risks—liver damage, cardiovascular issues, and hormonal disruption—remain real, particularly when misused. The FDA has issued warnings against their non-prescribed use, underscoring the importance of clinical oversight.
On the bright side, scientific momentum continues to build. Current research is exploring:
- Targeted delivery systems that increase localization to bone tissue.
- SARMs with dual action, combining anabolic activity with anti-resorptive effects.
- New scaffolds with enhanced metabolic stability and improved receptor selectivity.
- Potential use of SARMs in combination therapy alongside bisphosphonates or monoclonal antibodies.
As our global population ages, the burden of osteoporosis will rise. Millions stand to benefit from treatments that restore bone strength, improve mobility, and enhance quality of life—without the risks associated with traditional hormone therapies.
SARMs may not yet be on pharmacy shelves, but they represent one of the most compelling frontiers in osteoporosis drug development. With each new clinical trial, each structural insight, and each patient-focused innovation, we move closer to a future where selective androgen receptor modulation is no longer experimental—it’s standard care.
 LinkPeptide
LinkPeptide